Recently, AZoNano spoke with researchers from ETH Zurich about their work that has created a gold nanoparticle coating for antifogging applications on transparent surfaces.
Please can you introduce yourselves and your professional background?
Iwan Haechler: My name is Iwan, a fourth-year Ph.D. student, and I study under Prof. Dimos Poulikakos. I've been doing my Ph.D. on the interaction between light and matter and how we can use this for interesting phase-change phenomena with water.
Nicole Ferru: I'm Nicole. I'm a mechanical engineering Masters's student. I did my bachelor's and my Master's in mechanical engineering at ETH, and I entered the project two years ago. I have been working on it as a research assistant for the past two years.
What are some of the everyday issues that inspired you to focus on antifogging technology?
Iwan: Our lab has actually been doing a lot of research on anti-fogging coatings for about a decade now and how we can alter the surface wettability. Most of the anti-fogging coatings or sprays currently render a surface hydrophilic. The alternative option is basically that you render a surface hydrophobic so water can’t really condense on it.
But the issue related to these two approaches is that in one case, if a surface likes to attract water and be hydrophilic, it also attracts everything else: dust, dirt, etc. On the other hand, with the hydrophobic surfaces, it's very challenging to scale these up. As a result, we started to look at alternative ideas, such as how we can employ the energy of light to prevent this effect.
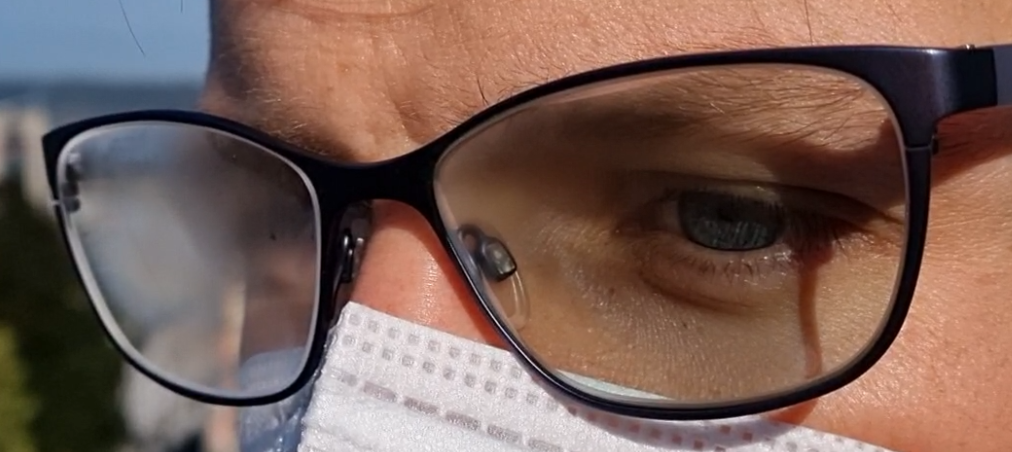
Image Credit: ETH Zurich
It’s extremely powerful because heat is, thermodynamically speaking, highly effective at preventing condensation. We know this from the back of our cars where you have copper wires that heat up the rear windshield.
Nicole: What makes this project so special is that we are really looking at transparent surfaces, and this changes which approach you can apply or how effective that approach can be. And that's really something that for me was very interesting throughout this project - the idea of wanting to keep the transparency in order for the technology to stay applicable to real-life problems.
You have managed to develop an ultrathin gold-based transparent coating for glass that produces heat from sunlight. Please could you describe the structure/make-up of this coating?
Nicole: The active parts of this material are gold nanoparticles. We all know that gold is not transparent, but if you have only singular nanoparticles then you can get a transparent surface.
Iwan: It is well known that gold nanoparticles absorb light quite efficiently. A lot of approaches actually use gold nanoparticles already, and when you shine a light on them they have some resonance effects and absorb the light. It is possible to take different particle sizes and merge them together, such that every particle absorbs at a specific wavelength. In the end, you have quite interesting broadband absorption behavior.
But the challenge was actually to develop a coating that selectively absorbs sunlight. How can it retain its transparency, yet absorb the infrared part of the light, which in fact makes up 50% of solar energy?

Image Credit: ETH Zurich
When gold is deposited, individual particles first settle on the surface. The more gold you deposit, the more continuous your film becomes. If you find that sweet spot where these individual nano islands start to communicate electrically or touch each other, then electrons can flow through the material, called the percolation threshold. Here we see some very, very anomalous absorption behavior because a metal film at this very specific thickness has a broadband absorption behavior above a certain wavelength, which means they broadly absorb light and also it's the maximum absorption you can achieve in a material.
The second material we employed was basically titanium dioxide, and we made a little sandwich where we encapsulated our gold layer between the buns on top and on the bottom. The reason for that was, first of all, it protects our gold layer, and second of all, it enhances the absorption even further.
At what levels of light will this technology function?
Nicole: Of course, you do need sunlight. So in total darkness, the system wouldn't work. But we have proven both in the lab as well as in real-world conditions that with very, very low solar intensities, we are already able to show this effect.
It was a very important question in the review process: how does this material perform under different solar intensities? So as well as the solar intensity that people use in research, we even showed it down to 60% of this intensity. Next, we measured it down to 20%. So in the real world, we showed that if it is windy or cloudy it is still robust.
eth_1
Iwan: I remember exactly the first day we tested it outside. You measure all that stuff in the lab on the setup and technically the physics holds and it should work, but I was pretty amazed when I saw it actually perform. Hence, it was quite astonishing to me, especially when we tested it in the mountains. After the experiments, in the evening when I checked the level of solar radiation during the day, I was baffled by how low the radiation actually was.
How much heat energy can be produced?
Nicole: We quantify the energy that is produced by how many degrees Celsius the surface increases by. Of course, this is always dependent on what substrate you put this coating on. But what we measured in our lab was that with one solar unit, we had eight degrees of increase in the temperature of the substrate, which is pretty significant if you think about how thin the active surface is.
Can you apply this coating to other types of materials?
Nicole: So in principle, this works on any surface. Of course for the way that the coating is conceived, it should be applied to transparent surfaces only. We actually tested a lot of different transparent surfaces including plastic foils, to which we were able to apply our coating and measure the desired optical properties.
Iwan: We basically tested it on five different surfaces. Glass acrylic glass (PMMA), polystyrene, polycarbonate, and on a flexible polyester foil (an overhead transparency). We also tested the robustness against bending; for this, we bent it back and forth 300 times and checked the optical properties. The properties before, during (in a bent state) and after bending were exactly the same and remained completely unaffected because the coating was so thin.
Are there any applications beyond glass anti-fogging that this technology could be adapted to in the future?
Iwan: Of course, sunglasses are a potential market that we foresee. We are also looking at car headlights, windshields, and any other application where you can localize heat on the nanoscale (nano-scale heaters).

Image Credit: ETH Zurich
If we compare our material to alternatives at the same level of transparency, and if we look at how much sunlight this material absorbs per thickness, our coating outperforms other photothermal antifogging coatings by up to 60 times. For this reason, we do see potential applications for nanoscopically localized heating applications.
How do these coatings compare to conventional anti-fogging sprays when it comes to performance?
Nicole: In the lab, we exposed the surface to different humid conditions to prove its functionality, and we wanted to use a camera to take images of the effect. Of course, our camera began to fog because it was exposed to the same conditions. First, we tried to use conventional anti-fogging sprays to keep the camera from fogging and be able to take pictures. But, those sprays did not hold the anti-fogging properties that our surface actually could, and in the end we had to completely change the setup. So already during this research, we saw the performance of our coating is much better than that of conventional sprays. It's the thermal approach that is much more efficient than these sprays that work with the surface properties.
How cheap and scalable can this technology be?
Iwan: In the end, the entire design strategy was to develop a coating which can actually be used outside of the laboratory conditions. We knew that this coating could only be used if the methods to fabricate it were scalable and we produced it with the probably most heavily used machines in the cleanroom. For this reason, the coating was basically developed on weekends, because the two tools were so heavily used during weekdays. We therefore think that it can definitely be fabricated over large areas.
Could gold be substituted for a cheaper metal?
Iwan: We filed a patent application which covers a range of materials. and which is not limited to gold. However, in the end, the gold deposition is literally five nanometers. That's about a thousand to 10,000 times thinner than a human hair, which means the amount of gold is very minute. This results in material costs for gold of about 0.20 USD per pair of glasses.
We are currently looking at potential markets and a business case.
20211130_140150
Where do you see this research in the next couple of years?
Iwan: Hopefully we see it on many glasses by then! Right now, I have to focus on finishing my Ph.D., but after, we will see how we can bring the technology to the market..
What was the most exciting part of this project for you?
Iwan: The biggest lesson to me was to experience how effective heat is to prevent fogging. I believe it was crucial to convince our peers by showing them that we can nano-engineer and we can take it out into the real world and simply show you it works. We tried to answer many of the questions by simply demonstrating that it does work and we can indeed just fabricate it. Hence, it was probably quite convincing to actually demonstrate the real-world product. This kind of nicely wraps up the whole experience we had with this project.
About Iwan Haechler
 Iwan is a Ph.D. student in the Laboratory of Thermodynamics in Emerging Technologies at ETH Zurich under Professor Dimos Poulikakos. His research focuses on nanoengineering the interaction of light with matter and how it can be used for sustainable water-related technologies.
Iwan is a Ph.D. student in the Laboratory of Thermodynamics in Emerging Technologies at ETH Zurich under Professor Dimos Poulikakos. His research focuses on nanoengineering the interaction of light with matter and how it can be used for sustainable water-related technologies.
About Nicole Ferru
 Nicole is a Mechanical Engineering Masters's student at ETH. She has been working in the Laboratory of Thermodynamics in Emerging Technologies as a research assistant.
Nicole is a Mechanical Engineering Masters's student at ETH. She has been working in the Laboratory of Thermodynamics in Emerging Technologies as a research assistant.
Disclaimer: The views expressed here are those of the interviewee and do not necessarily represent the views of AZoM.com Limited (T/A) AZoNetwork, the owner and operator of this website. This disclaimer forms part of the Terms and Conditions of use of this website.