High performance Dynamic Light Scattering (DLS) has been used to determine the hydrodynamic diameters of CdSe nanocrystals as well as CdSe cluster molecules in a size range of 1 to 10 nm. The method enables the determination of their particle size, including their ligand shells, in solution.
Dynamic Light Scattering Results Compared to other Methods
The results are consistent with the blue shift of the absorption bands, as well as Transmission Electron Microscope (TEM) experiments. The sizes of the cluster molecules were estimated from space filling models constructed from the results of a single crystal X-ray structure determination.
DLS gave comparable results for the size of both types of compound, indicating that it is potentially an important additional measurement technique to TEM, which uses harsh measurement conditions, and powder X-ray diffraction, which is difficult to interpret below 5 nm.
Introduction
The synthesis of nanoparticles of a wide variety of materials has received a remarkable amount of interest in the last few years, due to the possibility of changing a material's properties by simply changing its size.
Cadmium Selenide Nanoparticles
One frequently studied system is that of semiconductor nanoparticles, in particular CdSe. Applications for this material range from fluorescence markers in biological systems to the development of optical computing components. CdSe cluster molecules show the same quantum confinement properties as larger crystals, which allows their use as molecular models.
Measurement of the Size of Cluster Molecules
As the physical properties of the nanocrystals depend so strongly on their size, a precise and fast method of measurement is required. In the case of the cluster molecules which form three-dimensional crystal lattices, the size can be estimated from space filling models constructed from single-crystal X-ray crystallography data. Measurements of the nanocrystals however are usually done from high-resolution TEM and powder X-ray diffraction.
Limitations of Measurement Techniques
The TEM measurements are limited by the high temperatures generated in the sample, and powder X-ray diffraction (XRD) by the difficulty of result interpretation of samples smaller than 5nm.
Recent progress in the development of a Dynamic Light Scattering technology called NIBS, offers the possibility of measuring the size of the materials, including their ligand shells, under the same conditions as are used for optical spectroscopy.
Experiment
The CdSe nanoparticles and the four cluster molecules were prepared as described elsewhere.
These were a neutral [Cd ] cluster (1), a neutral [Cd ] cluster (2), an ionic mixed cluster {[Cd ] [Cd ] } (3) and a neutral cluster [Cd ] (4). For the DLS measurements, the particles were dissolved in appropriate solvents. The solutions were then filtered through syringe membrane filters with pores less than 0.4 µm, then centrifuged at 3400 rpm for 20 min. The measurements were carried out using a HPPS (High Performance Particle Sizer) using the new NIBS technology. This system combines a highly sensitive detector with backscatter optics to give the performance required for this measurement.
Measurements were made at 25°C in sealed quartz cuvettes.
Sample concentration was 1x10-3 mol l-1. This comparatively high concentration was used to reduce the effect of traces of dust, as any dust particles would then be a small proportion of the sample.
Results
The values of the peak means for the hydrodynamic diameters, show the expected trend from the smallest cluster 1, to the largest cluster 4.
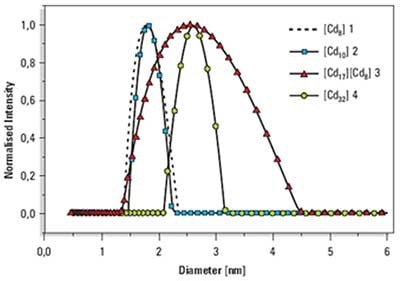
Figure 1. Overplot of size distributions of samples 1, 2, 3 and 4.
The larger peak width for sample 3 is in good agreement with structural features including the ionic forces between the clusters. However it should be noted that details of the size distribution would not be determined by this technique.
The comparison of the hydrodynamic diameters from DLS with the single crystal structure show reasonable agreement. (Table 1).
Table 1: Hydrodynamic diameters of cluster molecules 1, 2, 3 and 4 from dynamic light scattering and x-ray diffraction.
|
Sample
|
DLS dia (nm)
|
Single crystal XRD dia (nm)
|
|
1[Cd8]
|
1.80
|
2.20
|
|
2[Cd10]
|
1.82
|
2.18
|
|
3[Cd17]
|
2.50
|
2.52
|
|
4[Cd32]
|
2.60
|
3.14
|
To cover a wider size range, measurements were made on CdSe Nanoparticles synthesised in trioctylphosphine oxide (TOPO).
The sizes measured for these samples were significantly larger than determined by TEM. On average this difference was an increase in diameter of 2.6nm, and can be interpreted as a 1.3nm monolayer of TOPO. (Table 2).
Table 2: Hydrodynamic diameters determined by Dynamic Light Scattering (DLS) and core diameters by Transmission Electron Microscopy (TEM) of CdSe nanocrystals coated with Trioctylphosphine oxide (TOPO).
|
Sample
|
DLS dia (nm)
|
TEM dia (nm)
|
Thickness of ligand shell (nm)
|
|
NP1
|
4.8
|
2.4
|
1.2
|
|
NP2
|
5.6
|
3.4
|
1.1
|
|
NP3
|
6.2
|
3.8
|
1.3
|
|
NP
|
8.4
|
5.0
|
1.7
|
Confirmation of Analysis Methods
To test the reliability of the method, a measurement was made of a mixture of one of the CdSe cluster molecules (Sample 2) and CdSe nanoparticles measured on their own as 6.2 nm. The results clearly show separation of the two peaks (Figure 2). The means are slightly shifted to smaller sizes, and this shows the limit of the applicability of the DLS technique for mixtures.
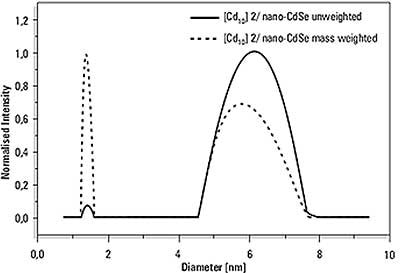
Figure 2. Size distribution result of the analysis of a mixture of CdSe cluster molecules (Sample 2) with 6.2nm CdSe (NP3) nanoparticles.
Conclusions
The results of the measurements show that Dynamic Light Scattering using NIBS technology is applicable to the measurement of very small particles and molecules such as CdSe cluster molecules and nanoparticles. It was possible to distinguish between the narrow monosize peaks of neutral cluster molecules and the broader distributions of ionic species.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information please visit Malvern Panalytical.