Dynamic light scattering (DLS) is a valued sizing technique for proteins, colloids and dispersions, which comfortably extends to the sub 1 nm region. The observation of scattered light helps determine defining characteristics of a particle dispersion or molecular solution such as particle size, molecular weight and zeta potential.
This article examples the benefits and limitations of DLS, focuses on those aspects of system design that are crucial in defining performance.
The Basics of DLS
Small particles in a dispersion or solution are subject to Brownian motion. DLS is driven by collisions with the solvent molecules present, which are in constant movement due to their thermal energy. The speed of Brownian motion can be directly measured from the scattered light pattern produced by the moving particles, a technique known as photon correlation spectroscopy (PCS) or quasi-elastic light scattering (QELS) but presently referred to as DLS.
The relationship between the speed of Brownian motion of a particle and that particle’s size is defined by the Stokes-Einstein equation:
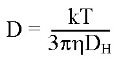
Where D = Diffusion speed, k = Boltzmann’s constant, T = absolute temperature, η = viscosity, and DH = hydrodynamic radius.
This relationship shows how size can be determined from diffusion speed provided that the temperature and continuous phase viscosity of the sample are known.
The Benefits of DLS
The benefits of DLS are:
- Operators can attain usable, detailed data without needing to have significant expertise.
- Sample volumes are small, down to just a few microliters, making this an appealing technique for early stage research where valuable materials are involved.
- DLS is essentially very good at measuring particle size across the range ~ 0.1nm to ~ 10µm.
- Its ability to measure small particles is especially valuable, with most systems giving accurate, reproducible data for particles from 2nm and above.
- It is possible to measure both dilute and turbid systems with the concentration range for analysis reaching down as low as 0.1ppm and up to 40% w/v.
- This size and concentration range, together with the high reproducibility of the technique makes DLS suitable for a wide range of applications.
Limitations of DLS
The limitations of DLS are:
- Large particles if present in even small quantities may be accounted during data analysis
- Sedimentation - This is particularly likely with more dense particles. Increasing the density of the dispersant by, for example, introducing sucrose is a helpful strategy but only for density matching up to around 1.05 g/ml.
- Low resolution - When size populations are closely spaced, less than a factor of three difference in size, DLS will not precisely characterize a polydisperse sample, making it advisable to apply separation prior to measurement.
- Multiple light scattering - Multiple scattering is when the scattered light from one particle is scattered by another before reaching the detector, and it compromises the accurate calculation of particle size in more concentrated samples.
- Dispersant choice – Even though most dispersants are suitable, those with a viscosity greater than 100 mPa.s inhibit reliable measurements. Furthermore, light absorption by the dispersant can interfere with detection
Defining Features of DLS Instrumentation
Key features of the optical set-up are:
- The laser - A stable laser with low noise characteristics, as exemplified by certain Helium-Neon gas lasers, is the most suitable.
- The optical arrangement – Measuring only at 90o enables the production of a simple, cost-effective system that provides an appropriate level of sensitivity for many applications as shown in Figure 1. For experiments that require higher sensitivity levels or for more concentrated samples, measuring at wider angles is preferable.
- The detector(s)- Detectors are one of two types: cheaper, less sensitive photomultipliers or more expensive, higher performance avalanche photodiode detectors (APD).
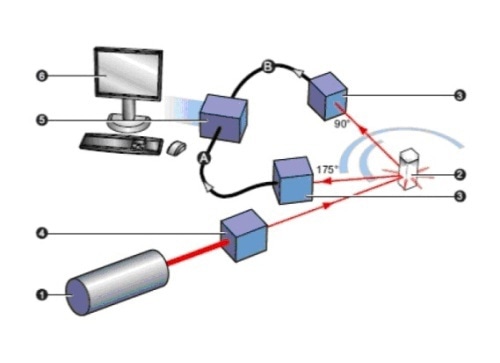
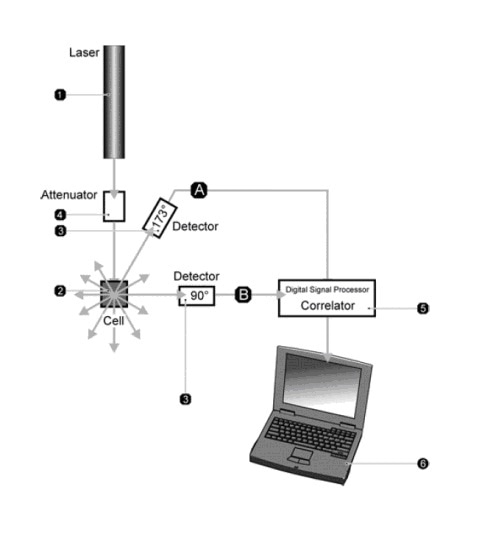
Figure 1. The key elements of a DLS system are the laser (1), measurement cell (2), detector (3), attenuator (4), correlator (5) and data handling PC (6). Detectors can be placed at either 90o or at a wider angle as exemplified by the NIBS detector shown at here at 173o.
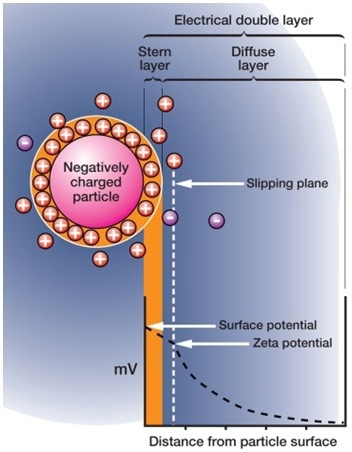
Figure 2. Zeta potential quantifies the repulsion between particles in a suspension aiding stability studies.
Another essential requirement for precise DLS measurements is good temperature control. Built-in temperature control is easier to use than attaching an external water bath set-up. In addition, instruments with high-performance control systems may offer fast system warm-up and the ability to rapidly change temperature in order to study temperature-dependent effects such as protein melting.
Assessing the Extras
DLS is accepted for particle size measurement, also the combination of components that make up a highly specified DLS system also lend themselves to the measurement of:
- Absolute molecular weight
- Zeta potential
- Protein charge
- Microrheology
Selection
While selecting the instrument, these factors must be considered:
- What is most important: speed or accuracy?
- What sample size one needs to measure?
- What type of samples are going to be measured – are they toxic for example, or especially corrosive?
- Are the operators likely to be expert or novice users? How much light scattering expertise is available?
Conclusion
DLS is an established method extensively used for molecular and particle size measurement, for a truly diverse range of sample types. This makes it important to closely match user requirements to the capabilities of a system while choosing an instrument.
Alongside particle size, light scattering systems may also determine molecular weight, protein charge and zeta potential, or even have microrheology capabilities. There is a variation in sensitivity considerably from system to system, defining the size of particles or molecules that can be measured robustly, as does the ability to measure at high concentrations.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information please visit Malvern Panalytical.