The bulk properties of polymer solutions are sensitive to polymer concentration. Polymer solutions are typically described as being either dilute, semi-dilute, or concentrated. The concentration defining the border between the dilute and semi-dilute regions is defined as C*, and is representative of the concentration at which polymer chain entanglement is first observed.
Bulk Viscosity and C*
Bulk viscosity is a property that can be used to establish C*. Within the dilute concentration regime, an increase in polymer concentration leads to a linear increase in the bulk viscosity. When chain entanglement occurs, the bulk viscosity deviates from linearity, due to the added viscoelastic effects of chain slippage and polymer interactions. Consider Figure 1 for example, which shows the concentration dependence of the bulk viscosity for a low molecular weight polymer (trade name Synapse) in an organic solvent, and indicates a C* value of ~ 50 mg/ml.
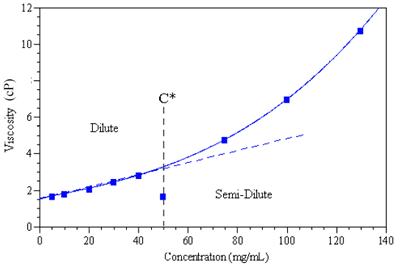
Figure 1. The concentration dependence of the bulk viscosity of Synapse in an organic solvent, indicating a C* value of ~ 50 mg/ml.
Scattering Intensity and Polymer Molecular Weight
In its simplest form, the scattering intensity of a polymer solution can be described as being proportional to the concentration and the square of the molecular weight. For a constant molecular weight polymer then, the scattering intensity should increase proportionally with the increase in sample concentration. For polymer solutions in the dilute region, a linear concentration dependence is the norm. Within the semi-dilute region however, multiple scattering reduces the number of photons arriving at the detector at higher concentrations, resulting in a decrease in scattering intensity with increasing concentration. The effects of multiple scattering are evident in Figure 2, which shows the concentration dependence of the scattering intensity and the bulk viscosity for the Synapse polymer, and indicates a peak in the sample scattering intensity that is consistent with C*.
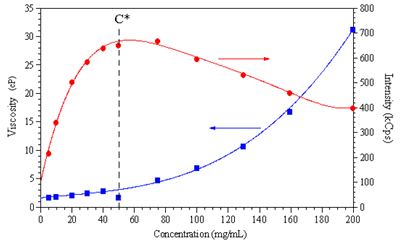
Figure 2. The residual scattering intensity and bulk viscosity of Synapse as a function of polymer concentration.
The size distributions for the Synapse polymer are shown in Figure 3, which indicates non-aggregated monodisperse distributions (~ 5.5 nm diameter) for all sample concentrations.
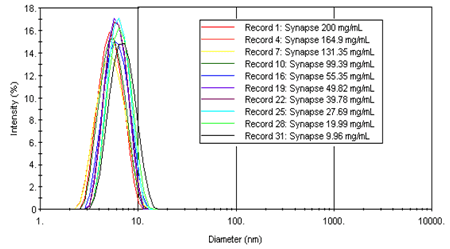
Figure 3. Intensity size distributions results for Synapse as a function of polymer concentration.
Concentration Dependence of the Scattering Intensity and Polymer Molecular Weight
The concentration dependence of the scattering intensity in the dilute regime can also be used to measure the molecular weight of the polymer. The Rayleigh ratio (Rθ) is the ratio of the sample to incident scattering intensities. For particles smaller than 100 nm diameter, the concentration dependence of the Rayleigh ratio can be described as given below, where K is an optical constant, C is the analyte concentration, M is the weight average molecular weight, and A2 is the 2nd virial coefficient (representative of the magnitude of particle - solvent interactions).
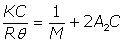
The Rayleigh expression given above is represented graphically in the form of a Debye plot showing the concentration dependence of KC/Rθ. The Debye plot for the Synapse data is shown in Figure 4, which indicates a molecular weight of 18.55 kDa and a 2nd virial coefficient of 0.001878 ml mol/g2.
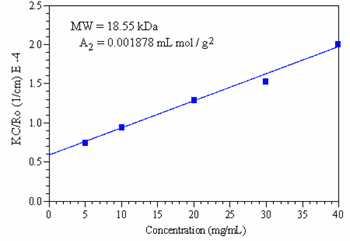
Figure 4. Debye plot for Synapse, showing the linearity of KC/Rθ with concentration, and indicating a molecular weight of 18.55 kDa and a 2nd virial coefficient of 0.001878 ml mol / g2.
Comparison with Other Data
The measured 18.55 kDa molecular weight of Synapse is consistent with size exclusion chromatography results, which give an estimated weight average molecular weight of 20 kDa. The positive value of the 2nd virial coefficient indicates that solvation is more energetically favourable than polymer aggregation, and suggests a high solubility limit in the organic solvent used in this study.
Summary
The size and mass measurements for the Synapse samples used in this study were collected using a Malvern Panalytical Zetasizer Nano ZS system. The Zetasizer Nano ZS is a static, dynamic, and electrophoretic light scattering instrument, and is the first commercial system to combine the technologies for accurate and absolute size, mass, and zeta potential measurements in a single instrument. The backscatter angle and the patented optical design of the system facilitates measurements across size (0.6 nm to 6 µm) and concentration (0.1 mg/ml lysozyme to 40 W%) ranges greater than any other commercially available light scattering instrument.

This information has been sourced, reviewed and adapted from materials provided by Malvern Panalytical.
For more information please visit Malvern Panalytical.