|
Researchers have used MS Modeling's CASTEP to study the role of oxygen on the structural and optoelectronic properties of silicon nanodots.
Potential Application of Silicon Nanodots
Such an understanding will enable these properties to be manipulated, leading to commercially viable nanoscale solid-state lighting devices - a major commercial application of nanotechnology.
What Are Nanodots?
Nanodots, also known as quantum dots, consist of 100s-1000s of atoms of inorganic semiconductor nanoparticles and are approximately one billionth of a meter in size. Developed in the mid-1980s for optoelectronic applications, they have interesting structural, electronic, and optical properties - they strongly absorb light in the near UV range and re-emit visible light that has its color determined by both the nanodot size and surface chemistry. And as the size of nanodots can be controlled during synthesis with nanoscale precision, so the optical properties can be manipulated. In addition, nanodots have a longer life than organic fluorophores, and have a broad excitation spectrum. These factors combined make the use of quantum dots as light-emitting phosphors a strong candidate for a major application of nanotechnology in the future.
What is so Special About Silicon Nanodots
Silicon nanodots have, in particular, have emerged over the last 10 years as a hot area of research due to the fact that a reduction in size of this semiconducting material to the nanometer scale dramatically alters their physical properties. In addition, the 1990 discovery that porous silicon exhibits photoluminescence properties, has led to a flurry of research activity, with commercially viable solid-state lighting devices made from Si nanostructures seemingly within reach.
Reactions of Porous Silicon with the Atmosphere
As porous silicon reacts with the atmosphere, leading to major structural (and thus optical properties change), the theoretical role of different passivation species (to passivate is to coat (a semiconductor, for example) with an oxide layer to protect against contamination and increase electrical stability) must be fully understood.
The Effect of Passivating Oxygen Properties of Nanodots
With the knowledge that silicon nanocrystals dispersed in SiO2 show an optical gain researchers at the University of Modena and Regio Emilia, Italy, used MS Modeling's CASTEP to study the role of passivating oxygen on the structural and optoelectronic properties of silicon nanodots.
Marcello Luppi and Stefano Ossicini used density functional theory (DFT) to investigate:
- The changes in the optoelectronic properties when O is absorbed onto hydrogenated Si nanocrystal
- The different role played by single and double Si–O bonds
- How a SiO2 matrix influences the physical properties of a Si nanocrystal
Using CASTEP, the scientists showed:
- In hydrogen covered Si nanocrystals single-bonded oxygen atoms lead to small variations in the electronic properties yet large changes in structure
- However, double-bonded oxygen atoms lead to small geometry variations yet a large energy gap reduction, explaining the huge photoluminescence red shift observed in high porosity silicon after oxygen exposure
The studies on Si nanocrystals embedded in a SiO2 matrix revealed:
- The presence of the nanocrystals only slightly deforms the SiO2 cage determining the formation of an interface region of stressed SiO2 between the nanocrystals and the matrix
- New electronic states originate in the SiO2 band gap
- Both nanocrystal Si atoms and interface O atoms affect optical properties
|
|
|
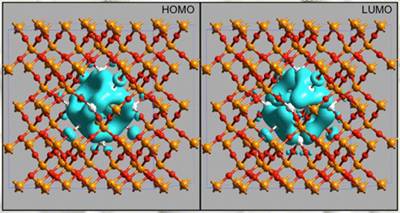
Figure 1. The HOMO and LUMO isosurfaces at fixed value show that the distribution is totally confined in the Si NC region with some weight on the interface O atoms. These dot-related states originate strong absorption features in the optical region. These features are entirely new and can be at the origin of the photoluminescence observed in the red optical region for Si nanocrystals immersed in a SiO2 cage.
|
Computer Modelling Results Agree with Experimental Results
These findings help to explain experimental optical property observations, and should lead to the fine tuning of nanodot optical properties - paving the way to commercially viable solid-state lighting devices based on nanodot technology.
Conclusion
Dr Marcello Luppi comments, "The use of CASTEP enabled us to perform a first principle study on semiconductor nanodots embedded in an insulator host matrix in which for the first time the whole system has been geometrically optimized. Thanks to the performance of the code we were able to handle hundreds of atoms and to study the electronic and optical properties of the system in a very accurate and efficient way. The graphic user interface was the perfect tool for drawing our models and for analyzing the results".
|