Sponsored by HORIBAReviewed by Olivia FrostMar 26 2026
The antioxidant properties of curcumin, a compound found in turmeric (Curcuma longa), have attracted considerable scientific interest. Understanding how curcumin interacts with proteins is key to exploring its potential health benefits, and several studies have already examined these interactions.
Fluorescence spectroscopy is a particularly effective technique for uncovering molecular interactions due to its inherent sensitivity. Time-resolved fluorescence adds further value, as the fluorescence lifetime remains largely unaffected by factors like concentration or illumination intensity, providing an absolute measure.
Curcuminoids themselves show weak fluorescence in water, but their quantum yield increases when they bind to proteins such as human serum albumin (Figure 1). This makes fluorescence techniques well-suited for analysing these interactions.
Figure 1. Human serum albumin interacting with a curcuminoid. Image Credit: HORIBA
Experiment
A microsense cuvette was used to enable fluorescence measurements from volumes less than 5 µL (Figure 1).
This cuvette, which has external dimensions similar to those of a standard 10 mm path-length fluorescence cell, was placed within the sample chamber of a HORIBA DeltaFlex fluorescence lifetime spectrometer (Figure 2).
The DeltaFlex was run in kinetic Time-Correlated Single Photon Counting (TCSPC) mode, enabling the capture of up to 10,000 sequential fluorescence decay measurements, with acquisition times spanning from one millisecond to one minute.
The collection time depends on several factors, including the quantum yield of the sample (i.e., the number of photons that can be collected), the timescale of the kinetic process to be observed, and the fluorophore lifetime.
Data was collected every 10 ms and 10,000 decays were collected, alongside the instrumental response, for subsequent analysis via reconvolution.
This analysis was conducted in batch mode using DAS6 analysis software.

Figure 2. DeltaFlex fluorescence lifetime system. Inset: Microsense cuvette. Image Credit: HORIBA
DeltaDiode-425L was used as an excitation source, operating at a repetition rate of 100 MHz. The DeltaHub timing electronics within the system manage a low (<10 ns) dead time, which enhances photon collection efficiency at this high excitation rate.
Detection occurred at 530 nm using a PPD-650 detection module. The experiment involved placing 2 µL of curcuminoid solution (DMSO/phosphate-buffered saline, pH 7.4) into the Microsense cuvette, followed by the addition of 3 µL of human serum albumin in phosphate-buffered saline during the kinetic TCSPC run.
The instrumental response was measured using the same cuvette, but with 5 µL of Ludox® employed as a scattering solution.
Results
The 10,000 sequential fluorescence decays, each recorded over 10 ms, were analysed using a bi-exponential decay model to calculate the average lifetime.
Figure 3 shows the average lifetime plotted against time. It is worth noting that the time axis switches to a logarithmic scale after the break, and for clarity, only a selection of data points is shown.
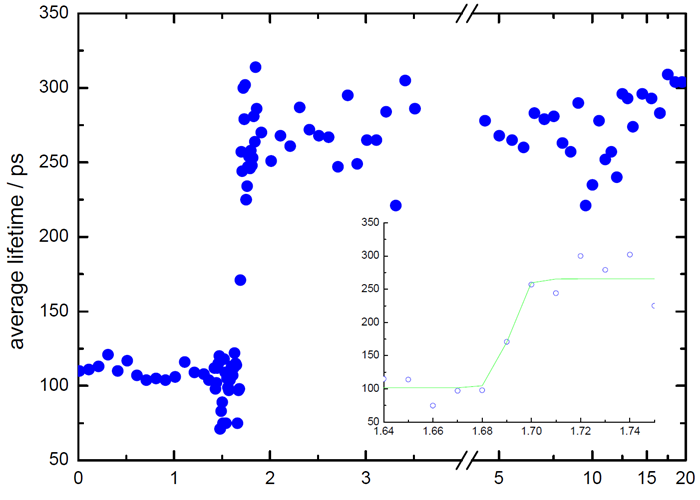
Figure 3. Lifetime vs time. Note logarithmic scale after break in time-axis. Image Credit: HORIBA
Figure 4 shows two representative fluorescence decays, illustrating shorter and longer average lifetimes. A clear increase in average decay time was observed, indicating an interaction between the curcuminoid and the protein.
Using fluorescence lifetime offers a key advantage, as it is unaffected by the dilution of the probe volume when the larger protein solution is added. By contrast, steady-state measurements would show a drop in intensity due to the reduced effective concentration of the probe.
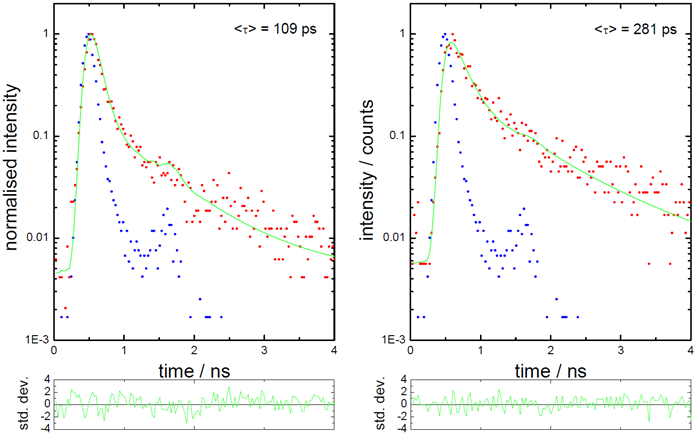
Figure 4. Fluorescence decays with shorter and longer lifetimes. Image Credit: HORIBA
Conclusions
The DeltaFlex system, equipped with the Microsense cuvette, can perform time-resolved fluorescence measurements to gather kinetic information on processes occurring on the millisecond timescale in microliter volumes.
In this instance, the interaction between a dye and a protein was monitored, with rapid and efficient data acquisition facilitated by a 100 MHz excitation rate combined with the low dead time of the timing electronics.
The application of time-resolved fluorescence offers a more comprehensive understanding by introducing an additional fluorescence parameter, the fluorescence lifetime, which is notably independent of concentration.

This information has been sourced, reviewed and adapted from materials provided by HORIBA.
For more information on this source, please visit HORIBA