Nicotinamide-loaded peptoid nanotubes restored cellular energy status, reduced inflammatory injury signals, and improved histopathological outcomes in preclinical models of acute neonatal brain injury.
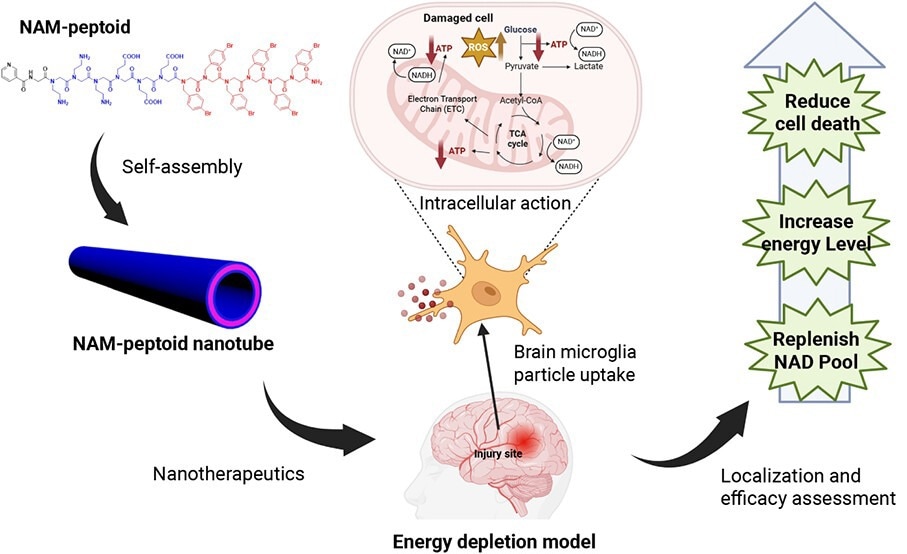
Nicotinamide-Loaded Peptoid Nanotubes for Energy Regeneration in Acute Brain Injury
A new study in the journal ACS Nano reports a nanotechnology-driven strategy for restoring cellular energy following acute brain injury. The researchers demonstrate that nicotinamide-loaded peptoid nanotubes enable microglia-associated intracellular delivery of an NAD+ precursor, overcoming key limitations in conventional therapies. The findings establish a new framework for using sequence-defined nanomaterials to modulate cellular metabolism, with important implications for treating energy-depleted neurological disorders.
Targeted Nanotherapy to Address Energy Failure in Brain Injury
Acute brain injuries disrupt cellular metabolism, sharply reducing Adenosine Triphosphate (ATP) levels while increasing oxidative stress and inflammation. A key driver of this dysfunction is the depletion of nicotinamide adenine dinucleotide (NAD+), an essential coenzyme in cellular respiration and DNA repair. Although replenishing NAD+ or its precursors shows therapeutic potential, translating this approach to clinical use remains challenging due to poor cellular uptake, rapid degradation, and limited cell-specific targeting.
The researchers address the barriers by developing a nanomaterial-based delivery system using peptoid nanotubes. These sequence-defined polymers self-assemble into stable, biocompatible tubular structures. By conjugating nicotinamide (NAM) to the nanotubes, the engineered NAM-PNTs improve intracellular delivery of an NAD+ precursor in brain-relevant injury models.
The study shows that NAM-PNTs increase intracellular ATP levels, enhance cell viability, and reduce inflammatory responses after injury. A single dose produces measurable benefits across multiple preclinical models, including microglial cells, organotypic brain slices, and neonatal rat models. This work addresses a key gap in the field by introducing a biocompatible, microglia-targeting platform to restore cellular energy metabolism in the injured brain.
Design and Experimental Framework of Peptoid Nanotube Delivery
The researchers used a bottom-up nanotechnology approach to design and synthesize peptoid nanotubes. Amphiphilic, sequence-defined peptoids containing hydrophobic and polar domains self-assembled into well-defined tubular structures. The team covalently attached nicotinamide molecules to the peptoid backbone to ensure stable drug incorporation and controlled delivery.
The team prepared the nanotubes through controlled evaporation-induced crystallization. The prepared nanotubes were characterized by atomic force microscopy (AFM), transmission electron microscopy (TEM), and X-ray diffraction (XRD), confirming highly ordered, crystalline nanotube architectures. Further, the researchers tuned nanotube length via sonication, thereby optimizing cellular uptake and tissue penetration.
Researchers used multiple biological models to assess biocompatibility and efficacy. In vitro studies employed BV-2 microglial cells exposed to oxygen-glucose deprivation (OGD) to simulate ischemic injury. Ex vivo experiments used organotypic whole hemisphere brain slices to preserve tissue architecture. In vivo validation was performed using a neonatal rat hypoxia-ischemia model.
Additional experiments investigated cellular uptake mechanisms and showed that microglia internalize the nanotubes primarily through fluid-phase phagocytosis and caveolae-mediated endocytosis. This comprehensive methodology enabled evaluation of both nanoscale design and biological performance.
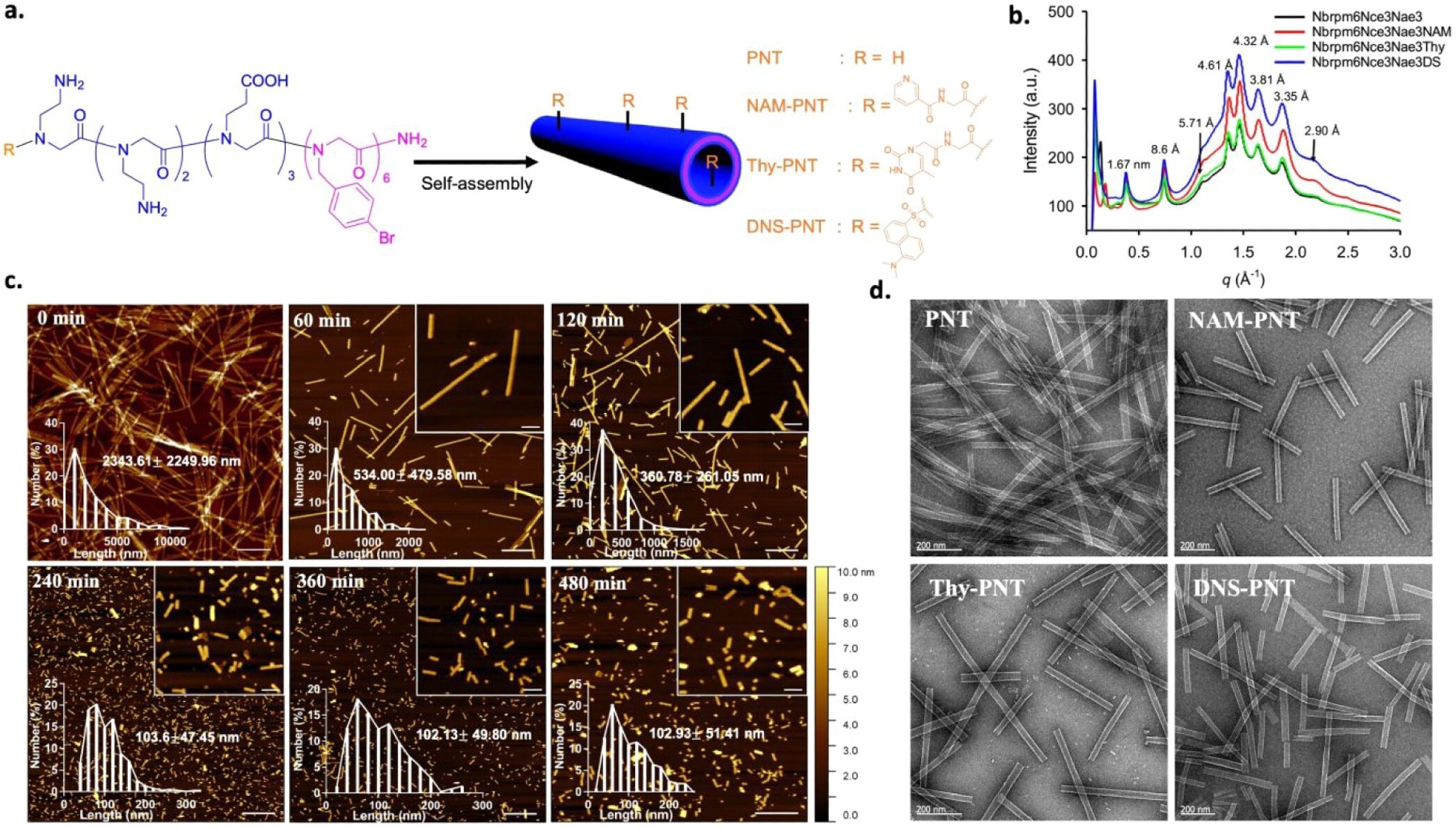
Characterizations of the PNT, NAM-PNT, Thy-PNT, and DNS-PNT. a, Schematic illustration of the peptoid structure and self-assembly. R indicates four different end groups in the peptoid. b, XRD data of PNT, NAM-PNT, Thy-PNT, and DNS-PNT showing similar crystalline structures for all peptoids. c, AFM images showing NAM-PNTs with various lengths after 0, 60, 120, 240, 360, and 480 min of sonication. Scale bars: 1 μm and 200 nm (insets). d, TEM images of PNT, NAM-PNT, Thy-PNT, and DNS-PNT, respectively. Scale bar: 200 nm.
Enhanced Cellular Energy, Reduced Inflammation, and Improved Histopathological Outcomes
The results show that NAM-PNTs effectively improve cellular energy metabolism after injury. In oxygen-glucose-deprived microglial cells, NAM-PNTs increased metabolic activity and ATP levels, whereas free nicotinamide showed limited benefit. This suggests that nanotube-mediated delivery improves the intracellular availability of therapeutic molecules.
Nanotube length strongly influenced therapeutic performance. Intermediate-length nanotubes, around 120 nm in the in vivo model and NAM-PNT120 in brain slices, achieved the best balance between cellular uptake and tissue penetration. Longer nanotubes showed limited diffusion, while shorter nanotubes produced weaker effects, possibly due to rapid clearance.
In organotypic brain slices, NAM-PNT treatment reduced cell death and restored intracellular NAD(H) levels toward healthy-slice levels. The treatment also increased cell proliferation, particularly in microglia and oligodendrocytes. In addition, NAM-PNTs modulated inflammatory responses by lowering proinflammatory cytokines, including IL-1β, IL-6, and TNF-α, while increasing the anti-inflammatory cytokine IL-10.
In vivo studies further showed that in neonatal rats subjected to hypoxia-ischemia, a single systemic dose of NAM-PNTs reduced brain tissue loss and improved histopathological outcomes. Treated animals showed lower lesion scores in key brain regions, including the cortex and thalamus.
Gene expression analysis showed reduced markers of cell death, inflammatory activation, and nitric oxide-related injury responses, along with enhanced anti-inflammatory signaling. Imaging studies confirmed that nanotubes preferentially localized within microglia in the injured brain, indicating cell-associated therapeutic delivery. Overall, these results establish NAM-PNTs as an effective preclinical platform for improving cellular energy status and reducing injury-induced damage at both cellular and tissue levels.
Conclusion and Future Implications for Nanomedicine
This study introduces a nanotechnology platform that addresses a central challenge in brain injury treatment: restoring cellular energy metabolism. By integrating precise molecular design with microglia-associated delivery, NAM-PNTs overcome key limitations of traditional NAD+ supplementation. The nanotubes enable efficient intracellular delivery, protect therapeutic cargo from degradation, and support preferential localization in microglia.
The findings show that nanotechnology-driven NAD+ restoration can increase intracellular ATP levels, reduce inflammation, and support tissue preservation after injury. Consistent results across in vitro, ex vivo, and in vivo models highlight the preclinical translational potential of NAM-PNTs. Beyond acute brain injury, this NAM-PNT platform may potentially extend to neurodegenerative diseases such as Alzheimer’s and Parkinson’s disease, where energy depletion and mitochondrial dysfunction play central roles. However, this possibility remains speculative and was not tested in neurodegenerative disease models. The modular design of peptoid nanotubes also supports the incorporation of different therapeutic agents, broadening their potential across biomedical applications.
Future studies should evaluate long-term safety, pharmacokinetics, optimized dosing strategies, repeat dosing, and functional outcomes. Overall, this work represents a significant advance in nanomedicine and demonstrates how engineered nanomaterials can address complex biological challenges and improve outcomes in preclinical models of neurological disorders.
Download your PDF copy by clicking here.
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.