| Nanoparticles of increasingly smaller particle size and various material compositions are being developed for the pharmaceutical, biomedical, electronics, optoelectronics, energy, catalyst and ceramics industries. These particles are formed, or alternatively dispersed after formation, in a liquid medium, typically water. Various non-aqueous continuous media such as alcohols are also used. The Importance of Particle Size and Particle Size Distribution The particle size and particle size distribution (PSD) of these materials are of great importance to the end user because they affect key colloid properties such as rheology, film gloss, surface area and packing density. Additionally, to prevent the aggregation of fine particles into much larger, undesirable units, steps must be taken to prevent particles from sticking together (aggregating) due to inter-particle collisions in the liquid medium. This can be accomplished by creating an interparticle electrical and/or steric energy barrier. For very fine particles, a combination of both electrical and steric barriers may be necessary to prevent aggregation. Zeta Potential The strength of the particle electrical barrier is measured in terms of an electrical potential termed the “zeta potential.” Zeta potential should be measured at process conditions, i.e., without prior sample dilution, as ions can adsorb or desorb from the particle surface upon dilution and change the zeta potential. Traditional Methods for measuring Zeta Potential Until recently, those wishing to obtain zeta potential results had to resort to tedious and time-consuming “microelectrophoresis” methods that require extreme dilutions and consequently stringent sample handling precautions. As particle sizes decrease to the nanometer size range, the light scattering methods employed in microelectrophoresis become increasingly difficult to use due to extreme Doppler broadening of the scattered light from the fine particles. Thus, detectors must be placed very close to the incident laser beam direction, requiring superior mechanical and environmental stability. Electroacoustic Zeta Potential Measurement However, a new instrument, the ESA-9800 from Matec Applied Sciences is changing this process. Developed during the last 15 years, the instrument employs electroacoustic principles that provide accurate measurements while preventing the zeta potential probe from being susceptible to environmental noise or vibrations. Electroacoustic Zeta Potential Measurement in Action The electroacoustic instrument measures the electrokinetic properties of a particle by an electroacoustic method. A high frequency electric field is applied to the dispersion, and the particles move electrophoretically in the applied field. If there is a density difference between the particle and the liquid, this motion will generate an alternating acoustic wave. The application of the electric field and the resulting acoustic wave provides a truly non-intrusive measurement and does not alter the properties of the sample. Evaluation of the Electroacoustic Zeta Potential System on a Titanium Dioxide System Tests were carried out on various materials to demonstrate the capabilities of the new instrument. Figure 1 shows the measured zeta potential of an 8.3% wt solids fine (~270 nanometer particle diameter) titanium dioxide system as a function of pH. The variation of electrical conductivity as a function of pH is also measured simultaneously during this automated “potentiometric titration.” This system has a zero of zeta potential (isoelectric point, IEP) at pH = 6.0. The location of the isoelectric point, in pH, is a characteristic of the particle surface and depends on the type of the metal oxide bond, the particle crystal structure, (rutile vs. anatase for titania) and on the type and level of impurities or other soluble species bonded to the titania particle surface. | 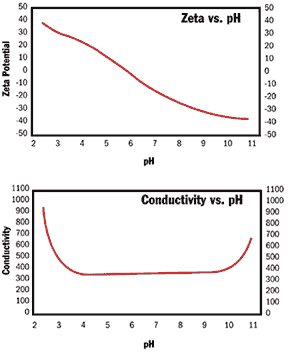
| | Figure 1. Measured zeta potential and conductivity of an 8.3%wt titania slurry as a function of pH. | Perhaps the single most important piece of information obtained from Figure 1 is that this titania system is unstable and will aggregate in the region of pH = 5 to pH = 7.3. This is because for particles in this size range (~270 nanometers), aggregation will occur in a concentrated system at a rapid rate for zeta potentials less than 20 millivolts in magnitude. For finer particles still, the zeta potential should be of even larger magnitude to prevent aggregation. In the extreme limit of very small nano-particles, sufficiently high zeta potentials cannot be achieved, and some level of steric stabilization is necessary. The results shown in Figure 1 can be obtained in concentrated systems (in the range of 0.1 to 50 volume %) by carrying out automatic pH titrations in conjunction with a computer-controlled burette on a vigorously stirred and/or flowing sample. Measurements can be made every thirty seconds while accumulating about 60 data points over the whole pH range. Evaluation of the Electroacoustic Zeta Potential System on a Silicon Dioxide System Figure 2 shows a potentiometric titration result on a 60-nanometer 10% wt silica particle system. For a pure silica surface, it is believed that the zeta potential approaches asymptotically an isoelectric point in the neighbourhood of pH = 2.0. The Si – OH2+ configuration, implying a positive silica surface, is believed not to exist for the silica surface. This idea seems to be supported by the results of Figure 2. For this very fine particle system, it is estimated that the region of stability against aggregation would start from about pH = 7.0 and go higher in pH. Increasing pH much above 10 will again result in a decrease in the magnitude of the zeta potential due to compression of the electric double layer. This occurs as the ionic strength increases with pH. Thus, the stability region is restricted to pH higher than 7 for a very fine silica particle system. | 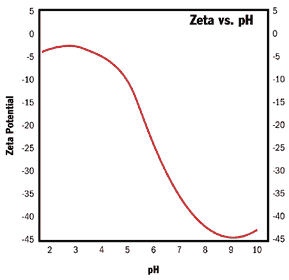
| | Figure 2. Zeta potential vs pH for a very fine 10% wt silica suspension. | The previous two examples display the utility of the zeta potential concept applied to particle stability considerations. Zeta potential measurements can also be used to determine the fractions of two different metals in mixed metal oxide catalyst particles, to determine the fraction of one crystal structure of a mixed structure metal oxide and many other applications too numerous to list here. |