What is a Cryostat?
In a vacuum-insulated sample environment that uses liquid cryogens, a cryostat employs helium, nitrogen, or a mechanical cooler to cool a sample's temperature. Depending on the boiling point of the cryogen used, base temperatures of 77 K (nitrogen) or <4 K (helium) can be reached. By combining the cooling power of the cryogen or cooler with a voltage applied across an electrical heater element, accurate temperature control can be achieved.
When purchasing an optical cryostat system, which can be overwhelming, there are many different types of cryostats and several things to consider. Cryostats, for instance, can have different cooling regimes, different shapes of windows, and different window types and can be better suited for either samples or specific experiments. Samples can be held in different environments under vacuum or in an exchange gas.
Cryostats can also use different mechanisms for cooling, for instance, a liquid cryogen like helium or nitrogen or a cryogen-free system. These points will be expanded within this article, along with explanations of the options available.

Figure 1. Different types of cryostats, nitrogen, dry, and helium. Image Credit: Andor Technology Ltd.
Uses for Cryostats
Cryostats have a wide range of applications, from cooling liquid to powder, to solid samples for spectroscopy and microscopy experiments.
Table 1. Source: Andor Technology Ltd.
| Microscopy Applications |
Spectroscopy Applications |
- Ultraviolet-Visible Spectroscopy
- Micro-Raman
- Micro-FTIR
- Micro-photoluminescence
- Fluorescence Spectroscopy
|
- THz spectroscopy
- UV/Visible reflectivity & absorption Photo/Electroluminescence
- Raman Scattering
- Electrical transport measurements
- Ultrafast spectroscopy
|
Types of Cryostats
With no need to continuously refill from a storage Dewar, bath cryostats contain enough liquid cryogen to allow for an adequate experimentation time. A continuous flow of liquid cools continuous flow cryostats, fed from an external transport Dewar. To cool the sample, dry cryostats or cryogen-free cryostats use a mechanical cooler.
What is a Liquid Cryogen?
A liquid cryogen is a gas cooled below its boiling point and then stored in a vacuum-insulated transport vessel. For cooling experiments or storing samples, they can be used to cool to extremely low temperatures, <4.2 K for Helium and 77 K for Nitrogen. The helium cost calculator. helps clients to work out the cost of running their cryostat with helium.
The Difference Between a Nitrogen and Helium Cryostat
As helium and nitrogen have different physical properties, the design of cryostats will vary accordingly.
Although liquid nitrogen (LN2) can exist in an uninsulated vessel for a period of time, eventually, it will boil away, resulting in icing around the vessel as the water vapor freezes. Nitrogen cryostats have a vacuum space to reduce the boil-off and isolate the outer surfaces from the cold liquid. A charcoal sorption pump is fitted within the space to improve the vacuum as the boiling point of LN2 (77 K) is not cold enough to freeze the gases left in the vacuum space (cryopump).
Due to radiated and conducted heat loads, Liquid Helium (LHe) is more difficult to store without losing liquid. It can be more efficient to store the liquid in an external transport Dewar and use a "low loss" transfer line to pass the helium through for small cryostats.
LHe cryostats also have an intermediary radiation shield to intercept radiated heat loads from room temperature (300 K) in addition to the vacuum space between the sample and room temperature. This requires a window in the shield in addition to the outer casing when used for optical cryostats.
The Difference Between a "Wet" Cryostat and a "Dry" Cryostat
A liquid-cooled cryostat uses liquid cryogens, like nitrogen or helium, to lower a sample's temperature. Either the liquid is fed through the cryostat from an external transport vessel (flow cryostats), or these cryostats hold the liquid within the unit (bath cryostats).
Instead of requiring liquid cryogens, cryogen-free or dry cryostats use a mechanical cooler to lower the temperature of the sample space. Though these can use PTR (Pulse Tube Refrigerator) coolers, they are typically GM coolers (Gifford-McMahon).
What Are the Advantages of Dry Cryostats Over Helium Cryostats?
As noted, dry or cryogen-free cryostats do not require any liquid cryogens. Dry or cryogen-free cryostats can have a lower cost of operation because they do not need liquid helium, although they may cost more to purchase initially.
Alternatively, with a cryogen storage Dewar, transfer tube, and a system for helium recovery, a helium cryostat will require a set-up, outlined in figure 2 below.
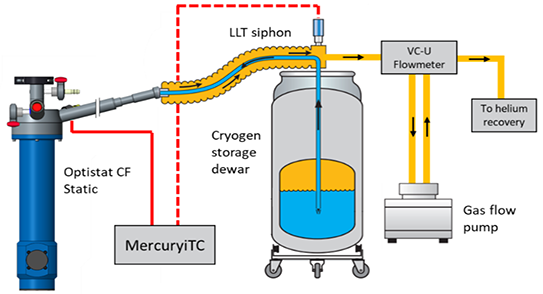
Figure 2. Helium cryostat experimental set-up. Image Credit: Andor Technology Ltd.
It is vital to consider the infrastructure required to obtain, store and manage liquid gases when considering the running costs of a wet vs. dry system. An electrical supply cost is associated with the compressor (3-6 kW) and a need to cool it using either a water chiller or fan, although these costs do not apply to dry systems.
Running Costs of a Cryostat
Cryostat running costs will depend on its type. Running a helium cryostat, for instance, will cost more than a dry or cryogen-free model over the experiment's lifetime. The helium cost calculator. allows users to determine the costs of running their cryostat with helium.
Controlling the Temperature of a Cryostat
A temperature controller with a proportional-integral-derivative (PID) controller can typically be purchased with cryostats. Andor's cryostats, for example, can be bought as standard with the Mercury iTC controller with one sensor/heater PID loop as standard. The temperature controller works with the heat exchanger in the cryostat, and heating and cooling are balanced via the voltage across the heater with cryogen or coolant's flow.
Another advantage of using a suitable, high-quality controller is that it consistently monitors the cryogen flow, the voltage, and the temperature. It makes the cryostat more efficient over time by slowly lowering the voltage and the flow to consume the minimum amount of cryogen.
Choosing Between a Cryostat With the Sample in an Exchange Gas or in Vacuum
In an exchange gas or within the isolation vacuum, cryostats can be designed to mount the user's samples. Different environments require different types of samples. For instance, powders or liquids can be contained in a cuvette and cuvette holder and subsequently loaded into a sample tube filled with an exchange gas (usually a small amount of helium gas).
It is highly impractical to load liquid samples into a vacuum space. The sample can be changed very quickly in the exchange gas by simply lifting it out, which only takes a couple of minutes. Uniformly cooling the sample aids in the presence of the gas.
The need for an additional window between the sample and the vacuum space is one of the downsides of having a sample tube with an exchange gas, as shown in figure 3 below. There are three windows for a helium cryostat with a gas exchange sample between the sample and the outside world.
Firstly, there is the sample tube with an inner window; secondly, the radiation shield with the middle window; and finally, the outer vacuum casing with the exterior window. It is important to note that only two windows are required as there is no radiation shield on nitrogen cryostats.
Changing the sample for sample-in-vacuum systems takes longer as the whole cryostat must first be warmed to room temperature. There are fewer windows in the beam path, but liquid samples cannot be used. Powdered samples can first be compressed into pellets before mounting to cool them in a vacuum cryostat.
Table 2. Source: Andor Technology Ltd.
| Sample Type |
Sample Exchange in Gas |
Sample in Vacuum |
| Powder |
Yes |
(As compressed pellets) |
| Liquid |
Yes |
No |
| Solid |
Yes |
Yes |
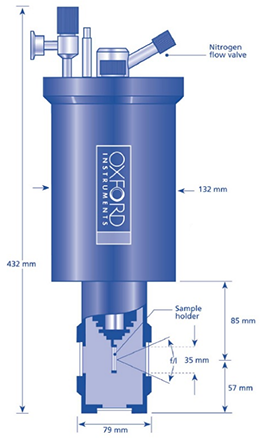
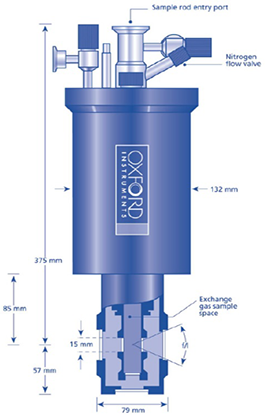
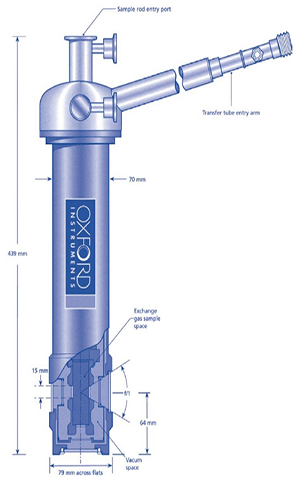
Figure 3. Top: sample in vacuum (1 window). Middle: Schematic of nitrogen cryostat with sample in exchange gas (2 windows). Bottom: Schematic of helium cryostat with sample in exchange gas (3 windows). Image Credit: Andor Technology Ltd.
Cryostat Window: Explained
Between the sample environment and either the vacuum space or the outside environment, the window on a cryostat forms a "transparent" barrier. It is essential to check that the window type is suitable for your experiment, as each window type has its own transmissive properties and is "transparent" to a unique portion of the light spectrum.
Why Are Multiple Window Options Needed for a Cryostat?
For different experimental needs, different window options are required. Some researchers, for instance, may need a window with high transmission across the spectrum or in the far infrared. The individual experimental requirements and the budget available will determine the window choice. Spectrosil B or WF, sapphire, crystalline quartz, zinc selenide, calcium fluoride, polythene, KRS-5, and Mylar are available for material options.
Many window options are available from Andor for their Optistat and Microstat ranges, which cover a wide range of materials and wavelengths. See their Optistat and Microstat specifications for more information. Outer, middle, and inner window fittings are shown in Figure 4.
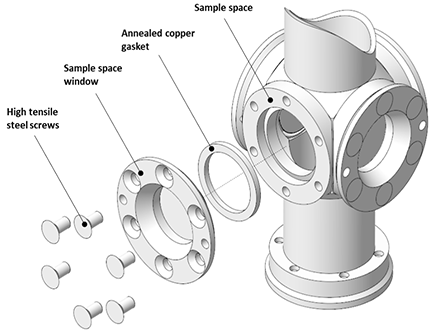
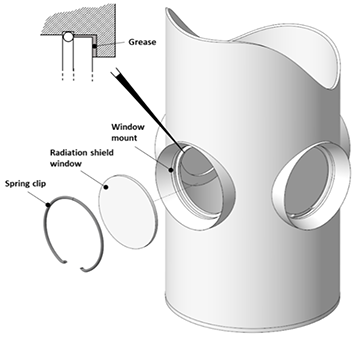
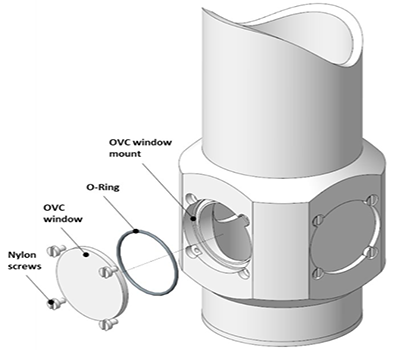
Figure 4. Top: Outer window mounts. Middle: Middle - radiation shield - window mounts. Bottom: Inner window mounts. Image Credit: Andor Technology Ltd.
Optistat Windows: Are They Interchangeable?
Andor's optical cryostats have interchangeable windows and can be changed by the customers themselves. However, it is essential to check with the supplier, since this will depend on the manufacturer of your cryostat.
Cryostats: How Much Do They Cost?
Depending on the type and methods used for cooling and the windows used, cryostat costs can vary. For simple nitrogen bath cryostats to £15 - 20 K for helium flow cryostats, prices can range from £5-6 K GBP. Depending on design and performance, cryogen-free cooler systems can cost upwards of £30 - 60 K
Dry Cryostats: Why Are They More Expensive to Buy Than Helium Cryostats?
Due to the mechanical build, materials, and control electronics, all cryostats have associated costs. The additional cost of the GM cooler and compressor is attached to the dry (cryogen-free ) systems, which is often the most significant cost related to a new cryostat.
Andor’s Optistat and Microstat Ranges: What Are the Differences?
Whilst the Microstat range is suitable for microscopy; the Optistat range is ideal for spectroscopy. An overview of the product range is depicted in the table below.
Andor's Optistat range for spectroscopy includes two cryogen-free, or Cryofree® systems and nitrogen cryostats (DN-V or DN-X) liquid helium-flow cryostats (CF-V or CF-X), all of which are designed for general spectroscopy.
Each forms a cube with five windows - four radial and one on the bottom - to a sample position to make the same optical tail design. This design allows for good all-around optical access to either excite the sample with light or measure the light that is emitted.
The Microstat range has two helium-flow cryostats (He and HiRes) and one nitrogen (N). Though they are different shapes and sizes, they work the same way as the Optistat range. They have been specifically designed to get your microscope objective as close to your sample as possible. A close-up of the sample to a couple of millimeters is facilitated by the fact that, with these systems, there is a single window in the top and one in the bottom for transmission. Within the Microstat range, all samples are held under vacuum.
Table 3. Source: Andor Technology Ltd.
| |
Spectroscopy (Optistat range) |
Microscopy (Microstat Range) |
| Sample in vacuum |
Sample in exchange gas |
Sample in vacuum with standard vibration |
Sample in vacuum with low vibration |
| Liquid Nitrogen |
 |
OptistatDN-X |
|
 |
| OptistatDN-V |
OptistatDN-X |
|
MicrostatN |
| Liquid Helium |
 |
 |
 |
 |
| OptistatCF-V |
OptistatCF-X |
Microstat He |
MicrostatHiRes |
| Cryogen Free |
 |
 |
|
|
| OptistatDry BLV |
OptistatDry TLEX |
|
|
Common Acronyms Used with Cryostats
Table 4. Source: Andor Technology Ltd.
| . |
. |
| PID controller |
Proportional-integral-derivative controller |
| OVC |
Outer vacuum casing |
| LN |
Liquid Nitrogen |
| LHe |
Liquid Helium |
| LLT |
Low Loss Transfer |

This information has been sourced, reviewed and adapted from materials provided by Andor Technology Ltd.
For more information on this source, please visit Andor Technology Ltd.