A high entropy alloy Al8Co17Cr17Cu8Fe17Ni33, has been developed from an equiatomic AlCoCrCuFeNi alloy. It has promising properties such as oxidation resistance, high corrosion and high thermal stability and is a candidate for a number of applications at high temperatures such as furnace parts, tools and moulds.
An important challenge in today’s material science is the exploration of new metallic systems for high temperature applications. However, an increase of the alloy strength is desirable and needs further optimization.
To improve these mechanical properties, knowledge of the microstructure is necessary. Therefore, Al8Co17Cr17Cu8Fe17Ni33 high entropy alloy has been analyzed using energy dispersive X-ray spectroscopy (EDS).
High Entropy Alloys
High entropy alloys have been defined as consisting of five or more elements, each one having a concentration between 5 and 35 at%. These alloys are crystalline materials that mostly form simple solid solutions, mainly of body-centred or face-centred cubic structures.
High entropy alloys promise an interesting combination of properties, such as oxidation resistance, thermal stability, high strength and soft magnetic behaviour. The high entropy alloy most studied is AlCoCrCuFeNi with equiatomic composition.
Dendritic solidification takes place within a large variation of casting conditions. The dendrites comprise two phases, a Fe/Cr rich phase and an Al/Ni rich phase, whereas interdendritic regions are enriched in Cu.
It has also been observed that the Fe/Cr-rich phase decomposes into Fe-rich and Cr-rich domains. A length scale between these domains of just a few nanometres indicates spinodal decomposition as the formation process, which is often reported for Fe-Cr systems.
The ductile Al8Co17Cr17Cu8Fe17Ni33 alloy shows mainly two fcc phases in transmission electron microscopy, an ordered phase of L12 structure and a disordered fcc matrix. The ordered phase is nickel and aluminum rich as measured by atom probe tomography. At the boundaries, very few copper-rich precipitates are found.
In the present study, an Al8Co17Cr17Cu8Fe17Ni33 high entropy alloy that was subjected to cold rolling to reveal the microstructural properties as well as elemental distributions within the specimen. The Al8Co17Cr17Cu8Fe17Ni33 alloy was prepared in a vacuum induction furnace. The alloy constituents were of 99.99% purity. The ingots were remelted three times to achieve a better homogenization. Analysis specimens were ground mechanically and polished with an OP-U colloidal silica suspension.
The investigated samples were subjected to cold rolling, with up to 70% decrease in thickness. The elemental distribution maps created by EDS and EBSD, given in the present work, were obtained using Oxford Instruments Ultim® Max and Symmetry detectors.
The acceleration voltages and beam currents applied were 15kV and 10nA. EDS and EBSD acquisitions, and analyses, were performed using the AZtec software package. Evaluation of stored EBSD patterns for strain/stress analysis within individual grains were conducted using CrossCourt 3 (BLG Productions).
Results
Figure 1 shows the scanning electron micrograph that reveals a high density of extended structural defects within individual grains. The EBSD maps (using fcc Ni as reference) in Figure 2 show that along linear features, the local orientation is changed when compared with the matrix of the Al8Co17Cr17Cu8Fe17Ni33 specimen.
The distributions of the ε11, ε22, and ε33 strain tensor components, calculated from the EBSD data, indeed indicates that strain modulation is done across these linear features, with peak values of about 6 % strain.
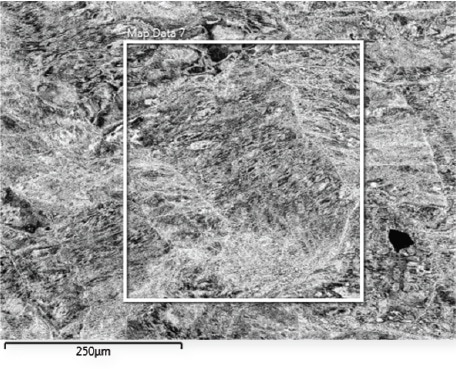
Figure 1. Scanning electron micrograph of Al8Co17Cr17Cu8Fe17Ni33 alloy. On the region highlighted by a white frame, EBSD and EDS measurements were performed

Figure 2. EBSD band contrast as well as orientation-distribution maps of the region highlighted by a white frame in Figure 1. The local orientations are given as colors (see legend) for the x, y, and z directions.
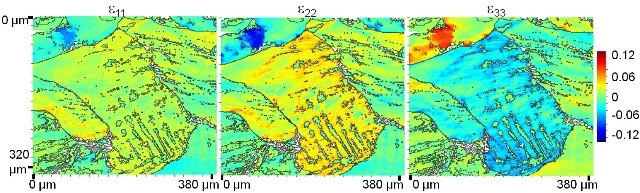
Figure 3. Distributions of the ε11, ε22, and ε33 strain tensor components, as calculated from the EBSD data (same identical position as in Figs. 1 and 2) by means of the CrossCourt software.
It can be seen from the elemental distribution maps obtained as in Figure 4 that the Al8Co17Cr17Cu8Fe17Ni33 alloy decomposes after casting into dendrites mainly consisting of Al/Ni/Cu-rich and Cr/Fe/Co-rich regions, which complies to previous reports.
Furthermore, elemental distribution maps acquired in cross-section show the layered structure of the Al/Ni/Cu-rich dendrites. Together, the distribution of the Al/Ni/ Cu-rich dendrites appears similar to the distributions of the ε11, ε22, and ε33 strain tensor components.

Figure 4. EDS elemental distribution maps calculated, using the Al-K, Ni-L, Cr-L, Fe-L, Cu-L, and Co-L signals, acquired on the same identical position as in Figs. 1-3.

Figure 5. EDS elemental distribution maps calculated using the Al-K, Ni-L, Cr-L, Fe-L, Cu-L, and Co-L signals, acquired on a cross-section specimen (view perpendicular to the maps in Fig. 4).
Conclusion
The present study shows how correlative imaging, as well as EBSD and EDS analysis in scanning electron microscopy on identical specimen positions, may reveal the microstructural properties and the elemental distributions in an Al8Co17Cr17Cu8Fe17Ni33 alloy. The distributions of Al/Ni/Cu-rich and Cr/Fe/Co-rich were detected and correlated to the distributions of the ε11, ε22, and ε33 strain tensor components.

This information has been sourced, reviewed and adapted from materials provided by Oxford Instruments Nanoanalysis.
For more information on this source, please visit Oxford Instruments Nanoanalysis.