Using a one-of-a-kind instrument designed and built at the National Institute of Standards and Technology (NIST), an international team of researchers have unveiled a quartet of graphene’s electron states and discovered that electrons in graphene can split up into an unexpected and tantalizing set of energy levels when exposed to extremely low temperatures and extremely high magnetic fields.
Published in the Sept. 9th, 2010, issue of Nature,* this new research raises several intriguing questions about the fundamental physics of this exciting material and reveals new effects that may make graphene even more powerful than previously expected for practical applications.
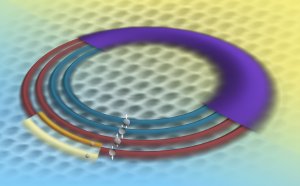 This artist's rendition illustrates the electron energy levels in graphene as revealed by a unique NIST instrument. Because of graphene's properties, an electron in any given energy level (the wide, purple band) comprises four quantum states (the four rings), called a "quartet." This quartet of levels split into different energies when immersed in a magnetic field. The two smaller bands on the outermost ring represent the further splitting of a graphene electronic state. Credit: T. Schindler and K. Talbott/NIST
This artist's rendition illustrates the electron energy levels in graphene as revealed by a unique NIST instrument. Because of graphene's properties, an electron in any given energy level (the wide, purple band) comprises four quantum states (the four rings), called a "quartet." This quartet of levels split into different energies when immersed in a magnetic field. The two smaller bands on the outermost ring represent the further splitting of a graphene electronic state. Credit: T. Schindler and K. Talbott/NIST
Graphene is one of the simplest materials—a single-atom-thick sheet of carbon atoms arranged in a honeycomb-like lattice—yet it has many remarkable and surprisingly complex properties. Measuring and understanding how electrons carry current through the sheet is important to realizing its technological promise in wide-ranging applications, including high-speed electronics and sensors. For example, the electrons in graphene act as if they have no mass and are almost 100 times more mobile than in silicon. Moreover, the speed with which electrons move through graphene is not related to their energy, unlike materials such as silicon where more voltage must be applied to increase their speed, which creates heat that is detrimental to most applications.
NIST recently constructed the world’s most powerful and stable scanning-probe microscope, with an unprecedented combination of low temperature (as low as 10 millikelvin, or 10 thousandths of a degree above absolute zero), ultra-high vacuum and high magnetic field. In the first measurements made with this instrument, the team has used its power to resolve the finest differences in the electron energies in graphene, atom-by-atom.
Because of the geometry and electromagnetic properties of graphene’s structure, an electron in any given energy level populates four possible sublevels, called a “quartet.” Theorists have predicted that this quartet of levels would split into different energies when immersed in a magnetic field, but until recently there had not been an instrument sensitive enough to resolve these differences. The experiment, according to the research team, revealed unexpected complex quantum behavior of the electrons in a high magnetic field at extremely low temperatures. The electrons apparently interact strongly with one another in ways that affect their energy levels.
One possible explanation for this behavior, the team says, is that the electrons have formed a “condensate” in which they cease moving independently of one another and act as a single coordinated unit. If so, the work could point the way to the creation of smaller, very-low-heat-producing, highly energy efficient electronic devices based upon graphene.
The research team includes collaborators from NIST, the University of Maryland, Seoul National University, the Georgia Institute of Technology and the University of Texas at Austin. For more details, see NIST's Sept. 8th, 2010, news announcement, “NIST Researchers Hear Puzzling New Physics from Graphene Quartet’s Quantum Harmonies” online at www.nist.gov/cnst/graphene_quartet.cfm.
* Y.J. Song, A.F. Otte, Y. Kuk, Y.Hu, D.B. Torrance, P.N. First, W.A. de Heer, H. Min, S. Adam, M.D. Stiles, A.H. MacDonald and J.A. Stroscio. High resolution tunneling spectroscopy of a graphene quartet. Nature. Sept. 9, 2010.