The production and research of nanocarbon for both diamond and graphite forms has witnessed a significant growth in recent times.
Nanocarbon graphite finds applications in fuel cells, textiles, polymers, and bio-nano applications. The nanodiamond was reported to have been employed in polishing, dry and oil lubricants, and drug delivery.
As the name implies, carbon nanotubes (CNT) are cylindrical carbon atoms with a few nanometers in diameter, and a length that is greater than the diameter by several million times. These structures are made of graphene sheets and have significant tensile, mechanical, thermal and electrical properties. Potential uses of CNTs include medical diagnostics, environmental contaminant removal, super capacitors, solar cells and electrical circuits.
This article reviews some examples that use Nanoparticle Tracking Analysis (NTA) to analyze carbon-based nanostructures.
Insight on Nanoparticle Tracking Analysis
Based on the principles of Brownian motion and light scattering, NTA determines the particle size distribution of samples in liquid suspension. On passing a laser beam through the sample chamber, the light beam is scattered by the particles in the suspension within the beam path in such a way that the particles can be observed using a 20x magnification microscope featuring a camera on top of it. The camera is operated at about 30 frames per second (fps), and it tracks the particles under Brownian motion within the field of view of about 100 x 80 x 10 µm, as shown in Figure 1. The particle motion is captured as a video file.
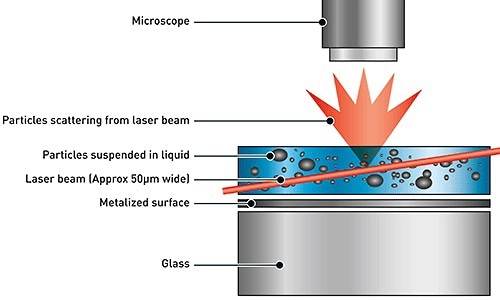
Figure 1. Schematic of the optical configuration used in NTA.
The center of each particle observed is simultaneously determined using the proprietary NTA software which also evaluates the average distance of each particle’s movement along the x and y planes. Using this value, the particle diffusion coefficient (Dt) can be determined. Further, the sphere-equivalent hydrodynamic diameter, d, of the particles can be obtained using Dt, known solvent viscosity η and sample temperature T, based on the Stokes- Einstein equation (Equation 1).
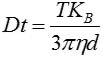
An example of the size distribution profile generated by NTA is shown in Figure 2. The movement of particles measured in a fixed field of view can be revealed using an illumination beam of 10 µm depth. The scattering volume of the target sample can be estimated using these figures.

Figure 2. An example of the size distribution profile generated by NTA. The modal size for this sample is found to be approximately 70nm, with larger sized particles also present.
Using NTA in Carbon and Carbon Nanotube Research
Preliminary studies have suggested the use of NTA to characterize different carbonaceous nanomaterials, including CNT-nematic liquid crystal composite materials (Trushkevych et al., 2007 and 2008) in addition to its use in the determination of the oxidative potential of a panel of carbonaceous and metallic nanoparticles (Hohl et al., 2009). However, more recently, NTA have been reported to analyze nanocolloids and carbon nanotubes as well.
Kim et al. (2011) produced water-based nano-size carbon colloid using an electro-chemical method to easily and inexpensively evaluate the removal efficiency of formaldehyde. They used NTA to investigate the carbon particle size in production. In another study by Lv et al. (2011), NTA was used to evaluate the graphene oxide nanoparticle size while producing graphene oxide membranes suitable for new optical devices.
NTA has been used to determine the sphere equivalent diameter of CNTs despite of their highly asymmetric shape. The initial state of colloidal stability of carbon nanotubes was investigated by Schwyzer et al. (2011) under natural conditions for a period of time. The cytotoxicity of a water-soluble, radioprotective C60 fullerene derivative (DF) generated upon the reaction of C60 fullerene with peracetic acid and subsequent hydrolysis was studied by Zemanova.
The NTA was used to show the lesser cytotoxicity of monodisperse DF to cell cultures when compared to an unfiltered, polydisperse equivalent coagulated on cell surfaces (Zemanova et al., 2011). Clements (2013) reported that the NTA particle size distribution for a C60 colloids sample used particles that are slightly larger than 100 nm.
Russ (2013) investigated the cellular toxicity of C60 fullerenes in RAW 264.7 immortalized macrophages. The results of the study showed the nanoparticle uptake and changes in the normal functions of the cell while exposing immune cells to C60 fullerenes. The size of C60 Fullerene and terbium endohedral Fullerene aggregates was analyzed using NTA.
Using single particle-inductively coupled plasma-mass spectrometry (spICPMS), Reed et al. (2013) detected single walled CNTs by observing embedded metals with trace catalytic metals intercalated in the CNT structure as substitutes for the nanotubes. It was evident from the split sample analysis, by both spICPMS and NTA, that the particle number concentration quantification using spICPMS has a lower magnitude than by NTA.
The researchers claimed that this was a result of metal content and/or size, due to the influence of CNTs that do not have sufficient metal over the instrument detection limit, leading to undercounting CNTs using spICPMS. They also concluded that, as it is impossible to detect CNTs at low ng L-1 concentrations using other techniques, spICPMS serves as a more sensitive technique to detect CNTs. Recently, Fahmy et al. (2013) have reported the activation of immune responses using carbon nanotube-based compositions and NTA data on analysis of their magnetite and CL-2 loaded PLGA nanoparticles.
The adsorption of size-selected Pt colloidal nanoparticles on high-surface-area graphene powders have been investigated by Sun et al. (2013) for activating methanol oxidation reaction. They observed that the adsorption of Pt colloidal nanoparticles on graphene surfaces depends on the process parameter in the mixing process, and that the different solution volumes lead to multiple catalyst morphologies.
Chen et al. (2013) have identified the interactions between protein and carbon black using NTA. The findings suggest the reaction of CB with 55 kDa and 70 kDa proteins following inhalation, and the chances of altering the functional structures of lung proteins, resulting in the activation of acute-inflammatory responses in the lungs.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information please visit Malvern Panalytical.