DOI :
10.2240/azojono0101
May 19 2005
Two of the 21st century’s most promising technologies are biotechnology and nanotechnology.
This science of nanoscale structures deals with the creation, investigation and utilization of systems that are 1000 times smaller than the components currently used in the field of microelectronics. Biotechnology deals with metabolic process with micro-oraganisms. Convergence of these two technologies results in growth of nanobiotechnology. This interdisciplinary combination can create many innovative tools.
The biomedical applications of nanotechnology are the direct products of such convergences.
However, the challenges facing Scientists and Engineers working in the field of nanotechnology are quite enormous and extraordinarily complex in nature.
Utility of nanotechnology to biomedical sciences imply creation of materials and devices designed to interact with the body at sub-cellular scales with a high degree of specificity. This could be potentially translated into targeted cellular and tissue-specific clinical applications aimed at maximal therapeutic effects with very limited adverse-effects.
Nanotechnology in biomedical sciences presents many revolutionary opportunities in the fight against all kinds of cancer, cardiac and neurodegenerative disorders, infection and other diseases.
This article presents an overview of some of the applications of nanotechnology in biomedical sciences.
Background
Nanotechnology is a new area of science that involves working with materials and devices that are at the nanoscale level. A nanometer is billionth of a meter. That is, about 1/80,000 of the diameter of a human hair, or ten times the diameter of a hydrogen atom. It manipulates the chemical and physical properties of a substance on molecular level. Nanotechnology alters the way we think, it blurs the boundaries between physics, chemistry and biology, the elimination of these boundaries will pose many challenges and new directions for the organization of education and research.
Richard Feynman’s speech called ‘There is plenty of room at the bottom’ in 1959 emphasised this concept - If our small minds, for some convenience, divide this universe into parts, physics, biology, geology, astronomy, psychology and so on – Remember that nature does not know it [1].
Nanobiotechnology is the unification of biotechnology and nanotechnology. This hybrid discipline can also mean making atomic-scale machines by imitating or incorporating biological systems at the molecular level, or building tiny tools to study or change natural structure properties atom by atom. Nanobiotechnology can have a combination of the classical micro-technology with a molecular biological approach. Biotechnology uses the knowledge and techniques of biology to manipulate molecular, genetic, and cellular processes to develop products and services, and is used in diverse fields from medicine to agriculture. Convergence, is an activity or trend that occurs based on common materials and capabilities-in this case the discipline that enables convergence is nanotechnology. The potential opportunities offered by this interface is truly outstanding; the overlap of biotech, nanotech and information technology is bringing to fruition many important applications in life sciences.
This technology is expected to create innovations and play a vital role in various biomedical applications (fig. 1), not only in drug delivery and gene therapy, but also in molecular imaging, biomarkers and biosensors. Target-specific drug therapy and methods for early diagnosis of pathologies are the priority research areas where nanotechnology would play a prominent role [2].
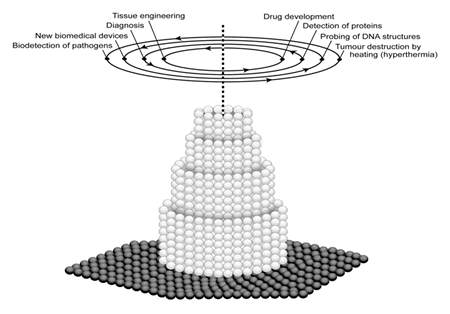
Figure 1. Schematic illustration of nanotechnology revolutionizing biomedical sciences.
The National Institutes of Health Bioengineering Consortium (BECON) held a symposium in 2000 entitled “Nanoscience and Technology: Shaping Biomedical Research″[3]. Eight areas of nanoscience and nanotechnology were addressed at the conference and believed to be the most pertinent to research in biomedicine. These areas included synthesis and use of nanostructures, applications of nanotechnology to therapy, biomimetic and biologic nanostructures, electronic-biology interface, devices for early detection of disease, tools for the study of single molecules, nanotechnology and tissue engineering.
The aim of BECON was to enhance communication between Biomedical Scientists and Engineers who bring different aspects of their skills and knowledge to bear on these problems and to make the biomedical community more aware of the emerging developments in the field of nanotechnology. The deliberations of the conference are now widely reinforced by day-to-day experience, increasing ability to manipulate individual molecules at a nanoscale and to combine biomolecules with other nanoscale structures. This ability provides the opportunity for untold new therapeutic and diagnostic applications by enabling the building of novel structures from the bottom up [4].
In the foreseeable future, the most important clinical application of nanotechnology will probably be in pharmaceutical development. These applications take advantage of the unique properties of nanoparticles as drugs or constituents of drugs or are designed for new strategies to controlled release, drug targeting, and salvage of drugs with low bioavailability [5-7].
Nanoscale polymer capsules can be designed to break down and release drugs at controlled rates, to allow differential release in certain environments, such as an acid medium, and to promote uptake in tumors versus normal tissues [8]. A lot of research is now focused on creating novel polymers and exploring specific drug-polymer combinations. Nanocapsules can be synthesized directly from monomers or by means of nanodeposition of preformed polymers [9]. Nanocapsules have also been formulated from albumin and liposomes. Implantable drug delivery systems that are being developed will make use of nanopores to control drug release.
One of the key issues in bio-availability is cell transfection in DNA gene therapy. Current methods have significant limitations, including the risk of inadvertent transmission of disease by viral vectors. This has led researchers to explore polymer-DNA complexes and liposome-DNA complexes for gene delivery [10]. It has also been shown that compacted DNA in the form of nanoparticles can be used to transfect postmitotic cells [11].
Despite the risk and limitations, viral vectors are an efficient biomimetic approach to drug targeting and delivery. The tat peptide from human immunodeficiency virus (HIV) and other viral proteins are being attached to DNA, proteins, and other materials for uptake into cells. These nano-assemblies mimic the action of the fusion proteins that make viral transfection efficient [12, 13]. Nanotechnology has also enabled the development of biochips and has a role in green manufacturing (e. g biocompatibility and biocomplexity areas). Other applications include the design of sensors for astronauts, soldiers, biofluids (for handling DNA and other molecules), in vitro fertilization of live stock, nanofiltration, bioprocessing ‘by design’ and traceability of genetically modified food (Table 1).
Table 1. List of Nanotechnology applications to Biomedical sciences.
|
|
|
|
Bio-detection of pathogens
|
15
|
|
|
Detection of proteins
|
16
|
|
|
Probing of DNA structure
|
17
|
|
|
Tissue engineering
|
18, 19
|
|
|
Heat destruction of tumour (hyperthermia)
|
20
|
|
|
Phagokinetic studies
|
21
|
|
|
MRI Contrast enhancement
|
22
|
|
|
Separation and purification of biomolecules and cells
|
23
|
|
|
Fluorescent biological markers
|
24, 25
|
|
|
Drug and gene delivery
|
26, 27
|
|
|
Artificial cells and their assemblies
|
28
|
|
Design of proteins for efficient electron transport or with mechanical features
|
29
|
|
Using dip pen technology
|
30, 31
|
|
Formation and growth of nanostructures in living biosystems (e.g by alfalfa plants)
|
32
|
|
Biosensors
|
33
|
|
Nanobiomotors
|
34-36
|
|
Biomineralization
|
37
|
|
Nanorobotics
|
14, 38
|
|
Nanocomputers
|
39
|
|
Nanorods for vaccination applications
|
40
|
Exploratory areas for nanotechnology will include research into the condition and/or repair of the brain and other areas for regaining cognition. It might also find application in designing pharmaceuticals as a function of patient genotypes and in applying chemicals to stimulate production as a function of plant genotypes. The synthesis of more effective and biodegradable chemicals for agriculture and the production of implantable detectors could be aided by nanotechnology with minimal quantities of blood. Employing this technology it should also be possible to develop methods that use saliva instead of blood for the detection of illnesses or that can perform complete blood testing within a short period of time. Broader issues include economic molecular medicine, sustainable agriculture, conservation of biocomplexity, and enabling emerging technologies.
Richard E. Smalley, winner of the 1996 Nobel Prize in Chemistry announced in his congressional testimony to the U.S. government about the increasing awareness in the scientific and technical community of our entry into a new golden age. Burgeoning interest in the medical applications of nanotechnology has led to the emergence of a new discipline known as nanomedicine [14]. On a wider scope, nanomedicine is the process of diagnosing, treating, preventing disease and traumatic injury, of relieving pain, and of preserving and enhancing human health, using molecular tools and molecular knowledge of the human body. The purpose of this review is to throw more light on the recent advances and impact of nanotechnology on biomedical sciences.
Recent Developments
Medical diagnosis with appropriate and effective delivery of pharmaceuticals are the medical areas where nanosize particles have found practical applications. However, there are many other interesting proposals for the use of nanomechanical tools in the fields of medical research and clinical practice. Such nanotools are awaiting construction, and presently are more like a fantasy. Nevertheless, they might be quite useful, and become a reality in the near future [41].
Nanodevices in medical sciences could function to replace defective or improperly functioning cells, such as the respirocyte proposed by Freitas [42]. This man-made red blood cell is theoretically capable of providing oxygen more effectively than an erythrocyte. It could replace defective natural red cells in blood circulation. Primary applications of respirocytes may involve transfusable blood substitution, partial treatment of anaemia, prenatal/neonatal problems, and lung disorders.
It has been reported that nanomachines could administer drugs within a patient’s body. Such nanoconstructions could deliver drugs to peculiar sites making treatment more accurate and precise [43]. Similar machines with specific ‘weapons’ could be used to remove obstacles in the circulatory system or in the identification and killing of tumor cells.
The other vital application of nanotechnology in relation to medical research and diagnostics are nanorobots. Nanorobots, operating in the human body, could monitor levels of different compounds and record the information in the internal memory. They could be rapidly used in the examination of a given tissue, surveying its biochemical, biomechanical, and histometrical features in greater detail. Just as biotechnology extends the range and efficacy of treatment options available from nanomaterials, the advent of molecular nanotechnology will again expand enormously the effectiveness, comfort and speed of future medical treatments while at the same time significantly reducing their risk, cost, and invasiveness.
Biotechnology permits tailor-made production and biopharmaceuticals and biotechnological drugs, many of which require special formulation technologies to overcome drug-associated problems. Such major challenges to solve include the following: poor solubility, limited chemical stability in vitro and in vivo after administration (i.e. short half-life), poor bioavailability and potentially strong side-effects requiring drug enrichment at the site of action (targeting) [44]. Nanoparticulate carriers have been developed as one solution to overcome such delivery problems, i.e. drug nanocrystals, solid lipid nanoparticles (SLN), nanostructured lipid carriers (NLC) and lipid-drug conjugate (LDC) nanoparticles [44]. The carriers as reported by Muller and colleagues are suitable to solve delivery problems with biotech drugs of different solubility. Targeting with these carriers can be realised by a very simple approach, the differential protein adsorption (PathFinder® technology). This technology proved to be efficient enough to accumulate sufficiently high amounts of drugs in the brain to reach therapeutic levels and also fulfill the major requirement to be pursued by a pharmaceutical company.
Quantum Dot with nanodots of a specific colour are believed to be flexible and could offer a cheap and easy way to screen a blood sample for the presence of a number of different viruses at the same time. It could also give physicians a fast diagnosis tool to detect, say, the presence of a particular set of proteins that strongly indicates the onset of myocardial infarction. On the research front, the ability to simultaneously tag multiple biomolecules both on and inside cells could allow scientists to watch the complex cellular changes and events associated with disease, providing valuable clues for the development of future pharmaceuticals and therapeutics (Quantum Dot Corporation) [45].
The National Heart, Lung, and Blood Institute (NHLBI) plans to foster the application of nanotechnology to HLBS (Heart, Lung, Blood and Sleep) research and disorders. A request for information (RFI) was developed, with advice from scientists and physicians with interests in nanotechnology, to canvas the broader scientific community on approaches to developing and applying nanotechnology to HLBS disorders. A working Group comprising scientists, engineers, and physicians with expertise across nanotechnology, nanoscience, and HLBS medicine met on February 28th, 2003, using the RFI responses as the starting point for discussions. The Working Group was entrusted with assessing the field of nanotechnology and suggesting ways for research. The Working Group cautioned against overly rigid or restrictive definition of nanotechnology, emphasizing the continuum of scale from the nanoscale to the microscale. The Group also identified areas of opportunity and challenges to further development associated with the application of nanoscience and nanotechnology to improved diagnosis, treatment, and prevention of HLBS disorders. It as well developed prioritized recommendations to facilitate the application of nanotechnology to biological questions and improved patient care [46].
The RESIST Group at the Welsh School of Pharmacy at Cardiff University and others have looked at how molecularly imprinted polymers could be medically useful in clinical applications such as controlled drug release, drug monitoring devices, and biological and antibody receptor mimics. Histamine and ephedrine molecularly imprinted polymers (MIPs) were studied as potential biological receptor mimics whilst a propanolol MIP was investigated for its use as a rate attenuating selective excipient in a transdermal controlled device [47].
The first artificial voltage-gated molecular nanosieve was fabricated by Charles R. Martin and colleagues [48] at Colorado State University in 1995. Martin’s membrane contains an array of cylindrical gold nanotubules with inside diameters as small as 1.6nm. When the tubules are positively charged, positive ions are excluded and only negative ions are transported through the membrane. When the membrane receives a negative voltage, only positive ions can pass. Similar nanodevices may combine voltage gating with pore size, shape, and charge constraints to achieve precise control of ion transport with significant molecular specificity. An exquisitely sensitive ion channel switch biosensor was built by an Australian research group [49].
The year 2003 could be termed a very special year for biomedical research because we celebrated the completion of the sequencing of the entire human genome which coincided with the 50th anniversary of the discovery of the DNA double helix structure by Watson and Crick. In biomedical imaging, we also witnessed the awarding of the Nobel Prize in Medicine and Physiology to two pioneers in Magnetic Resonance Imaging, Professor Paul Lauterbur and Sir Peter Mansfield. These landmark events helped to highlight the impact of the rapid development in many diverse disciplines to biomedical research. The leverage and tremendous advances in electronics and information technology has been brought about by biomedical imaging research [50]. The opportunities and challenges in future biomedical research lie in the incorporation of knowledge gained from molecular biology with chemistry, physics, engineering, information technology, and nanotechnology to understand the ambiguity and complexity of life and come up with new diagnostic and therapeutic methods.
Calcium phosphate nanoparticles present a unique class of non-viral vectors, which can serve as efficient and alternative DNA carriers for targeted delivery of genes. The design and synthesis of ultra-low size, highly monodispersed DNA doped calcium phosphate nanoparticles of size around 80nm in diameter has been reported [51]. The DNA encapsulated inside the nanoparticle is protected from the external DNase environment and could be used safely to transfer the encapsulated DNA under in vitro and in vivo conditions.
The application of a combination of nanomedicine with biophotonics for optically tracking the cellular pathways of gene delivery and the resulting transfection by using nanoparticles as a non-viral vector has been demonstrated recently [52]. Gene delivery is an area of considerable current interest; genetic materials (DNA, RNA, and oligonucleotides) have been used as molecular medicine and are delivered to specific cell types to either inhibit some undesirable gene expression or express therapeutic proteins.
Nano-DNA Technology
The discovery of the polymerase chain reaction (PCR) [53, 54] paved the way to a new era of biological research. The impact can be felt not only in the field of molecular biology, but also in other allied fields of science. Novel classes of semi-synthetic DNA-protein conjugates, self-assembled oligomeric networks consisting of streptavidin and double-stranded DNA, which can be converted into well-defined supramolecular nanocircles have been developed [55, 56].
The DNA-streptavidin conjugates are applicable as modular building blocks for the production of new immunological reagents for the ultrasensitive trace analysis of proteins and other antigens by means of immuno-PCR methodology [57-59]. Immuno-PCR is a combination of the specificity of an antibody-based immuno-assay with the exponential power of the amplification of PCR, hence resulting in a 1000-fold degree of sensitivity as compared with standard ELISA (Enzyme-linked immunosorbent assay) methods.
Self-assembled DNA-streptavidin conjugates have also been applied in the field of nanotechnology. For example, the conjugates are used as model systems for ion-switchable nanoparticle networks, as nanometre-scale ‘soft material’ calibration standards for scanning probe microscopy [60, 61], or as programmed building blocks for the rational construction of complex biomolecular architecture, which may be used as templates for the growth of nanometre-scale inorganic devices [62, 63]. Covalent conjugates of single-stranded DNA and streptavidin are used as biomolecular adapters for the immobilization of biotinylated macromolecules at solid substrates through nucleic acid hybridization. This ‘DNA-directed immobilization’ allows for reversible and site-selective functionalization of solid substrates with metal and semiconductor nanoparticles or, vice versa, for the DNA directed functionalization of gold nanoparticles with proteins, such as immunoglobulins and enzymes. The fabrication of functional biometallic nanostructures from gold nanoparticles and antibodies are applied as diagnostic tools in bioanalytics [64].
Nanobiotechnology in High-Throughput Single Nucleotide Polymorphism Analysis
Following the publication of a map of variation in the human genome sequence containing over two million single nucleotide polymorphisms (SNPs) (The International SNP Map Working Group, 2001), the next challenge is the development of the technologies to use this information in a cost-effective manner. Genotyping methods have to be improved in order to increase throughput by at least two orders of magnitude to enable pharmaceutical, biotechnological and academic research to uncover associations between genetic variants and diseases, with consequent potential for the development of novel diagnostics and therapies. New approaches to DNA extraction and amplification have curtailed the times required for these processes to seconds. Microfluidic devices enable polymorphism detection through very rapid fragment separation using capillary electrophoresis and high-performance liquid chromatography, together with mixing and transport of reagents and biomolecules in integrated systems [65]. The basic objectives in the development of a DNA extraction and purification system that will be compatible with high-throughput SNP genotyping requirements are:
• Release of the DNA from the cells into solution without either enzymatic (i.e. endonucleases) or mechanical (shearing) breakdown of the DNA;
• Removal of cellular debris (e.g. proteins) that may hamper DNA amplification or hybridization assays;
• High-throughput and economical DNA sample preparation with simplified protocols that reduce the number of procedures involved;
• Avoidance of hazardous chemical requirements as much as possible to minimize handling and disposal costs;
• Consistency of both quality and quantity of DNA yield among samples so that quantification is unnecessary, and subsequent amplification and/or hybridization can be to a high degree of reproducibility;
• A highly efficient process, to ensure enough supply for the enormous number of assays anticipated; and
• An interface that will enable direct loading of conventionally sampled biopsies on to the system [65].
The potential for nanotechnology to contribute to rapid high-throughput SNP analysis is most evident with smart biochip platforms. The development of an electronically addressable microarray platform as described by Heller L. et al 2000 [66] has given rise to Nanogen Inc. (San Diego, California, USA). The challenge of providing one or more technology platforms capable of SNP screening throughput of the order of 107 genotypes per day will need to be achieved, to allow significant associations between genes and diseases to be established. Additionally, the technology platform(s) will also need to deliver economies of scale, such that the cost per genotype will be less than 0.01$ for the magnitude of screening necessary to be feasible. From the rapidly developing field of nanotechnology, novel tools and processes have been introduced with the potential to provide the capabilities required [67-69].
Differences of SNPs occurring in close proximity to each other on the genome is normally correlated due to linkage during the process of replication, and the extent of this correlation is termed linkage disequilibrium. Where a significant association occurs between the genetic variation observed at specific SNPs and the presence of a disease, susceptible genes can be identified. The statistical estimations needed to eliminate false-positive results were reviewed by McCarthy and Hilfiker (2000) [70]. They suggest a linear increase in sample size is necessary for every order of magnitude increase in the number of markers tested. Hence, positive identification of a susceptible gene from a screening programme including 1 Million SNPs would require a minimum sample size of 1000 (i.e. a minimum of 109 SNPs have to be screened).
Nanoparticles as Biomarkers
Nanoparticles can be used for both quantitative and qualitative in vitro detection of tumor cells. They enhance the detection process by concentrating and protecting a marker from degradation, in order to render the analysis more sensitive. For instance, streptavidin-coated fluorescent polystyrene nanospheres Fluospheres® (green fluorescence) and TransFluospheres® (red fluorescence) were applied in single colour flow cytometry to detect the epidermal growth factor receptor (EGFR) on A431 cells (human epidermoid carcinoma cells) [71]. The results have shown that the fluorescent nanospheres provided a sensitivity of 25 times more than that of the conjugate streptavidin-fluorescein.
New tools can now be developed, designed at the intersection of proteomics and nanotechnology, whereby nanoharvesting agents can be instilled into the circulation (e.g. derivatized gold particles) or into the blood collection devices to act as ″molecular mops″ that soak up and amplify the bound and complexed biomarkers that exist [72-74]. These nanoparticles, with their bound diagnostic cargo, can be directly queried via mass spectrometry to reveal the low molecular weight and enriched biomarker signatures. Ultimately, utility of any approach for detecting disease is assessed on its clinical impact to patient outcome and disease-free survival [75]. What is urgently required in the study of diseases in general, is the development of biomarkers that can detect curable diseases earlier, and not detecting advanced disease better.
Contrast agents have been loaded onto nanoparticles for tumor diagnosis purposes. The physico-chemical features (particle size, surface charge, surface coating, stability) of the nanoparticles allow the redirection and the concentration of the marker at the specific site of interest. Labelled colloidal particles could be used as radiodiagnostic agents. On the other hand, some non-labelled colloidal systems are already in use and some are still being tested as contrast agents in related diagnosis procedures such as computed tomography and NMR imaging.
To date, a study of radionucleide use in diagnostic imaging with nanoparticles for cancer detection is yet to be published. However, as conventional colloidal particles can be cells of organs like the liver, the spleen, the lungs and the bone marrow and as long-circulating nanoparticles can have a compartmental localization in the blood circulation or the lymphatic system- all these organs being potential sites for tumor development, these colloidal systems could potentially improve tumor diagnosis.
In the future, nanoparticles that are engineered with specific binding affinities can be resuspended into the collected body fluids, or perhaps even injected directly into the circulation. The nanoparticles, together with the bound molecules, could be directly captured on engineered filters and directly questioned by ultra high-resolution mass spectrometry (e. g. Fourier Transform Ion Cyclotron Resonance).
Nanotechnology in Measurements of Dissolved Oxygen
Oxygen is one of the major metabolites in aerobic systems, and the measurement of dissolved oxygen is of vital importance in medical, industrial, and environmental applications. Recent interest in the methods for measuring dissolved oxygen concentration has been focused mainly on optical sensors, due to their advantages over conventional amperometric electrodes in that they are faster, do not consume oxygen, and are not easily poisoned [76, 77].
Optical PEBBLE (probes encapsulated by biologically localized embedding) nanosensors have been developed for dissolved oxygen using organically modified silicate (ormosil) nanoparticles as a matrix. The ormosil nanoparticles are prepared through a sol-gel-based process, which includes the formation of core particles with phenyltrimethoxysilane as a precursor followed by the formation of a coating layer with methyltrimethoxysilane as a precursor [78]. The highly permeable structure and the hydrophobic nature of the ormosil nanoparticles, as well as their small size, result in an excellent overall quenching response to dissolved oxygen and a linear response over the whole range, from 0 -100% oxygen-saturated water. This PEBBLE sensor has a higher sensitivity and a broader linearity as well as longer excitation and emission wavelengths, resulting in reduced background noise for cellular measurement. The PEBBLE sensors are excellent in terms of their reversibility and stability to leaching and long-term storage. A real-time monitoring of changes in the dissolved oxygen due to cell respiration in a closed chamber was made by gene gun delivered PEBBLE. This sensor is now being applied for simultaneous intracellular measurements of oxygen and glucose [78].
Application of Nanotechnology to P450 Enzymes
Cytochromes P450 are highly relevant to the bio-analytical area [79]. They form a large family of enzymes present in all tissues essential to the metabolism of most drugs in use today, playing a vital role in the drug development and discovery process. They act as catalysts for the insertion of one of the two atoms of an oxygen molecule into a variety of substrates (R) with quite broad regioselectivity, leading to concomitant reduction of the other oxygen atom to water as shown in the equation below [29].
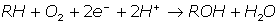
Several methods have been reported in the literature for the screening of substrate turnover by P450s in a high throughput format [80-83]. However, they all fall short of being limited to testing the activity of P450 enzymes through the detection of the conversion of a specific marker substrate, but Tsotsou et al 2002 [84] have been able to develop a method called the alkali method, which can detect the turnover of any NAD(P)H or NAD(P)+ dependent enzyme. The progress on these research fronts and their combinations provide a powerful platform for future applications of these enzymes, with particular reference to protein array technology.
Application of Nanotechnology to Tissue Engineering
Tissue engineering is based on the creation of new tissues in vitro followed by surgical placement in the body or the stimulation of normal repair in situ using bioartificial constructs or implants of living cells introduced in or near the area of damage. Though it is mainly concerned with using human material, either from the patient themselves (autologous) or from other human sources (allogeneic), material from other mammalian sources have also been applied in humans (xenogeneic).
The involvement of microelectronics or nanotechnology in creating a truly bioartificial tissue or organ that can take the place of one that is terminally diseased, such as an eye, ear, heart, or joint has been envisaged. Implantable prosthetic devices and nanoscaffolds for use in the growing of artificial organs are goals of nanotechnology researchers. Nanoengineering of hydroxyapatite for bone replacement is reasonably advanced [85, 86].
In the future, we could imagine a world where medical nanodevices are routinely implanted or even injected into the bloodstream to monitor wellness and to automatically participate in the repair of systems that deviate from established norms. These nanobots could be personalized by tailoring them to patient genotype and phenotype to optimize intervention at the earliest stage in the course of disease expression [4].
Growth of New Organs
Nanoscale building of cells can be accomplished by their programmed replication. The signals are transmitted back and forth with the instruction for the desired size and shape form the construction site. When complete instructions are finished, the organs can be grown according to the prerequisite specifications.
These organs could have the necessary DNA encoded to be compatible with the required human body immunological status. This can enhance integration of artificial structures with living tissues, presenting a more appropriate interface to biological systems. With the advantage in absence of immune reaction unlike today’s donor organ transplantation. In the years to come this can accomplish a Quantum leap in the management of organ failure disorders.
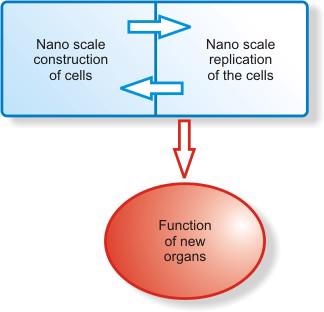
Figure 2. Graphical representation of the nanoscale construction and growth of new organs.
Molecular Imaging
New imaging approaches using genetically encoded fluorescent and bioluminescent reporters (i.e., illuminated or glowing identification tags) are offering revealing insights to the living body as never observed before. Information provided by these reporters can be used to enhance our understanding of human biology and the development of therapeutic approaches for many diseases, including cancer, infection, neurodegenerative and cardiovascular disease.
In addition to progresses so far made with molecular agents, industry leaders are also showcasing rapidly evolving imaging technologies that allow scientists to view organisms at the molecular level (Table 2).
Table 2. Latest products in Molecular Imaging and associated producing companies.
|
|
|
SPECT/CT hybrid imaging systems
|
Philips Medical Systems/Siemens Medical Solutions
|
|
|
GFAP-luc (glial fibrillary acid protein)
|
Xenon
|
|
|
Ultrasound bubbles
|
Schering AG
|
|
|
NeuroSpec™ (radiodiagnostic agent)
|
Tyco Healthcare/Mallinckrodt Inc.
|
|
|
eXplore Locus Ultra (Volumetric CT system)
|
GE Medical system
|
|
Definity® or Sonolysis™ (nanosurgery)
|
ImaRx
|
• SPECT/CT hybrid systems capture both functional information on molecular and cellular processes (growth and activity) and anatomical detail (size and shape) of a targeted molecular structure more quickly, efficiently and clearly than standard imaging devices. The images obtained from these systems can assist with the rapid identification of tumours, analysis of appropriate treatment, delivery of targeted therapy to precisely destroy target cells, and follow up to assess treatment effectiveness.
• Xenon presented its newer light producing transgenic animal models (GFAP-luc) during the Society for Molecular Imaging’s 3rd Annual Meeting. This model may prove to be an important model for tracking damage and repair in chronic neurological conditions such as post-ischemic stroke or Parkinson’s disease.
• An ultrasound contrast agent is made of tiny “microbubbles″ that scatter light and allow the clinician to see which part of the heart muscle is poorly functioning. The sensitivity and flexibility of ultrasound makes it the most sensitive method of imaging microbubbles because it deliberately disrupts the pattern and produces a very strong and highly characteristic transient effect. For example,
• Definity® otherwise known as Sonolysis™ are gas-filled microbubbles for novel therapeutic applications. For dissolving vascular thrombosis, microbubbles are administered intravenously to a patient or injected locally into a specific vascular structure such as a vascular graft. Ultrasound is applied externally (or can be applied internally via catheter) over the area of the blood clot to provide localized, targeted action. As the microbubbles perfuse the clot, they act as micromechanical devices where ultrasound pulses the bubbles and blows up the bubbles in the ultrasound field, leading to blood clot dissolution. Sonolysis nanosurgery is locally targeted nanoinvasive therapy for treatment of vascular thrombosis. Compared with alternative therapies for treating thrombosis, sonolysis affords the potential merits of being less invasive than mechanical thrombectomy and faster than conventional drug therapy with less risk of bleeding.
• NeutroSpec™ is a radiodiagnostic agent which labels white blood cells and myeloid precursors without the need for removal and re-injection of blood into patients. This new product is for patients with equivocal signs of appendicitis who are five-years-old and up. NeutroSpec also facilitates the visualization of images generated via gamma camera allowing physicians to quickly and easily locate the sites of infection thereby eliminating time delays and/or risks normally affiliated with alternative white blood cell labelling processes.
• eXplore Locus Ultra is a first-class volumetric CT system capable of quantitating physiological measurements and elaborate anatomy of tissues, tumours and organ perfusion. The Locus Ultra also performs image acquisition at the rate of a sub-second, enabling dynamic imaging.
Summary
The multidisciplinary field of nanotechnology’s application for discovering new molecules and manipulating those available naturally could be dazzling in its potential to improve health care. The spin-offs of nanobiotechnology could be utilised across all the countries of the world.
In the future, we could imagine a world where medical nanodevices are routinely implanted or even injected into the bloodstream to monitor health and to automatically participate in the repair of systems that deviate from the normal pattern. The continued advancement in the field of biomedical nanotechnology is the establishment and collaboration of research groups in complementary fields. Such collaborations have to be maintained not only on specialty field level, but internationally as well. The successful development and implementation of international collaborations fosters a global perspective on research and brings together the benefits to mankind in general.
However, nanotechnology in medicine faces enormous technical hurdles in that long delays and numerous failures are inevitable. Likewise, it should not be taken for granted the dangers and negative consequences of nanobiotechnology when applied in warfare, in the hands of terrorists and disasters associated with its application in energy generation when and wherever it strikes or the risks associated with nanoparticles in blood circulation. It should be appreciated that nanotechnology is not in itself a single emerging scientific discipline but rather a meeting point of traditional sciences like chemistry, physics, biology and materials science to bring together the required collective knowledge and expertise required for the development of these novel technologies.
Acknowledgements
The authors wish to express their gratitude to Prof. Guy M. Tremblay and Dr. Jakob Bonlokke for their critical review of the manuscript and helpful suggestions and also Ms Cecile Bilodeau, audio-visual department for designing Figure 1.
References
1. Richard Feynman, “Six Easy Pieces”, Addison-Wesley Pub. Co., Menlo Park, CA, 1963.
2. Sahoo K. S. and Labhasetwar V. “Nanotech Approaches to Drug Delivery and Imaging”, DDT Vol. 8, No. 24, 1112-1120, 2003.
3. BECON Nanoscience and Nanotechnology Symposium Report, June (2000). National Institutes of Health Bioengineering Consortium, 2000. National Institute of Health web site, accessed March 20th, 2005.
4. Thrall J. H. “Nanotechnology and Medicine. Radiology”, Vol. 230(2), 315-318, 2004.
5. Roy I., Ohulchanskyy T. Y., Pudavar H.E., et al., “Ceramic-Based Nanoparticles Entrapping Water-Insoluble Photosensitizing Anticancer Drugs: A Novel Drug-Carrier System for Photodynamic Therapy”, J. Am. Chem. Soc., 125, 7860-7865, 2003.
6. Brigger I., Dubernet C., and Couvreur P., “Nanoparticles in Cancer Therapy and Diagnosis”, Adv. Drug Deliv. Rev., 54,631-651, 2002.
7. Crommelin D. J., Storm G., Jiskoot W., Stenekes R., Mastrobattista E. and Hennink W. E., “Nanotechnological Approaches for The Delivery of Macromolecules”, J. Control Release, 87,81-88, 2003.
8. Na K. and Bae Y. H., “Self-Assembled Hydrogel Nanoparticles Responsive to Tumour Extracellular Ph From Pullulan Derivative/Sulphonamide Conjugate: Characterization, Aggregation and Adriamycin Release In Vitro”, Pharm. Res., 19,681-683, 2002.
9. Couvreur P., Barratt G., Fattal E., Legrand P. and Vauthier C., “Nanocapsule Technology: A Review”, Crit. Rev. Ther. Drug. Carrier Syst., 19, 99-134, 2002.
10. Xu L., Frederik P. and Pirollo K.F., “Self-Assembly of A Virus-Mimicking Nanostructure System for Efficient Tumour-Targeted Gene Delivery”, Hum. Gene Ther., 13, 469-481, 2002.
11. Liu G., Li D., Pasumarthy M.K., et al., “Nanoparticles of Compacted DNA Transfect Postmitotic Cells”, J. Biol. Chem., 278, 32578-32586, 2003.
12. Lewin M., Carlesso N., Tung C. H., et al, “Tat Peptide-Derivatized Magnetic Nanoparticles Allow in Vivo Tracking and Recovery of Progenitor Cells”, Nat. Biotechnol., 18, 410-413, 2000.
13. Reynolds A. R., Moein Moghimi S. and Hodivala-Dilke K., “Nanoparticle Mediated Gene Delivery to Tumour Neovasculature”, Trends Mol. Med., 9, 2-4, 2003.
14. Robert A. Freitas Jr., “Nanomedicine”, Volume I: Basic Capabilities, Landes Bioscience, Georgetown, TX, 1999
15. Edelstein R. L., Tamanaha C. R., Sheehan P.E., Miller M. M., Baselt D. R., Whitman L. J. and Colton R. J., “The Barc Biosensor Applied to The Detection of Biological Warfare Agents”, Biosensors Bioelectron., 14, 805-813, 2000.
16. Nam J. M., Thaxton C. C. and Mirkin C. A., “Nanoparticles-Based Bio-Bar Codes for The Ultrasensitive Detection of Proteins”, Science, 301,1884-1886, 2003.
17. Mahtab R., Rogers J. P. and Murphy C. J., “Protein-Sized Quantum Dot Luminescence can Distinguish Between ‘Straight’, ‘Bent’ and ‘Kinked’ Oligonucleotides”, J. Am. Chem. Soc., 117, 9099-9100, 1995.
18. Ma J., Wong H., Kong L. B. and Peng K. W., “Biomimetic Processing of Nanocrystalline Bioactive Apatite Coating on Titanium”, Nanotechnology, 14,619-623, 2003.
19. de la Isla A., Brostow W., Bujard B., Estevez M., Rodriguez J. R., Vagas S. and Castano V. M. “Nanohybrid Scratch Resistant Coating for Teeth and Bone Viscoelasticity Manifested in Tribology”, Mat. Resr. Innovat., 7, 110-114, 2003.
20. Yoshida J. and Kobayashi T., “Intracellular Hyperthermia for Cancer Using Magnetite Cationic Liposomes”, J. Magn. Mater., 194,176-184, 1999.
21. Parak W. J., Boudreau R., Gros M. L., Gerion D., Zanchet D., Micheel C. M., Williams S. C., Alivisatos A. P. and Larabell C. A., “Cell Motility and Metastatic Potential Studies Based on Quantum Dot Imaging of Phagokinetic Tracks”, Adv. Mater., 14,882-885, 2002.
22. Weissleder R., Elizondo G., Wittenburg J., Rabito C. A., Bengele H. H. and Josephson L., “Ultrasmall Superparamagnetic Iron Oxide:Characterization of a New Class of Contrast Agents for MR Imaging”, Radiology, 175,489-493, 1990.
23. Molday R. S. and Mackenzie D., “Immunospecific Ferromagnetic Iron Dextran Reagents for The Labelling and Magnetic Separation of Cells”, J. Immunol. Methods, 52, 353-367, 1982.
24. Bruchez M., Moronne M., Gin P., Weiss S. and Alivisatos A. P., “Semiconductor Nanocrystals As Fluorescent Biological Labels”, Science, 281, 2013-2016, 1998.
25. Chan W. C. W. and Nie S. M., “Quantum Dot Bioconjugates For Ultrasensitive Nonisotopic Detection”, Science, 281 , 2016-2018, 1998.
26. Mah C., Zolotukhin I., Fraites T. J., Dobson J., Batich C. and Byrne B.J., “Microsphere-Mediated Delivery of Recombinant AAV Vectors In Vitro and In Vivo”, Mol. Therapy, I, S239, 2000.
27. Panatarotto D., Prtidos C.D., Hoebeke J., Brown F., Kramer E., Briand J.P., Muller S., Prato M., and Bianco A., “Immunization with Peptide-Functionalized Carbon Nanotubes Enhances Virus-Specific Neutralizing Antibody Responses”, Chemistry & Biology, 10, 961-966, 2003.
28. Pohorille A. and Deamer D. “Artificial Cells: Prospects for Biotechnology”, Trends Biotechnol., 20, 123-128, 2002.
29. Gilardi G., Meharenna Y. T., Tsotsou G.E., Sadeghi S.J., Fairhead M. and Giannini S., “Molecular Lego: Design of Molecular Assemblies of P450 Enzymes for Nanobiotechnology”, Biosensors & Bioelectronics, 17, 133-145, 2002.
30. Hyun J., Ahn S.J., Lee W.K., Chilkoti A. and Zauscher S., “Molecular Recognition-Mediated Fabrication of Protein Nanostructures by Dip-Pen Lithography”, Nano. Lett., 2, 1203-1207, 2002.
31. Liu G. and Amro N., “Positioning Protein Molecules on Surfaces: A Nanoengineering Approach to Supramolecular Chemistry”, Proc. Natl. Acad. Sci. USA, 99, 5165-5170, 2002.
32. Gardea-Torresdey J.L., Parsons J.G., Gomez E., Peralta-Videa J., Troiani H.E., Santiago P., and Yacaman M.J., “Formation And Growth of Au Nanoparticles Inside Alfalfa Plants”, Nano. Lett., 2, 397-401, 2002.
33. Tiefenauer L. and Ros R., “Biointerface Analysis on A Molecular Level: New Tools for Biosensor Research”, Colloids Surfaces B: Biointerfaces, 23, 95-114, 2002.
34. Moore S. and Prevelige P., “DNA Packaging: A New Class of Molecular Motors”, Curr. Biol., 12, R96-R98, 2002.
35. Liu H., Schmidt J.J., Bachand G.D., Rizk S.S., Looger L.L., Hellinga H.W. and Montemagno C.D., “Control of A Biomolecular Motor Powered Nanodevices with an Engineered Chemical Switch”, Nat. Mater., 1, 173-177, 2002.
36. Roco M.C., “International Strategy for Nanotechnology Research and Development”, J. Nanoparticle Res., 3, 353-360, 2001.
37. Banfield J.F. and Navrosky A., “Nanoparticles and The Environment”, Rev. Mineralogy Geochemistry, 44, 6-16, 2002.
38. Drexler E.K., “Nanosytems: Molecular Machinery, Manufacturing and Computation”, John Wiley & Sons, New York, 1992.
39. Collier C.P., Mattersteig G., Wong E.W., Luo Y., Beverly K., Sampaio J., Raymo F.M., Stoddart J.F. and Heath J.R., “A (2) Catenane-Based Solid State Electronically Reconfigurable Switch”, Science, 289, 1172-1175, 2000.
40. Salem A.K., Hung C.F., Kim T.W., Wu T.C., Searson P.C. and Leong K.W., “Multi-Component Nanorods for Vaccination Applications”, Nanotechnology, 16, 484-487, 2005.
41. Kubik-Bogunia K. and Sugisaka M., “From Molecular Biology to Nanotechnology and Nanomedicine”, Biosystems, 65, 123-138, 2002.
42. Freitas R.A., “The Future of Computers”, Analog., 116, 57-73, 1996.
43. Triggle D.J. Rho Chi lecture, “Pharamaceutical Sciences in The Next Millennium”, Ann Pharmacother., 33, 241-246, 1999.
44. Muller R.H. and Keck C.M., “Challenges and Solutions for The Delivery of Biotech Drugs-A Review of Drug Nanocrystal Technology and Lipid Nanoparticles”, J. Biotechnol., 113, 151-170, 2004.
45. Quantum Dot Corporation web site accessed on April 20th 2005.
46. Buxton D.B., Lee S.C., Wickline S.A. and Ferrari M., “Recommendations of The National Heart, Lung, and Blood Institute Nanotechnology Working Group”, Circulation, 108, 2737-2742, 2003.
47. Allender C.J., Richardson C., Woodhouse B., Heard C.M. and Brain K.R., “Pharmaceutical Applications for Molecularly Imprinted Polymers”, Int. J. Pharm., 195, 39-43, 2000.
48. Nishizawa M., Menon V.P. and Martin C.R., “Metal Nanotubule Membranes with Electrochemically Switchable Ion-Transport Selectivity”, Science, 268, 700-702, 1995.
49. Cornell B., Braach-Maksvytis V., King L., Osman P., Raguse B., Wieczorek L. and Pace R., “A Biosensor that Uses Ion-Channel Switches”, Nature, 387, 580-583, 1997.
50. Li K.C., Pandit S.D., Guccione S. and Bednarski M.D., “Molecular Imaging Applications in Nanomedicine”, Biomedical Microdevices, 6 (2), 113-116, 2004.
51. Roy I., Mitra S., Maitra A. and Mozumdar S., “Calcium Phosphate Nanoparticles as Novel Non-Viral Vectors for Targeted Gene Delivery”, International Journal of Pharmaceutics, 250, 25-33, 2003.
52. Roy I., Ohulchanskyy T.Y., Bharali D.J., Pudavar H.E., Mistretta R.A., Kaur N. and Prasad P.N., “Optical Tracking of Organically Modified Silica Nanoparticles as DNA Carriers: A Non-viral, Nanomedicine Approach for Gene Delivery”, PNAS, 102 (2), 279-284, 2005.
53. Mullis K., Faloona F., Scharf S., Saiki R., Horn G. and Erlich H., “Specific Enzymatic Amplification of DNA In Vitro: The Polymerase Chain Reaction”, Cold Spring Harbor Symp. Quant. Biol., 51, 263-273, 1986.
54. Saiki R.K., Bugawan T.L., Horn G.T., Mullis K.B. and Erlich H.A., “Analysis of Enzymatically Amplified B-Globin and Hladqa DNA with Allele-Specific Oligonucleotide Probes”, Nature, 324, 163-166, 1986.
55. Niemeyer C.M., Adler M., Pignataro B., Lenhert S., Gao S., Chi L.F., Fuchs H. and Blohm D., “Self-Assembly of DNA-Streptavidin Nanostructures and Their Use as Reagents In Immunio-PCR”, Nucleic Acids Res., 27, 4553-4561, 1999.
56. Niemeyer C.M., Adler M., Gao S. and Chi L.F., “Supramolecular Nanocircles Consisting of Streptavidin and DNA”, Angew. Chem. Int. Ed. Engl., 39, 3055-3059, 2000.
57. Niemeyer C.M., Wacker R. and Adler M., “Hapten Functionalized DNA-Streptavidin Nanocircles as Supramolecular Reagents in a Competitive Immuno-PCR Assay”, Angew. Chem. Int. Ed. Engl., 40, 3169-3172, 2001.
58. Adler M., Langer M., Witthohn K., Eck J., Blohm D. and Niemeyer C.M., “Detection of rViscumin in Plasma Samples by Immunio-PCR”, Biochem. Biophys. Res. Commun., 300, 757-763, 2003.
59. Adler M., Wacker R. and Niemeyer C.M., “A Real-Time Immuno-PCR Assay for the Ultrasensitive Quantification of Proteins Suitable for Routine Diagnostics”, Biochem. Biophys. Res. Commun., 308, 240-250, 2003.
60. Gao S., Chi L.F., Lenhert S., Anczykowsky B., Niemeyer C.M., Adler M. and Fuchs H., “High Quality Mapping of DNA-Protein Complex by Dynamic Scanning Force Microscopy”, Chem. Phys. Chem., 2, 384-388, 2001.
61. Pignataro B., Chi L.F., Gao S., Anczykowsky B., Niemeyer C.M., Adler M. and Fuchs H., “Dynamic Scanning Force Microscopy Study of Self-Assembled DNA-Protein Oligomers”, Apply. Phys. A 74, 447-452, 2002.
62. Niemeyer C.M., “Nanotechnology. Tools for the Biomolecular Engineer”, Science, 297, 62-63, 2002.
63. Keren K., Krueger M., Gilad R., Ben-Yoseph G., Sivan U. and Braun E., “Sequence-Specific Molecular Lithography on Single DNA Molecules”, Science, 297, 72-75, 2002.
64. Niemeyer C.M., “Semi-synthetic DNA-Protein Conjugates: Novel Tools in Analytics and Nanobiotechnology”, Biochemical Society Transactions, 32 (part 1), 2004.
65. Galvin P., “A Nanobiotechnology Roadmap for High-Throughput Single Nucleotide Polymorphism Analysis”, Psychiatric Genetics, 12, 75-82, 2002.
66. Heller M.J., Forster A.H. and Tu E., “Active Microelectronic Chip Devices which Utilize Controlled Electrophoretic Fields for Multiplex DNA Hybridization and other Genomic Applications”, Electrophoresis, 21, 157-164, 2000.
67. Laval J.M., Mazeran P.E. and Thomas D., “Nanobiotechnology and Its Role in The Development of New Analytical Devices”, Analyst, 125, 29-33, 2000.
68. Vo-Dinh T., Cullum B.M. and Stokes D., “Nanosensors and Biochips: Frontiers in Biomolecular Diagnostics”, Sensors Actuators, B 74, 2-11, 2001.
69. Guetens G., Van Cauwenberghe K., De Boeck G., Maes R., Tjaden U.R., van der Greef J. et al. “Nanotechnology in Bio/Clinical Analysis”, J. Chromatogr., B 739, 139-150, 2000.
70. McCarthy J.J. and Hilfiker R., “The Use of Single Nucleotide Polymorphism Maps in Pharmacogenomics”, Nat. Biotechnol., 18, 505-508, 2000.
71. Bhalgat M.K., Haugland R.P., Pollack J.S. and Swan S., “Green- and Red-Fluorescent Nanospheres for The Detection of Cell Surface Receptors by Flow Cytometry”, J. Immunol. Methods, 219, 57-68, 1998.
72. Liotta L.A., Ferrari M. and Petricoin E., “Clinical Proteomics: Written in Blood”, Nature, 425, 885-989, 2003.
73. Mehta A., Ross S., Lowenthal M.S. et al, “Biomarker Amplification by Serum Carrier Protein Binding”, Disease Markers, 19, 1-10, 2003.
74. Zhou M., Lucas D.A. and Chan K., “Investigation into The Human Serum Interactome”, Electrophoresis, 25, 1289-1298, 2004.
75. Petricoin E. F., Ornstein D.K. and Liotta L.A., “Clinical Proteomics: Applications for Prostate Cancer Biomarker Discovery and Detection”, Science Direct-Urologic Oncology: Seminars and Original Investigations, 22 (4), 322-328, 2004.
76. Xu H., Aylott J.W., Kopelman R., Miller T.J. and Philbert M.A., “A Real-Time Ratiometric Method for The Determination of Molecular Oxygen inside Living Cells Using Sol-Gel-Based Spherical Optical Nanosensors with Applications to Rat C6 Glioma”, Anal. Chem., 73, 4124-4133, 2001.
77. McDonagh C., MacCraith B.D. and McEvoy A.K., “Tailoring of Sol-Gel Films for Optical Sensing of Oxygen in Gas and Aqueous Phase”, Anal. Chem., 70, 45-50, 1998.
78. Koo Lee Y.E., Cao Y., Kopelman R., Koo S.M., Brasuel M. and Philbert M.A. “Real-Time Measurements of Dissolved Oxygen Inside Live Cells by Organically Modified Silicate Fluorescent Nanosensors”, Anal. Chem., 76, 2498-2505, 2004.
79. Sadeghi S.J., Tsotsou G.E., Fairhead M., Meharenna Y.T. and Gilardi G., “Rational Design of P450 Enzymes for Biotechnology” In: De Cuyper M., Bulte J. (Eds.), Focus on Biotechnology.Physics and Chemistry Basis of Biotechnology”, Kluwer Academic Publisher, Dordrecht, 71-104, 2001.
80. Joo H., Lin Z. and Arnold F.H., “Laboratory Evolution of Peroxide-Mediated Cytochrome P450 Hydroxylation”, Nature, 399, 670-673, 1999.
81. Dmochowski I.J., Crane B.R., Wilker J.J., Winkler J.R. and Gray H.B., “Optical Detection of Cytochrome P450 by Sensitizer-Linked Substrates”, Proc. Natl. Acad. Sci. USA, 96, 12987-12990, 1999.
82. Grigoryev D.N., Kato K., Njar V.C.O., Long B.J., Ling Y.Z., Wang X., Mohler J. and Brodie A.M.H., “Cytochrome P450c17-Expressing E. Coli as a First Step Screening System for 17 Alpha-Hydroxylase C17, 20-Lyase Inhibitors”, Anal. Biochem., 267, 319-330, 1999.
83. Schwaneberg U., Schmidt Dannert C., Schmitt J. and Schmid R.D., “A Continuous Spectrophotometric Assay for P450 Bm3, A Fatty Acid Hydroxylating Enzyme and Its Mutant F87A”, Anal. Biochem., 269, 359-366, 1999.
84. Tsotsou G.E., Cass A.E.G and Gilardi G., “High Throughput Assay for P450 BM3 For Screening Libraries of Substrates and Combinatorial Mutants”, Biosens. Bioelectron., 17, 119-130, 2002.
85. Wang X., Li Y., Wei J. and de Groot K., “Development of Biomimetic Nanohydroxyapatite/Poly (Hexamethylene Adipamide) Composites”, Biomaterials, 23, 4787-4791, 2002.
86. Du C., Cui F.Z., Feng Q.L., Zhu X.D. and de Groot K., “Tissue Response to Nano-Hydroxyapatite/Collagen Composite Implants in Marrow Cavity”, J. Biomed. Mater. Res., 42, 540-548, 1998.
Contact Details
|
Ernest Herbert
Department of Medicine
Center for Research
Hospital Laval
2725 Chemin Sainte Foy
Quebec City
Quebec G1V 4G5
Canada
E-mail: [email protected]
Tel. +1 (418) 656-8711, ext 2653
Fax. +1 (418) 656-4509
|
Rahul Shetty (Corresponding Author)
Department of Cardiac Surgery
Hospital Laval
2725 Chemin Sainte Foy
Quebec City
Quebec G1V 4G5
Canada
E-mail: [email protected]
Tel. +1 (418) 656-8711, ext 2653
Fax. +1 (418) 656-4509
|