There's a new way to explore biology's secrets. With a flash of light, scientists from the U.S. Department of Energy's Lawrence Berkeley National Laboratory and the University of California, Berkeley zeroed in on the type of neural cell that controls swimming in larval zebrafish.
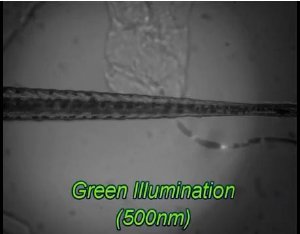
Using innovative light-activated proteins and gene expression techniques, the scientists zapped several zebrafish with a pulse of light, and initiated a swimming action in a subset of fish that was traced back to the neuron that drives the side-to-side motion of their tail fins.
The technique behind this needle-in-haystack search for the neural roots of a specific behavior could become a powerful way to learn how biological systems work. The light-activated protein could also become a handy tool in the field of synthetic biology, in which scientists seek to identify a useful network of proteins in one microbe and import it into another microbe — a method could be used to optimize the development of biofuels and disease-fighting therapies, to name a few applications. Their work is published in the Sept. 17 issue of the journal Nature.
“This is a very unique way of arriving at an individual cell: by starting with the behavior it controls,” says Ehud Isacoff, a biophysicist who holds joint appointments with Berkeley Lab's Physical Biosciences and Materials Sciences Divisions and UC Berkeley's Department of Molecular and Cell Biology. The research is an ongoing collaboration between Claire Wyart, a scientist in Isacoff's UC Berkeley lab, Filippo Del Bene of Herwig Baier's UC San Francisco lab, and Dirk Trauner of the University of Munich.
Currently, scientists often determine a neuron's function via correlation. If a group of neurons activates every time an animal performs a certain behavior, then chances are those neurons control that behavior. The same goes if the neurons are disrupted: if the behavior stops, then the affected neurons likely control it.
“But we want to move beyond correlation and actually test causality,” says Isacoff. “We want to play a behavior back to the nervous system and determine the individual neurons that are directly responsible. And this has been very difficult to do until now.”
The key to the scientists' success is an artificial, light-activated protein they developed a few years ago. The protein can be genetically engineered to express in a neuron or other type of cell and function as an optical switch. Zap the protein with one color of light, and it switches on and activates its host neuron. Zap it with another color of light, and it turns off and the neuron becomes dormant.
To test the light-activated protein, Isacoff and colleagues used it to search for the cell that drives the neural circuit that mediates swimming in larval zebrafish.
First, they randomly expressed the protein in the genome of hundreds of larval zebrafish. Some light-activated switches popped up in muscle cells, some in bone cells, and some in the central nervous system.
Next, they chose only those fish in which the light-activated protein was expressed in neural cells in the spinal cord, some of which are known to control locomotion.
They then zapped these fish with light. Like dutiful servants, a handful of fish spontaneously flicked their tails side to side in a swimming motion. Further analysis led the scientists to the neural source of this behavior: all of the swimmers had the optical switch expressed in a cell called the Kolmer-Agduhr neuron, whose existence has been known for more than 75 years, but whose function had remained a mystery.
“Our technique allows us to identify previously unknown parts of neural circuits that control a behavior,” says Isacoff. “And this approach can be broadly used. What we have done with locomotion can be done with any behavior and in many biological systems. “
The research was funded in part by Berkeley Lab's Directed Research and Development Program and the National Institutes of Health Nanomedicine Development Center for the Optical Control of Biological Function.
Neuron's function revealed