Jul 18 2013
Researchers at the University of Basel have successfully developed artificial organelles that are able to support the reduction of toxic oxygen compounds. This opens up new ways in the development of novel drugs that can influence pathological states directly inside the cell. The results have been published in the Journal «Nano Letters».
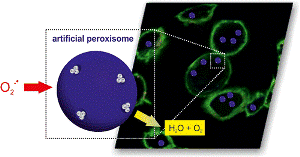 Inside the artificial peroxisome reactive oxygen radicals are being transformed into water (H2O) and oxygen (O2) via a reaction cascade with two enzymes. (Adapted with permission from Tanner et al. Nano Letters 2013 13 (6), 2875-2883. Copyright 2013 American Chemical Society.)
Inside the artificial peroxisome reactive oxygen radicals are being transformed into water (H2O) and oxygen (O2) via a reaction cascade with two enzymes. (Adapted with permission from Tanner et al. Nano Letters 2013 13 (6), 2875-2883. Copyright 2013 American Chemical Society.)
Free oxygen radicals are produced either as metabolic byproduct, or through environmental influences such as UV-rays and smog. Is the concentration of free radicals inside the organism elevated to the point where the antioxidant defense mechanism is overwhelmed, the result can be oxidative stress, which is associated with numerous diseases such as cancer of arthritis.
The aggressive molecules are normally controlled by endogenous antioxidants. Within this process, organelles located inside the cell, so-called peroxisomes, play an important part, since they assist in regulating the concentration of free oxygen radicals.
Nanocapsules Transform Radicals into Water and Oxygen
Prof. Cornelia Palivan and her research group at the University of Basel have successfully produced artificial peroxisomes that mimic the natural organelle. The researchers developed a cell organelle based on polymeric nanocapsules, in which two types of enzymes are encapsulated. These enzymes are able to transform free oxygen radicals into water and oxygen.
In order to verify the functionality inside the cell, channel proteins were added to the artificial peroxisome’s membrane, to serve as gates for substrates and products. The results show that the artificial peroxisomes are incorporated into the cell, where they then very efficiently support the natural peroxisomes in the detoxification process.
Novel Drugs
This type of effective principle targets the cell dysfunction directly on the cellular level, thus representing a further step towards the development of novel drugs that will make patient-oriented and personalized treatments possible in the future.
Source: http://www.unibas.ch/