A research team from the North Carolina State University has discovered that the bulkiness of ligands, which are organic molecules utilized in the synthesis of gold nanoparticles, determines the size of the nanoparticles, which means the larger the ligands, the smaller the nanoparticles.
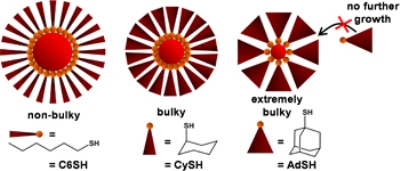 The bulkier the ligand, the fewer ligands can sit side-by-side -- leading to a smaller nanoparticle.
The bulkier the ligand, the fewer ligands can sit side-by-side -- leading to a smaller nanoparticle.
The researchers have also discovered that each kind of ligand synthesizes nanoparticles with a specific array of discrete sizes. Dr. Joseph Tracy, one of the researchers, informed that this research helps understand the formation of nanoparticles and provides a new tool to manipulate the size and properties of gold nanoparticles.
The study findings have been described in a paper titled ‘Bulky Adamantanethiolate and Cyclohexanethiolate Ligands Favor Smaller Gold Nanoparticles with Altered Discrete Sizes’ in the journal, ACS Nano.
In the process of gold nanoparticle synthesis, gold atoms are arranged effectively by the ligands to form the nanoparticles. During the process, the ligands assemble alongside to encircle the nanoparticles in all three dimensions.
To study the impact of the bulkiness of the ligands on the size of nanoparticles, the researchers tested three kinds of thiol ligands namely 1-adamantanethiolate (-SAd), cyclohexanethiolate (-SCy) and linear hexanethiolate (-SC6), of which -SAd is the bulkiest of the three and last in the order is -SC6.
The study has confirmed that the size of the nanoparticles is decided by the bulkiness of the ligands. Only fewer -SAd and -SCy ligands can sit side by side in three dimensions, thus bringing only fewer gold atoms together in the core, which in turn results in smaller nanoparticles. On the other hand, largest nanoparticles are created by -SC6, which is the least bulky among the three thiolates.
The researchers have also found that the formed smaller nanoparticles have very specific sizes, termed as discrete sizes. For example, some smaller nanoparticles may have 25 or 28 atoms but never have 26 or 27 atoms.
The research team has also discovered that the ligands’ bulkiness alters the nanoparticles’ discrete size. Tracy informed that this finding is important because the count of ligands and gold atoms varies with each discrete size, which may have an impact on the chemical behavior of the nanoparticle. However, this issue is yet to be explored.