May 7 2013
In a recent article in (Li X. et. al. Nature Methods advance online publication, 5 May 2013 doi:10.1038/nmeth.2472) researchers at the University of California, San Francisco (UCSF) report on the use of a breakthrough new camera and associated methods in the near-atomic-resolution reconstruction of the 700 kDa 20S proteasome protein complex using single particle cryo electron microscopy (cryo-EM).
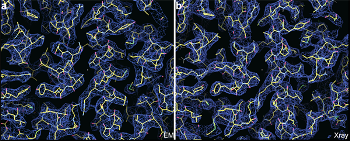 Indistinguishable results from EM (left) and X-ray (right) structures of the Thermoplasma acidophilum 20S proteasome.
Indistinguishable results from EM (left) and X-ray (right) structures of the Thermoplasma acidophilum 20S proteasome.
Prior studies using cryo-EM and film only yielded results down to 5.6 A resolution. At 3.3 A, the new reconstruction allows scientists to directly determine the location of each atom in the protein complex. (1 A or Angstrom is one-tenth of a nanometer or one-ten billionth of a meter.)
"Knowledge of the location of each atom in a protein complex is an incredibly important step in structural biology. It enables a deep understanding of fundamental biological processes, with the promise of developing novel drugs and therapeutics," says Dr. Christopher Booth, Life Science Product Manager at Gatan. "Until now only large, highly symmetric viruses could be studied at this level of detail using cryo-EM. Most atomic resolution protein structures come from X-ray crystallography, a technique that is limited to samples which can be crystalized, a challenging task that can sometimes take years of effort, and from nuclear magnetic resonance (NMR) spectroscopy which is limited to relatively small proteins or protein domains."
This breakthrough result on an older generation Transmission Electron Microscope (TEM) was enabled by Gatan's K2 Summit camera, the first of a new class of cameras, which, by counting the individual electrons in the TEM image, captures biological structures with unprecedented resolution and clarity.
Dr. Sander Gubbens, Gatan's Chief Operating Officer, states: "Electron microscopes have long been able to provide images at resolutions far greater than 3.3 A; however until now results in structural biology have been limited by the noise of the camera relative to the very small electron dose the fragile biological specimens can handle, as well as by specimen motion during imaging. The K2 Summit camera's electron counting allows near noise free imaging while its high 400 frames per second imaging speed enables the measurement and correction of specimen motion. The net result is a huge step forward in realizing the full potential of cryo-EM for the routine atomic resolution imaging of biological complexes in their native hydrated state."
"The K2 Summit has really changed the way we approach cryo-TEM experiments. Having achieved this near-atomic resolution benchmark opens a wide range of new problems for us to try to solve," says Drs. David Agard and Yifan Cheng, Professor and Associate Professor at UCSF. "We are very excited to be working at a time when technology is moving so quickly and gratified to have helped the development. For so many years we have been held back by the high intrinsic noise levels and slow readouts of our detectors. This seriously compromised the data. This camera is game changing for the field, and opens new ways to collect and process the data to ultimately achieve the highest resolution possible."
The K2 Summit is the result of a very successful collaboration between scientists at Gatan, UCSF, and the Lawrence Berkeley National Laboratory, with partially funding from the Howard Hughes Medical Institute and the National Science Foundation through an American Recovery and Reinvestment Act Major Instrumentation grant.
For more information about the K2 Summit camera, please visit: www.gatan.com/K2 or contact your local Gatan sales representative.
Source: http://www.gatan.com/