Nov 8 2006
.jpg)
Sourced from: Shutterstock
Just as the key building block to life on earth is the carbon atom, carbon is the key to one of the most promising branches of nanotechnology. Much of the current research and commercialisation of nanotechnology relies on tubes, wires and balls made from carbon atoms. All are known by a number of names. The carbon based tubes can be carbon nanotubes, buckytubes and very long tubes are often referred to as nanowires. The balls are known as fullerines or buckyballs.
Origins of the Buckyball
Russian researchers discovered carbon nanotubes in the 1950s but the research was only published in Russian and the Cold War meant this information was not widely available. In the 1980s knowledge surrounding carbon nanotubes and balls become more widespread. The balls resembled the geodesic dome shape designed by American, R. Buckminster Fuller, and the buckyball or fullerene was named in his honour. The simplest form of buckyball is the C60 Buckminsterfullerene. A shape instantly recognisable for it’s soccer ball like features.
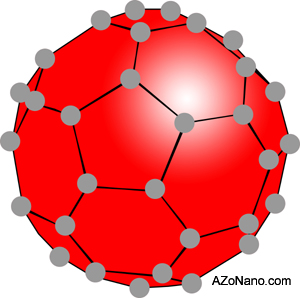
Figure 1. C60 variant of a buckyball
Buckyballs
In the simplest buckyball there are 60 carbon atoms with each atom covalently bonded to three adjacent atoms. The resultant sphere is around 1nm in diameter. Other fullerenes exist with their name given by the symbol for carbon, C, followed by the number of carbon atoms present, e.g. C60, C70 and C80.
Making Buckyballs
Although buckyballs spontaneously occur in nature from origins as simple as candle soot, their discovery is a very recent occurrence. Japanese researcher Eiji Osawa of Toyohashi University of Technology proposed the existence of buckyballs in 1970.
However, it took until August 1985 for Richard Smalley, Harry Kroto and Bob Curl to perform an experiment at Rice University in the USA that produced buckyballs. Their experiment vaporised carbon with a laser and produced a very small quantity of buckyballs. The quantity was enough to prove their existence, but not enough to study them in detail. For this work they were awarded the 1996 Nobel Prize in Chemistry.
In order to produce larger quantities for study purposes researchers from the Max Planck Institute and the University of Arizona combined forces to develop a method that used two carbon electrodes in a helium or neon atmosphere. When an arc was generated between the electrodes buckyballs were generated along with carbon soot that required removal by a solvent.
This was still not enough quantity for commercial purposes. Several methods now exist for making buckyballs in tonnage amounts but they tend to be a closely guarded commercial secret. The first bulk production method is known as combustion synthesis. Combustion synthesis burns a hydrocarbon and oxygen mixture at low pressure to produce 95% pure buckyballs.
Buckyball Applications
A range of promising applications exist for buckyballs. With buckyballs having hardness akin to or greater than that of diamond, researchers have seen promise for buckyball use within armour. This hardness also allows buckyballs to be added to various polymers to make them stronger.
Modified buckyballs are also being developed as antioxidants for use in humans. When a buckyball encounters a free radical, the unpaired electron in the free radical joins with a delocalised electron in the buckyball. The buckyball is modified in order to make it water soluble and suitable for medical use.
Modifying a buckyball by adding or replacing an atom in order to change the properties of the buckyball is called functionalization.
Functionalized buckyballs are being developed for targeted drug delivery. The buckyball encases a minute dose of a particular drug. By controlling the functionalization of the buckyball the drug remains encased until the buckytube reaches the site where the drug is required. The buckyball then releases it.
|