Humans have a long history of combining ointments, balms, and salves for therapeutic, protective, and cosmetic purposes. Cosmetics have long been a vital aspect of human civilization, representing care, self-confidence, and self-expression.1
They now integrate dermatological research and aesthetic design, using nutrient-rich ingredients and cutting-edge technologies to enhance well-being and beauty.
Every cream, serum, therapeutic drug, or medical device is the result of extensive study aimed at improving tolerance and performance while avoiding negative side effects.
Atomic force microscopy (AFM) drives innovation and product development in the cosmetic and healthcare industries, but it also serves as a powerful tool for quality control in routine manufacturing processes, ensuring consistent quality and reproducibility, which are critical factors in increasing customer confidence and brand loyalty.
This article explains how atomic force microscopes for biology (BioAFMs) can be utilized for a wide range of cosmetic and healthcare applications, providing detailed examples.
Introduction
AFM is a high-resolution microscopy technique that provides three-dimensional images of surface topography and thorough mechanical characterization of objects ranging in size from sub-nanometers to micrometers.
AFM can also measure friction forces, adhesion, and viscoelastic characteristics, identify specific binding sites, and investigate dynamic processes.
AFM's ability to operate in near-physiological conditions, such as body temperature and controlled humidity, makes it a powerful tool for investigating the effects of an active substance on living cells and tissues after application.2,3
The integration of AI and deep learning algorithms drives higher levels of automation, faster, more exact measurements, and more thorough analyses.4
Companies that produce healthcare items, medical equipment, and cosmetics can leverage these capabilities to build safer, more effective products and to substantiate efficacy claims with quantitative data.5
By incorporating AFM into development workflows, industries can better meet growing regulatory and consumer demands for evidence-based quality, ensuring that goods are scientifically demonstrated to be effective rather than merely claimed to be so.
Applications: A Brief Overview
The following section provides examples of how AFM is used to analyze skin, hair, teeth, contact lenses, and oral care items. They demonstrate AFM's capabilities and the advantages of its application in the healthcare and cosmetics industries.
Contact Lenses
Contact lenses used for vision correction must meet multiple requirements simultaneously, including optical performance, biocompatibility, and long-term comfort. These properties are determined by a precise balance of material composition, surface topography, and mechanical qualities.
Contact lenses' nanoscale characteristics significantly impact tear film stability, wettability, and protein adsorption, all of which are important factors in reducing irritation and infection risk.
Because no single material can meet all these specifications, modern contact lenses often combine optimized bulk materials with sophisticated surface coatings. AFM significantly contributes to the creation of new lens materials in industrial research and quality assurance during the manufacturing process.6
High-resolution AFM imaging reveals surface roughness and heterogeneities in polymer structures, and nanomechanical mapping measures elasticity across the lens.7
It is crucial for silicone hydrogel lenses to strike a balance between oxygen permeability and surface smoothness. Figure 1 shows representative AFM measurements on daily single-wear contact lenses, delivering:
- Large-area mapping of the lens curvature
- Stiffness measurements
- Nanoscale surface topography
- Viscoelastic properties
Advanced AFM modes, such as SmartMapping, perform large-area curvature mapping with nanoscale precision, enabling full-lens characterization on the millimeter scale.8
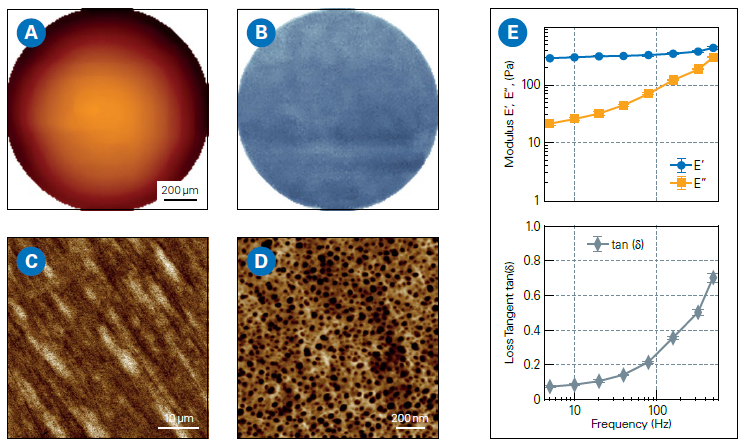
Figure 1. AFM measurements on daily contact lenses (single-use): A: Large-scale topography map (Z-range: 90 μm). B: Corresponding Young’s modulus map (Z-range: 326-506 kPa; average modulus: 405 kPa), acquired using SmartMapping mode. C, D: High-resolution AFM topography images of the lens surface. E: Frequency-dependent analysis of viscoelastic properties showing Storage Modulus (E’), Loss Modulus (E”), and Loss Tangent (ratio of E”/E’). Image Credit: Bruker Nano Surfaces and Metrology
AFM measurements reveal crucial links between structure, composition, and performance, enabling precise optimization of lens materials and design, and delivering quantitative gains in comfort, safety, and regulatory compliance.9,10
Next-generation lens research prototypes are being developed that incorporate sensors for measuring glucose or intraocular pressure, as well as augmented reality functionality.
AFM can play an important role in this advancement by enabling accurate material engineering and characterization of material properties while preserving lens performance and wearer comfort.
Dermatology and Skincare
Skin health is a key sign of general well-being. Early diagnosis of structural or compositional changes in the skin can greatly improve diagnostic and treatment results.
AFM enables nanoscale characterisation of skin by integrating high-resolution topographic imaging with nanomechanical mapping, yielding quantitative nanohistology.11
This method gives objective indicators that go beyond the limitations of traditional histology, which normally uses staining and light microscopy to examine tissue morphology and cellular architecture.
AFM can detect small variations in tissue stiffness, elasticity, and surface shape that frequently occur at the cellular or extracellular matrix level, before symptoms appear at the macroscopic level.12
These nanoscale measurements are particularly useful for distinguishing healthy from diseased skin, especially in disorders such as eczema, psoriasis, skin cancer, hypermobile Ehlers-Danlos syndrome (hEDS), or scleroderma.13,14
In a diagnostic study, cutaneous tissue sections from individuals with hEDS and scleroderma were examined using AFM.
Two sections of the same sample were extracted; one was stained with Picosirius Red to visualize structural changes in the dermal collagen using polarized light microscopy, while the other was left unchanged and studied with AFM.
This method allows for a direct correlation between classical histology and AFM-based nanomechanical investigation. The AFM nano-topography investigation revealed significant structural variations (Figure 2A).
Healthy skin had well-organized collagen fibrils with distinct D-banding, whereas diseased areas had disorganized arrangements of bent, randomly orientated, and twisted fibrils.
AFM-derived biomechanical measurements also revealed considerably lower modulus values in hEDS-scleroderma tissue, indicating decreased collagen stiffness and increased structural disorder. In comparison, healthy skin exhibited a uniform, unimodal stiffness distribution with modest fibril abnormalities (Figure 2B).
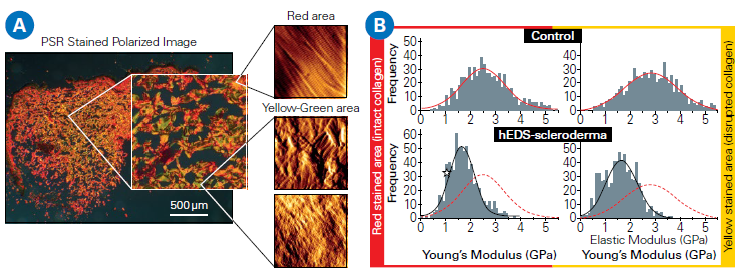
Figure 2. A: Correlated AFM and polarized light microscopy images (Picosirius Red-stained) reveal intact collagen regions (red) and compromised areas (yellow/green) in skin samples from control and hEDS-scleroderma patients. B: Distributions of collagen fibril Young’s modulus for healthy and disrupted regions. In the control specimen, both region types exhibit a unimodal stiffness distribution. In hEDSscleroderma samples, the modulus values are significantly lower, consistent with reduced collagen stiffness. The red dotted line represents the collagen stiffness distribution of the control group. Data courtesy of Laurent Bozec, University of Toronto, Canada. Image Credit: Bruker Nano Surfaces and Metrology
In addition to pathology, AFM may detect hydration-induced swelling and lipid structure, providing insights into the skin barrier function and irritation susceptibility.15,16
AFM's ability to function in physiologically realistic settings makes it ideal for ex vivo biopsies and other intriguing future uses. AFM, which provides structural and biomechanical insights, can improve diagnosis, track illness progression, and promote breakthrough skincare and cosmetics advances.
AFM provides quantitative measures that allow for exact assessment of collagen structure, hydration, and treatment results, hence promoting individualized skincare, targeted therapies, and scientifically supported product claims.
Human Hair
Human hair is a complex, keratin-based, anisotropic biomaterial that varies significantly in thickness, curl, stiffness, and wetness. Its structural integrity governs its visual appearance, mechanical robustness, and tactile feel.
Conventional evaluation methods, such as expert panel tests and macroscopic trials, capture user experience but lack the ability to quantify nanoscale variables, such as adhesion, friction, and cuticle wear, which heavily influence the feel and durability of hair.17
The hair cuticle, the outermost protective covering of hair composed of overlapping keratin scales oriented in the direction of hair growth, dominates the tribological aspects of hair, namely adhesion, friction, and wear.
AFM is especially appropriate for the quantitative analysis of these features and the evaluation of the performance of hair-care products.18-20
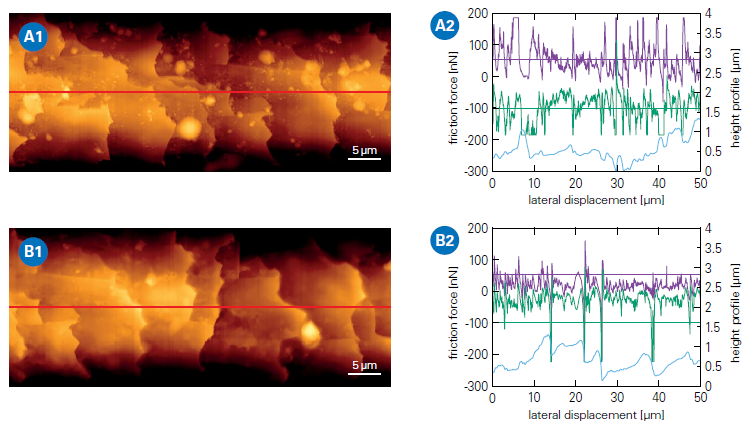
Figure 3. AFM analysis of a hair fiber before and after application of a conditioning hair care product. AFM topography images (left) of a hair fiber and corresponding friction measurements (right) along the hair fiber (indicated by the red line). The diagrams show the leveled height profile in blue and the corresponding friction forces: purple (trace) and green (retrace). A: Untreated hair fiber. Topography image (A1) and corresponding friction measurement (A2). B: Hair fiber after treatment with conditioner. Topography image (B1) and corresponding friction measurement (B2). Image Credit: Bruker Nano Surfaces and Metrology
Figure 3 shows tribological AFM measurements using a 1 µm spherical probe on a hair fiber before and after applying a conditioning agent.
Although the related topography photos show no evident visual changes between the hair surfaces other than small impurities, friction forces are dramatically reduced after conditioner treatment.
Transferring the friction force to the coefficient of friction using Amonton's law yields a scalable, quantitative metric that may be related to cosmetic product performance.18,19 The sliding speed can be changed to explore dynamic tribology.20
AFM's automated procedures enable high-throughput investigation of various hair fibers and treatments. The experiment's design can be altered to imitate realistic settings by mounting a section of a hair fiber on the cantilever, allowing the study of hair-to-hair contact.21
To simulate fingertip touch, customized collagen-coated colloidal probes can be used to take tribological measurements under conditions similar to those found in ordinary use. BioAFM systems and their related environmental add-ons are intended to work in liquids at controlled temperatures and humidity levels.
This enables the examination of shampoos, conditioners, and other cosmetic products under shower-like and everyday circumstances, revealing insights into their performance and impacts on hair vitality, nutrition, and cuticle protection.22,23
Oral Care and Dental Protection
Oral health, particularly teeth, shows the close relationship between aesthetic appearance and general health. White teeth are widely regarded as a sign of good health and social prestige, which explains why whitening solutions are popular as a cosmetic therapy.
Maintaining dental health, specifically preventing dental decay (caries) and treating dentin hypersensitivity, is a top healthcare goal.
Modern oral hygiene products, such as toothpaste, are multipurpose, serving protective and whitening purposes. Effective whitening needs bleaching solutions, which might impair the integrity of the teeth and enhance hypersensitivity by exposing dentin tubules.24,25
Oral care solutions must consequently strike a balance between cleaning and whitening while also protecting enamel. AFM contributes to the creation of such goods by characterizing tooth surfaces at the micro- and nanoscale levels, as well as under conditions that closely resemble reality.
AFM assesses surface roughness, mineral crystallite orientation, and micro-defects, which are crucial for understanding how abrasives, bleaching chemicals, and biofilms alter tooth structure.26-28
This is critical for assessing structural integrity and predicting the long-term efficacy of dental care therapies. Figure 4 shows the effect of a bleaching chemical on the surface structure of dentine as detected by AFM. The evident alterations in surface structure upon application illustrate the bleaching agent's aggressive properties.

Figure 4. AFM analysis of animal tooth cross-section. The sample was prepared by sanding, polishing, and etching with 0.5M EDTA for 2 minutes. A: AFM topography image of the dentin surface showing dentin tubules (Z-range: 618 nm). B: Zoomed-in AFM topography image of dentin surface after brushing with toothpaste for 30 min and subsequent cleaning with water (Z-range: 150 nm). C: AFM topography image of dentin surface after application of bleaching gel, revealing structural changes (Z-range: 150 nm). Image Credit: Bruker Nano Surfaces and Metrology
Desensitizing toothpastes reduce dentine hypersensitivity by, for example, using chemicals that plug dentin tubule apertures. Figure 5 shows how AFM was used to assess the effectiveness of a desensitizing toothpaste.
By evaluating multiple tubule openings, AFM can determine the efficiency of toothpaste by calculating the proportion of sealed dentin tubules. Nanomechanical mapping evaluates local stiffness and hardness, allowing for immediate assessment of enamel loss or remineralization following treatment with fluoride or calcium-based formulations.
Contact resonance AFM, in particular, may provide high-resolution mechanical maps of highly rigid surfaces, such as enamel and dentin, with greater accuracy than standard analytical methods.29

Figure 5. AFM analysis of the cross section of an animal tooth. The sample was prepared by sanding, polishing, and etching with 0.5M EDTA for 2 minutes. AFM topography images of openings of dentin tubule before (A) and after (B) application of a desensitizing toothpaste, revealing that the tubule is sealed after treatment. (Z-range: 500 nm). Image Credit: Bruker Nano Surfaces and Metrology
Figure 6 shows a high-resolution mechanical map of the dentin-enamel junction created by contact resonance AFM on the cross-section of a human milk tooth. In addition to the topography information, the stiffness differential between enamel and dentin is plainly visible and consistent with published values.
These findings emphasize AFM's capacity to investigate chemical erosion and evaluate dental care treatments, providing a thorough assessment of dental integrity.
Tribological analysis under simulated brushing conditions could help optimize the development of dental hygiene products and formulations by determining the effects of abrasive particle size on performance, ensuring effective cleaning and minimal enamel erosion.
Similarly, advanced AFM modes can help with the design and evaluation of protective coatings that minimize bacterial adhesion and biofilm formation, which are crucial for long-term dental health and caries prevention.
These techniques underscore the potential of AFM to guide the development of next-generation oral care products that combine beauty and oral health benefits.

Figure 6. Contact resonance AFM imaging of the enamel-dentin junction of a human milk tooth cross-section. Young’s modulus map overlaid onto the 3D-rendered topography, showing a strong mechanical contrast (50 GPa for enamel vs. 18 GPa for dentin), consistent with literature studies (scan size: 50×50 μm²). The sample was prepared by sanding and polishing, then cleaned with distilled water. Image Credit: Bruker Nano Surfaces and Metrology
Key Benefits of AFM in the Cosmetic and Healthcare Industries
Versatility
AFM provides nanoscale insights into surface structure and dynamics across a wide range of materials, including contact lenses, skin, hair, teeth, and care items, enabling innovation in both the cosmetic and healthcare industries.
Product Performance
High-resolution imaging and nanomechanical mapping enable precise evaluation of material integrity, wear resistance, and treatment effects, resulting in optimal formulations and dependable quality control.
Realistic Testing
BioAFMs operate in physiologically relevant settings, enabling ex vivo studies of samples, materials, and healthcare products before, during, and after application, resulting in accurate, thorough performance assessment.
Advanced Tools
Techniques such as contact resonance AFM and SmartMapping produce quantitative, multiparametric data that far outperforms standard analytical methods. The increasing application of machine learning and AI-driven automation in AFM is speeding up procedures and enhancing reproducibility.
Future Potential
AFM is projected to drive next-generation solutions, such as smart contact lenses, individualized skincare, and enhanced oral care coatings, while also supporting manufacturers in meeting regulatory and customer needs for evidence-based results.
Acknowledgments
This article was produced using materials originally written by Maximilian Seuss, Jörg Barner, Randhir Kumar, and Thomas Henze.
References and Further Reading
- McMullen, R.L. and Dell’Acqua, G. (2023). History of Natural Ingredients in Cosmetics. Cosmetics, (online) 10(3), p.71. DOI: 10.3390/cosmetics10030071. https://www.mdpi.com/2079-9284/10/3/71.
- Alsteens, D. (2024). Probing living cell dynamics and molecular interactions using atomic force microscopy. Biophysical Reviews, 16(6), pp.663–677. DOI: 10.1007/s12551-024-01258-7. https://link.springer.com/article/10.1007/s12551-024-01258-7.
- Magazzù, A. and Marcuello, C. (2023). Investigation of Soft Matter Nanomechanics by Atomic Force Microscopy and Optical Tweezers: A Comprehensive Review. Nanomaterials, 13(6), p.963. DOI: 10.3390/nano13060963. https://www.mdpi.com/2079-4991/13/6/963.
- Di Guardo, A., et al. (2025). Artificial Intelligence in Cosmetic Formulation: Predictive Modeling for Safety, Tolerability, and Regulatory Perspectives. Cosmetics, (online) 12(4), p.157. DOI: 10.3390/cosmetics12040157. https://www.mdpi.com/2079-9284/12/4/157.
- Bhushan, B. and LaTorre, C. (2008). Structural, Nanomechanical, and Nanotribological Characterization of Human Hair Using Atomic Force Microscopy and Nanoindentation. Springer Handbook of Nanotechnology, pp.1325–1485. DOI: 10.1007/978-3-540-77608-6_24. https://link.springer.com/chapter/10.1007/978-3-540-77608-6_24.
- Last, J. A., et al. (2010). The Applications of Atomic Force Microscopy to Vision Science. Investigative Ophthalmology & Visual Science, 51(12), 6083–6094. DOI: 10.1167/iovs.10-5470. https://iovs.arvojournals.org/article.aspx?articleid=2126233.
- Sharma, V., et al. (2022). Surface characterization of an ultra-soft contact lens material using an atomic force microscopy nanoindentation method. Scientific Reports, 12(1). DOI: 10.1038/s41598-022-24701-9. https://www.nature.com/articles/s41598-022-24701-9.
- Bruker Nano Surfaces SmartMapping. In: AFM Modes - SmartMapping. Available at: https://www.bruker.com/en/products-and-solutions/microscopes/bioafm/bioafm-modes/smartmapping.html
- Mitrovic, A., et al. (2018). Characterization of surface roughness of new nanophotonic soft contact lenses using lacunarity and AFM method. Hemijska industrija, 72(3), pp.157–166. DOI: 10.2298/hemind170924004m. https://doiserbia.nb.rs/Article.aspx?ID=0367-598X1800004M.
- Kim, S.H., et al. (2020). ToF-SIMS and AFM Characterization of Brown Cosmetic Contact Lenses: From Structural Analysis to the Identification of Pigments. Journal of Analytical Methods in Chemistry, 2020, pp.1–11. DOI: 10.1155/2020/6134627. https://onlinelibrary.wiley.com/doi/10.1155/2020/6134627.
- Huang, S., et al. (2023). Quantitative nanohistology of aging dermal collagen. Frontiers in Aging, (online) 4. DOI: 10.3389/fragi.2023.1178566. https://www.frontiersin.org/journals/aging/articles/10.3389/fragi.2023.1178566/full.
- Torebjörk, H.E., et al. (2023). Age-related changes in dermal collagen physical properties in human skin. PLOS ONE, 18(12), pp.e0292791–e0292791. DOI: 10.1371/journal.pone.0292791. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0292791.
- Kotova, S.L., et al. (2015). Collagen Structure Deterioration in the Skin of Patients with Pelvic Organ Prolapse Determined by Atomic Force Microscopy. Microscopy and Microanalysis, 21(2), pp.324–333. DOI: 10.1017/s1431927615000148. https://academic.oup.com/mam/article-abstract/21/2/324/6897158?redirectedFrom=fulltext.
- Mehrnoosh Neshatian, et al. (2024). Investigation of dermal collagen nanostructures in Ehlers-Danlos Syndrome (EDS) patients. PLoS ONE, 19(8), pp.e0307442–e0307442. DOI: 10.1371/journal.pone.0307442. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0307442.
- Qin, X., Dong, M. and Li, Q. (2024). Insight into the hydration friction of lipid bilayers. Nanoscale, 16(5), pp.2402–2408. DOI: 10.1039/d3nr05517e. https://pubs.rsc.org/en/content/articlelanding/2024/nr/d3nr05517e.
- Marcott, C., et al. (2013). Nanoscale infrared (IR) spectroscopy and imaging of structural lipids in human stratum corneum using an atomic force microscope to directly detect absorbed light from a tunable IR laser source. Experimental Dermatology, 22(6), pp.419–421. DOI: 10.1111/exd.12144. https://onlinelibrary.wiley.com/doi/10.1111/exd.12144.
- Lodge, R.A. and Bhushan, B. (2006). Surface characterization of human hair using tapping mode atomic force microscopy and measurement of conditioner thickness distribution. Journal of Vacuum Science & Technology A: Vacuum, Surfaces, and Films, 24(4), pp.1258–1269. DOI: 10.1116/1.2180275. https://pubs.aip.org/avs/jva/article-abstract/24/4/1258/101928/Surface-characterization-of-human-hair-using?redirectedFrom=fulltext.
- Smith, J.R., et al. (2013). AFM friction and adhesion mapping of the substructures of human hair cuticles. Applied Surface Science, 285, pp.638–644. DOI: 10.1016/j.apsusc.2013.08.104. https://www.sciencedirect.com/science/article/abs/pii/S016943321301595X?via%3Dihub.
- Bowen, J., et al. (2016). Friction and wear of human hair fibres. Surface Topography: Metrology and Properties, 4(2), p.024008. DOI: 10.1088/2051-672x/4/2/024008. https://iopscience.iop.org/article/10.1088/2051-672X/4/2/024008.
- Weiand, E., et al. (2023). Nanoscale friction of biomimetic hair surfaces. Nanoscale, 15(15), pp.7086–7104. DOI: 10.1039/d2nr05545g. https://pubs.rsc.org/en/content/articlelanding/2023/nr/d2nr05545g.
- Luengo, G.S., Mizuno, H. and Rutland, M.W. (2017). Hair-Hair Contact Dynamics and Interactions Studied with Atomic Force Microscopy. Agache’s Measuring the Skin, (online) pp.835–845. DOI: 10.1007/978-3-319-32383-1_118. https://www.diva-portal.org/smash/record.jsf?pid=diva2%3A1303844&dswid=-9211.
- Fellows, A.P., Casford, M.T.L. and Davies, P.B. (2020). Nanoscale Molecular Characterization of Hair Cuticle Cells Using Integrated Atomic Force Microscopy–Infrared Laser Spectroscopy. Applied Spectroscopy, 74(12), pp.1540–1550. DOI: 10.1177/0003702820933942. https://journals.sagepub.com/doi/10.1177/0003702820933942.
- Fellows, A.P., Casford, M.T.L. and Davies, P.B. (2021). Chemically characterizing the cortical cell nano-structure of human hair using atomic force microscopy integrated with infrared spectroscopy (AFM-IR). International Journal of Cosmetic Science. DOI: 10.1111/ics.12753. https://onlinelibrary.wiley.com/doi/10.1111/ics.12753.
- Dahl, J.E. and Pallesen, U. (2003). Tooth Bleaching - a Critical Review of the Biological Aspects. Critical Reviews in Oral Biology & Medicine, 14(4), pp.292–304. DOI: 10.1177/154411130301400406. https://journals.sagepub.com/doi/10.1177/154411130301400406.
- de Freitas, M.R., et al. (2021). Effectiveness and Adverse Effects of Over-the-Counter Whitening Products on Dental Tissues. Frontiers in Dental Medicine, 2. DOI: 10.3389/fdmed.2021.687507. https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2021.687507/full.
- Pattem, J., et al. (2022). The dynamic interplay of dietary acid pH and concentration during early-stage human enamel and dentine erosion. Frontiers in dental medicine, 3. DOI: 10.3389/fdmed.2022.1040565. https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2022.1040565/full.
- Murchie, B.D., et al. (2025). The early wear susceptibility of dentine following exposure to citric acid: In vitro study. Archives of Oral Biology, 178, p.106363. DOI: 10.1016/j.archoralbio.2025.106363. https://www.sciencedirect.com/science/article/pii/S0003996925001918?via%3Dihub.
- Zhai, Y., et al. (2024). Effects of Acid Etching on the Microstructure and Stiffness of Human Teeth. Journal of Engineering and Science in Medical Diagnostics and Therapy, 8(3). DOI: 10.1115/1.4066624. https://asmedigitalcollection.asme.org/medicaldiagnostics/article-abstract/8/3/031005/1206872/Effects-of-Acid-Etching-on-the-Microstructure-and?redirectedFrom=fulltext.
- Bruker. (2026). Contact Resonance Module. (online) Available at: https://www.bruker.com/en/products-and-solutions/microscopes/bioafm/bioafm-accessories/contact-resonance-module.html.

This information has been sourced, reviewed and adapted from materials provided by Bruker Nano Surfaces and Metrology.
For more information on this source, please visit Bruker Nano Surfaces and Metrology.