Molybdenum disulfide (MoS2) is a two-dimensional (2D) nanostructured material that has been used for many years as an industrial lubricant in its bulk form. The 2D form of the material was not discovered until 2011, when scientists succeeded in producing a transistor made from this new material.
For years, researchers have been struggling with the technical challenges of building electronic circuits from graphene, but with molybdenum disulfide researchers have already been able to develop a wide range of electronic components. Molybdenum disulfide in its 2D form could help development of futuristic products such as flat panel lighting covering entire walls, clothing embedded with electronics, and contact lenses with built in head-up displays.
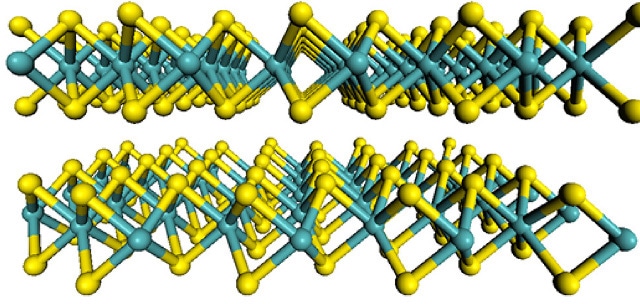
The flat structure of molybdenum disulfide. Molybdenum atoms are shown in teal, and sulfur atoms in yellow. Image credit: Wang et al, MIT News.
Properties of Molybdenum Disulfide
The properties of molybdenum disulfide are listed below:
- It is thin and transparent.
- It is capable of being deposited on other materials.
- It is capable of retaining huge amounts of elastic strain.
- It is naturally semiconducting - unlike graphene, the bandgap does not need to be physically induced.
- The bandgap varies with the strain on the material, allowing configurable electronic properties
Potential Applications of Molybdenum Disulfide
Molybdenum disulfide finds use in the following applications:
- It has a bandgap that allows easy usage of molybdenum disulfide to build transistors.
- A wide range of basic electronic components can be fabricated on this material.
- It could be used for high-efficiency solar cells, as varying the strain across an MoS2 sheet would vary the bandgap, allowing different parts of the cell to absorb different frequencies of light.
- It can be used as a transparent electrode in large-screen displays such as computer monitors and television sets.
- It can be used along with other 2D or thin-film materials to form flat light-emitting devices.
- It can be used to produce displays on windows and eyeglasses.
Advantages Over Graphene
Molybdenum disulfide has some of graphene’s properties but has an edge over graphene as this new 2D nanomaterial has a band gap in its electronic structure which is absent in graphene. Band gaps are the special electronic property of semiconductors which allows them to be made into devices that can be turned on or off - transistors, the building block of all modern electronic devices. Graphene lacks this band gap, which makes it an excellent electrical conductor, but hampers its direct use to fabricate electronic components.
Graphene does have other incredible properties, and can be used in optics, electronics and structural materials, but molybdenum disulfide may cover a range of applications in electronics that graphene cannot. This provides nanotechnology researchers with another tool to work with, which could speed up the development of nanotechnology-enhanced products that could bring about beneficial changes in all walks of life.
Conclusions
Molybdenum disulfide looks to be a promising class of material for next-generation electronics, as it is the only 2D material yet discovered which has an inherent band gap. With this feature, molybdenum disulfide paves way for the development of a totally new domain of electronic devices and materials.
With the significance of nanotechnology spreading far and wide, the discovery of nanostructured materials like molybdenum disulfide is accelerating scientific research, which is now focusing on studying the benefits and exploring the application areas of this nanomaterial.
The future will likely see a range of wearable devices, flexible electronics and even smart bandages featuring transistors made of molybdenum disulfide, which perform well even when bent.
Sources