Titanium dioxide (TiO2) nanoparticles are one of the main ingredients of sunscreen formulation, which helps to block the sun's harmful UV rays. With the increasing use of nanoparticles in consumer products, there has become a bigger concern to assess the health and environmental effects of nanoparticles.
When applied on the skin, sunscreens can be easily washed off with water, so the TiO2 nanoparticles can make their way into environmental and biological systems. For this reason, it is critical to evaluate the size and size distribution of these nanoparticles and their impact on the environment and human health
Different methods - including microscopy, dynamic light scattering, flow fractionation (FFF) and other techniques - have been used as the standard to determine the nanoparticle size characteristics.
More recently, single particle inductively coupled plasma mass spectrometry (SP-ICP-MS) has become a popular way to measure and characterize nanoparticles.
The key benefits of SP-ICP-MS include it's speed and the quantity of information obtained from the analysis. This article discusses the characterization of TiO2 nanoparticles in sunscreens employing SP-ICP-MS with a simple sample preparation.
Experimental
Sample Preparation
Table 1 shows the label information of sunscreens purchased from a local store. The sample preparation involves adding 15 mL of sunscreen to a 50 mL container and mixing for three minutes to form a homogeneous sample. Following this, 0.2 g of the homogenized sample was transferred into another container and 200 mL 1% Triton-X solution was added.
The solutions were then sonicated until the disintegration of all the aggregates. Using deionized water, serial dilutions were carried out on the samples to generate particle concentrations of 100,000 - 200,000 particles/mL.
Table 1. Label information for sunscreen products
| Sunscreen |
SPF |
TiO2 Content (%) |
| 1 |
60+ |
4.9 |
| 2 |
50 |
6 |
| 3 |
45 |
0 |
| 4 |
50 |
6 |
| 5 |
45+ |
5.1 |
Gold nanoparticles (50, 80, 100 nm, nanoComposix, San Diego, California, U.S.A.) were used to determine transport efficiency. Using deionized water, these nanoparticles were diluted to a final nominal concentration of 100,000 particles/mL.
Before starting the analysis, the standards were sonicated for five minutes to ensure dispersion of any agglomerated particles. In order to check the accuracy of the TiO2 size measurements, 40 nm TiO2 (US Research Nanomaterials Houston, Texas, U.S.A.) spikes were added to different samples.
Titanium calibration standards include 2, 5, and 10 ppb standards formed in deionized water from a 1000 mg/L titanium standard (PerkinElmer, Shelton, Connecticut, U.S.A.).
Instrument Conditions
The Syngistix™ Nano Application Software Module was used to carry out all of the analysis on a PerkinElmer NexlON® 350D ICP-MS. The instrumental and method parameters were shown in Tables 2 and 3, respectively. The quadrupole settling time is automatically eliminated when using the Syngistix Nano Application Module to avoid any particles from being missed. With a short dwell time and the elimination of quadrupole settling time, multiple data points can be collected for each particle.
Table 2. NexION 350D ICP-MS instrument parameters
| Parameter |
Value |
| Nebulizer |
Glass concentric |
| Spray chamber |
Glass cyclonic |
| Sample uptake rate |
0.26-0.28 mL/min |
| RF power |
1600 W |
| Analysis mode |
Standard |
| Quadrupole settling time |
0 µs |
Table 3. Method parameters
| Parameter |
Value |
| Analyte |
Ti48 |
| Dwell time |
100 µs |
| Data acquisition time |
100 sec |
| Density |
4.23 g/cm3 |
| Ti mass fraction |
60% |
Results and Discussion
A blank solution containing only the reagents (i.e. Triton X-100 in deionized water) was analyzed to verify the Ti background of the system and reagents. The resulting signal is displayed in Figure 1. Not many signal spikes were observed. Most of the Ti background is less than two counts, indicating the cleanliness of the system.
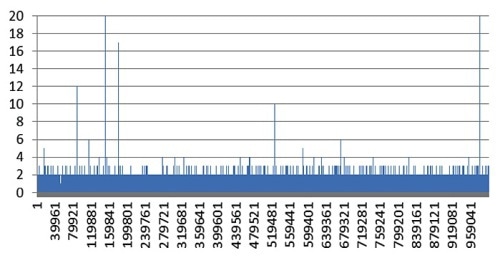
Figure 1. Ti background signal for blank
The ability to identify TiO2 particles in a sunscreen matrix was analyzed using sunscreen that is free of any TiO2 particles. The TiO2 particle signal from a sunscreen without TiO2 (according to its label) is shown in Figure 2A. This demonstrates that the background is almost similar to the blank, aside from a few spikes.
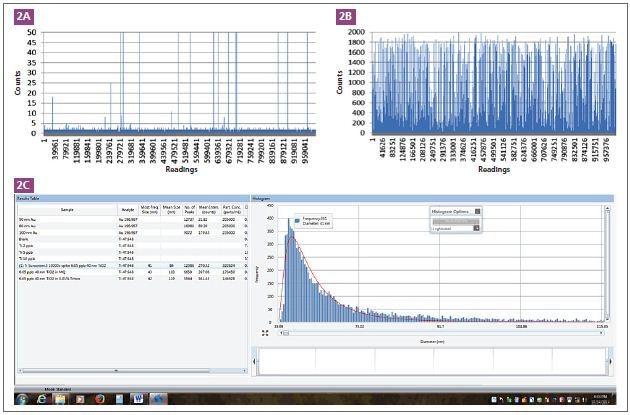
Figure 2. TiO2 signals from a sunscreen which does not contain TiO2 (2A), and spiked with 40 nm TiO2 (2B) particles. The screen shot shows the processed data of spiking 40n m TiO2 into a sunscreen which does not contain any TiO2 nanoparticles (2C).
The sunscreen solution was then spiked with 40 nm TiO2 particles at a 6.65 μg/L concentration. The resulting signal is displayed in Figure 2B which indicates that each spike represents a particle. This ensures the presence of TiO2 particles in a sunscreen matrix.
The most frequent size particle was observed to be 37.7 μm, which demonstrates the measurement accuracy. The TiO2 particle size distribution for a 20,000 times-diluted sunscreen (Sunscreen 5) is shown in Figure 3.
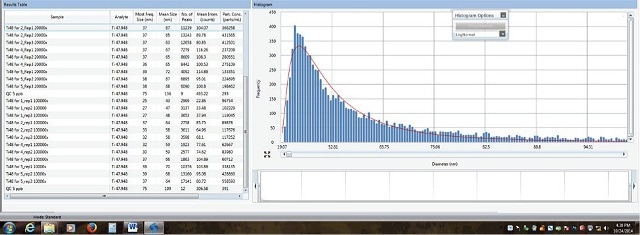
Figure 3. Processed data for 20,000 times-diluted sunscreen which contains TiO2 nanoparticles
Since the ability to measure TiO2 particles in sunscreen accurately was determined, other sunscreen samples were analyzed thrice and their results are listed in Table 4.
Table 4. TiO2 nanoparticle analysis in commercial sunscreens
| Sunscreen |
Dilution Factor |
Most Frequent Size (nm) |
Particle Size Distribution (nm) |
Particle Concentration (particles/mL) |
| 1 |
100,000 |
32 |
24 – 58 |
102,229 |
| 2 |
100,000 |
34 |
24 – 64 |
117,252 |
| 3* |
100,000 |
- |
- |
- |
| 4 |
100,000 |
33 |
24 – 61 |
63,000 |
| 5 |
20,000 |
42 |
28 – 67 |
198,462 |
*This sunscreen contains no nanoparticles, as shown in Table 1.
The presence of TiO2 nanoparticles ranging from 24 to 67 nm with even size distributions is evident from these results. However, Sunscreen 5 is clearly different from the other samples. It requires smaller dilution factor to achieve less than 200,000 particles/mL. This clearly shows a lower number of TiO2 particles in this sample when compared to the others.
Conclusion
This article provides an experimental demonstration of TiO2 nanoparticles measurement in commercial sunscreen samples. The combination of both the NexION 350 ICP-MS and the Syngistix Nano Application Software Module has made the analysis rapid and simple, and enabled clear differentiation of TiO2 content in different samples.

This information has been sourced, reviewed and adapted from materials provided by PerkinElmer Inc.
For more information on this source, please visit PerkinElmer Inc.