Mar 6 2013
The term nanomedicine encompasses a broad range of technologies and materials. Types of nanomaterials that have been investigated for use as drugs, drug carriers or other nanomedicinal agents include:
- Dendrimers
- Polymers
- Liposomes
- Micelles
- Nanocapsules
- Nanoparticles
- Nanoemulsions
Around 250 nanomedicine products are being tested or used in humans, according to a new report that analyzed evolving trends in this sector. According to experts, the long-term impact of nanomedicinal products on human health and the environment is still not certain.
During the last 10 years, there has been steep growth in development of devices that integrate nanomaterials or other nanotechnology. Enhancement of in vivo imaging and testing has been a highly popular area of research, followed by bone substitutes and coatings for implanted devices.
Active and passive cell targeting will continue to be an important focus in nanomedicine. Targeted nano-enhanced solutions have been shown to often enhance existing treatments, and some nanomedicinal techniques are being developed which work as diagnosis and treatment stages simultaneously.
The unknown factor as far as nanotechnology is concerned is whether the increased production, exposure and handling of products and nanomaterials will result in serious impact on the environment and humans. It is possible that toxicity will be the restricting factor for the public acceptance and commercial success of nanotechnology-based products.
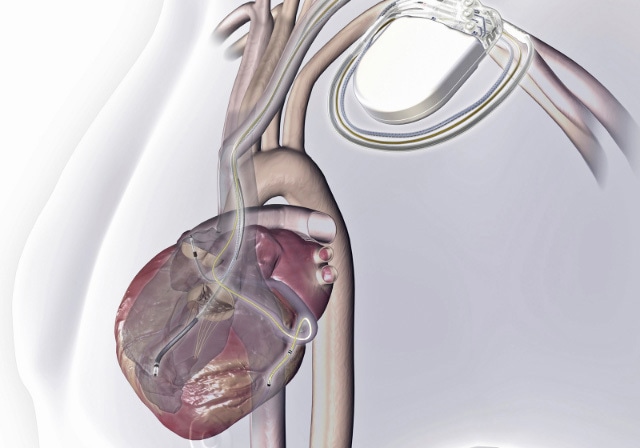
Advances in modern medicine are increasingly relying on electronic devices implanted inside the patient's body. Nanotechnology allows us to create materials and coatings to construct these devices that are fully biocompatible. Image credit: NASA
Implantable Biosensors
Micro-electromechanical systems (MEMS) and silicon chips that are capable of implantation within the human body may permit interfacing semiconductor devices with living tissues.
This will pave the way for implantable biosensors that can evaluate disease indicators or symptoms and regulate drug release to help in disease treatment.
For example, an implanted glucose sensor can be coupled with an insulin release system and help sufferers of diabetes control their sugar levels without the need for insulin injection or pin-prick tests.
While biocompatibility and long-term stability are being addressed, a number of prototypes have begun to emerge for the management of patients having acute diabetes or to treat epilepsy and other debilitating neurological disorders, and to monitor patients suffering from heart disease.
Integration with Monitoring Systems
Virtual Medical World published an article in November 2005 that stated that a research project financed by the Academy of Finland was underway to develop of minute subcutaneous sensors that can be used for active monitoring of the heart or prosthetic joint function even over long time periods.
For instance, a subcutaneous EKG monitor can detect cardiac arrhythmia, and this data can be wirelessly transmitted to the PC or mobile phone of the physician.
Chronic Disease Monitoring
Guidant is a specialist in treating vascular and cardiac disease and has invested in CardioMEMS based on an article published in Virtual Medical Worlds in November, 2005. CardioMEMS develops novel devices based on MEMS technology to help physicians monitor remotely the progress of chronic diseases like heart failure.
The University of Texas received a grant in 2006 to fund the research and development of an implantable intravascular biosensor that will monitor disease and health progression.
The nano pressure sensor can monitor pressure within the cardiovascular system while the data is transmitted to a wristwatch-like data collection device. The data is transmitted by this external device to a central remote monitoring station where it can be seen by health care providers in real time.
Implantable Cardioverter-Defibrillators
The implantable cardioverter-defibrillator (ICD) has transformed treatment of patients at risk for sudden cardiac death because of ventricular tachyarrhythmias.
The Medtronic CareLink Monitor is a small, convenient device that allows patients to gather information by holding an antenna over the implanted cardiac device. The data is automatically downloaded by the monitor and sent through an internet connection directly to the secure Medtronic CareLink Network. The patient’s data is accessed by clinicians by logging onto a website from any internet-connected computer in their home or office or through the laptop while travelling.
The ICD systems also include portable computer systems that program the implantable cardioverter defibrillators or pacemakers. This interactive system has an LCD touch screen with a user-friendly interface that helps clinicians retrieve and study patient information during routine follow-up visits and easily makes programming changes to the implanted devices.
This video demonstrates how an Implantable Cardioverter Defibrillator or ICD is used to treat dangerously fast or irregular heart beats. Run time: 0:58s.
Implantable Drug Delivery Systems
More and more advances in modern medicine are relying on electronic devices implanted inside the patient's body, to minimize the need for regular examinations, surgery, or in-patient time. Nanotechnology allows us to create materials and coatings to construct these devices that are fully biocompatible, so that they integrate seamlessly with the body's systems.
Implantable drug delivery systems can deliver small amounts of drugs on a regular basis, so that the patient does not need to be injected. Implantable drug delivery systems give a more consistent drug level in the blood compared to injections, which often makes the treatment more effective and reduces side effects.
By using active monitoring capabilities built into the device, the dosage can be adjusted to suit changes in physical activity, temperature changes and other variables.
In treatments such as chemotherapy, which are usually aimed at a specific area of the body, the device can be implanted near the target area, keeping drug concentration much lower in the rest of the body.
Smart implantable insulin pumps are designed so as to offer relief for people with Type I diabetes. These are implantable, active drug delivery devices that build on and go beyond the capabilities offered by passive glucose biosensors.
Regulatory Challenges
Nanomaterials and nanotechnology offer significant promise in the medical device community, as well as many other industry sectors. They also pose a number of regulatory challenges, which as time goes by will become more pressing than the technical challenges. Some of the difficulaties in regulating nanomedical devices are as follows:
- It is important to determine the intended use of the product, but it can be difficult to define uses among several stakeholders.
- The indicated patient population must be understood, and there should be clarity about the claimed benefits of a product.
- Throughout the submission process of products for market approval, it is important to communicate with the FDA or other relevant authority. Manufacturing processes are highly critical for a successful submission. Marketing, sales, labeling and international issues, training and education are all part of this effort.
Conclusions
Nanomedicine will transform healthcare in the coming years, changing the day-to-day business practices of health care organizations and improving how patient care is provided.
Health care organizations must monitor innovations continually, perform clinical trials and developments related to this area and also other evolving health IT solutions.
There is a lot of research going on in this area; however not many products have reached the commercialization stage. There is still a long way to go before all these promising devices become a part of our daily lives.
Sources