Feb 2 2022
Micron-sized particles are available on the commercial market in a broad range of materials such as polymers, glass, ceramics and (magnetic) metals, and they can be used in the fields of biology, medicine, material science and engineering.
These particles are employed as filler materials, as contrast agents in e-displays, for targeted delivery or timed release of drugs, altering the viscosity of slurries in the casting-industry, or to fabricate dentures, amongst other things.
The physical properties of the particles, particularly their size distribution and density, are crucial for the control of certain functionalities, including magnetic properties, flow dynamics or packing density. Thorough characterization of these beads’ composition, surface area and size or mass is a key factor relative to their function.
This article presents novel mass/size measurements of micrometer-sized particles using the synergistic combination of two innovative technologies: PicoBalance and FluidFM®.
Hollow cantilevers that are commercially available (FluidFM probes) were specifically used to reliably aspirate, hold and subsequently release single particles. PicoBalance - a technology that measures the mass of beads by measuring the frequency shift of a resonating cantilever - was used to quantify the mass of the particles.
PicoBalance
PicoBalance is a cantilever-based method that can be applied to establish the mass of a sample that is affixed to the free end of the cantilever (see Figure 1). Generally, the particle is attached to a tipless cantilever, and the shift in resonance frequency is determined before and after attachment to measure the mass.1-3
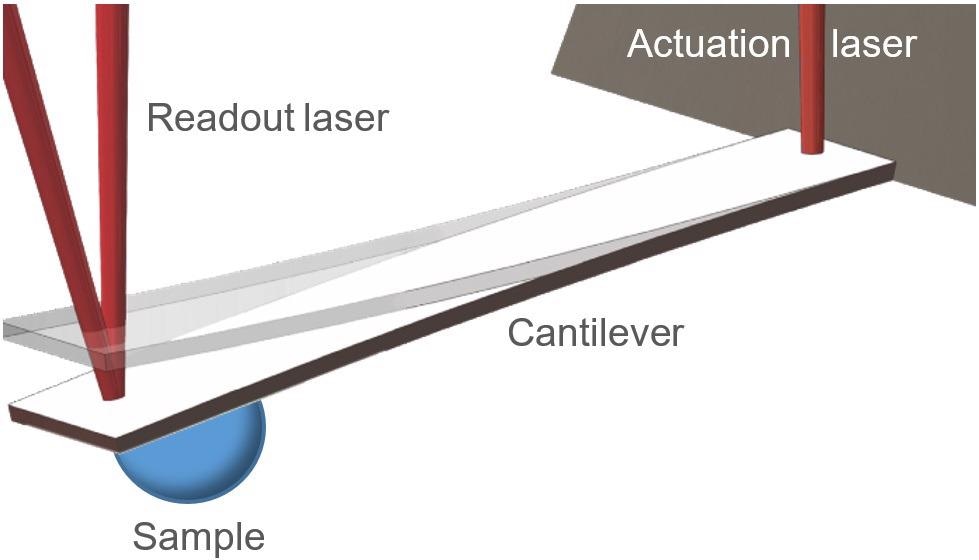
Figure 1. PicoBalance schematic. The mass of a sample is measured by determining the mass-induced resonance frequency shift of an actuated cantilever. Actuation and readout are done via red lasers. Image Credit: Nanosurf AG
Three parameters are required for mass calculation:
- The shift in resonance frequency (see Fig. 2)
- The cantilever’s spring constant
- The position of the attached mass on the cantilever
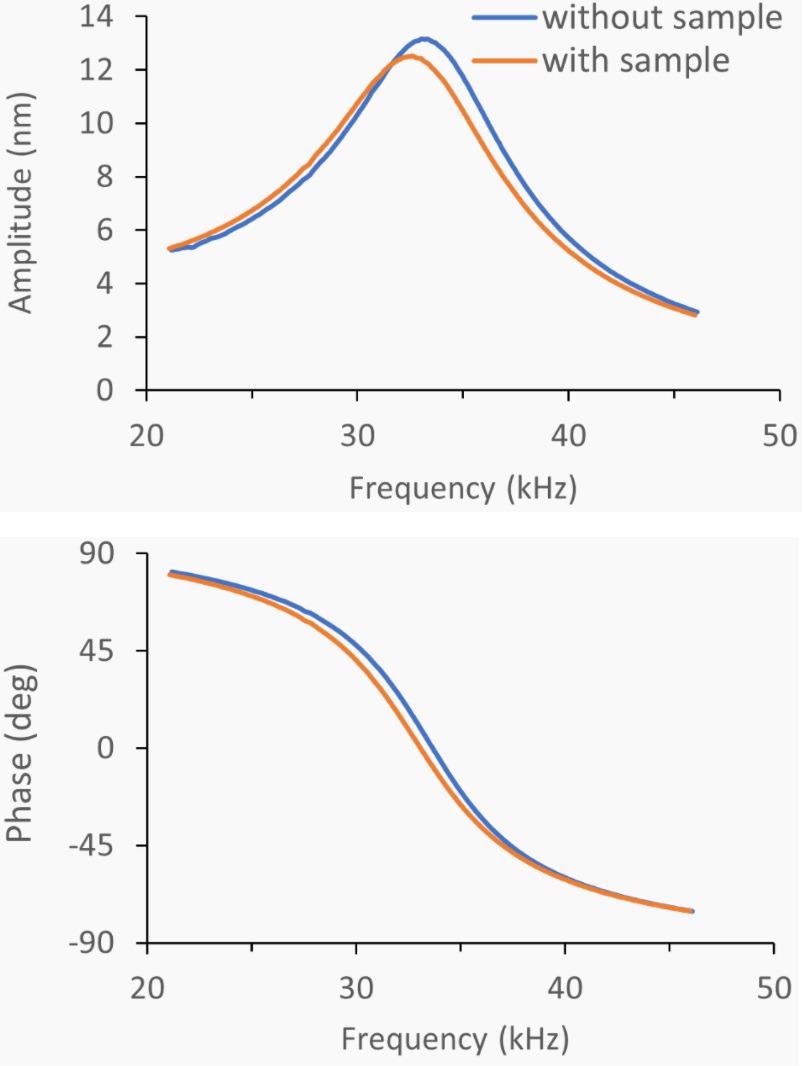
Figure 2. Attachment of a bead to the cantilever leads to a resonance frequency shift that allows the mass determination of the bead. Image Credit: Nanosurf AG
For precise mass measurements, a clean and stable actuation of the cantilever is necessary. Clean and stable actuation using acoustic (piezo) actuation is especially challenging in liquids, where a phenomenon called the “forest of peaks”4,5 obstructs the cantilever resonance.
To prevent this artifact, PicoBalance utilizes the cantilever’s photothermal actuation (CleanDrive) - the resulting textbook-like amplitude and phase response - to facilitate the accurate tracking of the resonance frequency.
Measuring the spring constant can be conducted in a number of ways prior to the experiment if it is not already pre-calibrated.
Typically, the best results are acquired by applying the Sader method.6 This method utilizes the planar dimensions of the cantilever as well as its damping and resonance frequency that are measured in air, either by a thermal spectrum or by actuating the cantilever.
As the position changes, the mass sensitivity varies. Thus, the attached mass’s position needs to be accounted for. It can be established optically, however, when FluidFM probes are in use, it is well-defined.
FluidFM®
The cornerstone of the FluidFM technology is a hollow cantilever with an aperture at the free end (Figure 3). The hollow cantilever works like a micropipette using a reservoir controlled by a precision pump.7
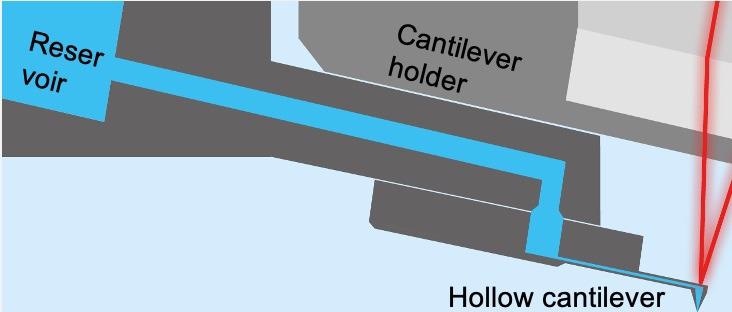
Figure 3. Schematic of FluidFM. A hollow FluidFM cantilever is connected to a pressurized fluid reservoir. Image Credit: Nanosurf AG
Particle attachment is fast and reversible. Practically, the cantilever is brought within proximity (~10 µm) to the particle that is to be attached, and a minor underpressure (- 50 mbar) is applied as the particle is fixed onto the cantilever using suction.
Once measurements of the particle’s mass are complete, the particle can be released via overpressure.
The position of the attached particle corresponds to the position of the aperture, and it is known and repeatable. In the example presented (Figure 4), the aperture is 8 µm in diameter and 4.4 µm distance from the free end. Without FluidFM, glue or chemical functionalization is required to attach particles.

Figure 4. Left, 10 µm bead attached to FluidFM probe. Right, 15 µm bead attached to FluidFM probe. Scale bar is 15 µm. Image Credit: Nanosurf AG
These techniques can cause uncertainties in the particle mass, and the attachment process is irreversible. A new cantilever is required for each measurement. However, a chemical functionalization could be better suited for some biological samples.
In air or vacuum environments, manipulating small beads can be a challenge, and they may also aggregate due to powerful electrostatic interactions. Working in liquids at the appropriate pH (i.e., in their storage buffer solution) can inhibit aggregation, and FluidFM technology readily allows the selection of individual particles.
The smallest shift detectable in the resonance frequency of the FluidFM probe was 15 Hz, which is equal to a mass resolution of 0.12 ng.
Mass Distribution of Silicon Dioxide and Polystyrene Beads
As an experimental model, silicon dioxide (SiO2) beads with nominal diameters of 10, 11, 13, 14 and 15 µm, and polystyrene beads (PS) with diameters of 10 and 15 µm were measured8 utilizing PicoBalance with FluidFM probes. Sequential measurements of 25 different specimens of each type of bead were carried out.
The results of all 175 measurements are displayed in Figure 5. The expected mass (x-axis) and uncertainty was inferred using the manufacturer’s specifications for the density and nominal diameter.9,10
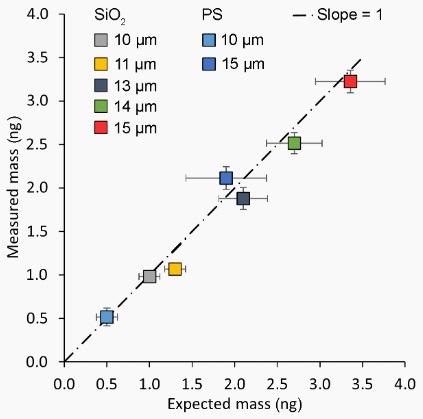
Figure 5. Results of 175 mass measurements on different beads. The uncertainty as represented by the error bars is lower than expected from the manufacturer specifications. A straight line of slope 1 indicates the expected behaviour for an accurate, linear measurement. n=25 per datapoint. Image Credit: Nanosurf AG
The masses observed using the PicoBalance-FluidFM combination (y-axis) displayed an uncertainty of only 0.1 ng. To further improve this value, smaller cantilevers can be applied.
The close correlation between the expected and measured mass signifies that the frequency shift gives the total mass of the beads directly and does not take into account the buoyant mass (mass of the bead minus the mass of the displaced liquid).
The mass observed matches the expected mass predicated on the diameter of the bead and the manufacturer’s given density.
Indeed, the mass uncertainty declared in the manufacturer specifications is greater than the PicoBalance measurements, indicating that the cantilever-based mass sensing technique is much more accurate.
Summary
Bringing together FluidFM and PicoBalance technologies facilitates precise mass measurements of colloidal beads in solution and offers a user-friendly platform for aspirating and dispensing particles.
PicoBalance, supported by photothermal excitation (CleanDrive), is a precise technique for measuring total bead mass. Through the application of the FluidFM technology, the measurement process is both quick and highly repeatable.
The PicoBalance is the only solution commercially available to measure the total mass of samples in the range of picograms to nanograms.
Alternative, non-commercial mass measurement techniques that are cantilever-based have one of two drawbacks: measuring only the buoyant mass (total mass minus the mass of the sample displaced liquid) or inferior mass and time resolution. They are also deficient in simultaneously acquiring supportive optical information.
References
- Martinez-Martin, D., Fläschner, G., Gaub, B. et al. Nature 550, 500-505 (2017).
- Fläschner, G., Roman, C. I., Strohmeyer, N. et. al. Nat. Commun 12, 2922 (2021).
- Cleveland, J. P., Manne, S., Bocek, D. & Hansma, P. K. Rev Sci Instrum 64, 403-405 (1993).
- Nanosurf. CleanDrive: Photothermal Excitation of the Cantilever (2021).
- Carrasco, C., Ares, P., de Pablo, P. J. & Gomez-Herrero, J. Rev Sci Instrum 79, 126106 (2008).
- Sader, J. E., Larson, I., Mulvaney, P. & White, L. R. Rev Sci Instrum 66, 3789-3798 (1995).
- Meister, A., Gabi, M., Behr, P. et al. Nano Lett 9, 2501-2507 (2009).
- Gunstheimer, H. Improved mass calibration of an intertial pico-balance using micro-particles, TU Ilmenau (2021).
- MicroParticles GmbH (2021).
- Polysciences Inc. (2021).

This information has been sourced, reviewed and adapted from materials provided by Nanosurf AG.
For more information on this source, please visit Nanosurf AG.