SARS-CoV-2 (COVID-19) is an infectious disease caused by a novel strain of coronavirus. A study published in the journal Current Research in Pharmacology and Drug Discovery has collated the results from research investigating the effects of graphene oxide's antiviral activities.

Image Credit: PopTika/Shutterstock.com
Figure 1 illustrates commonly observed symptoms of the virus, including loss of smell and taste, shortness of breath and cough. The virus penetrates human cells via interaction with the angiotensin-2 converting enzyme (ACE2) in the lungs.
Graphene oxide (GO), the oxidized form of graphene, is a nanomaterial that can disperse in many solvents. Graphene and GO have a crucial role in the electronic and medical fields as they have been shown to demonstrate antibacterial and antiviral properties.
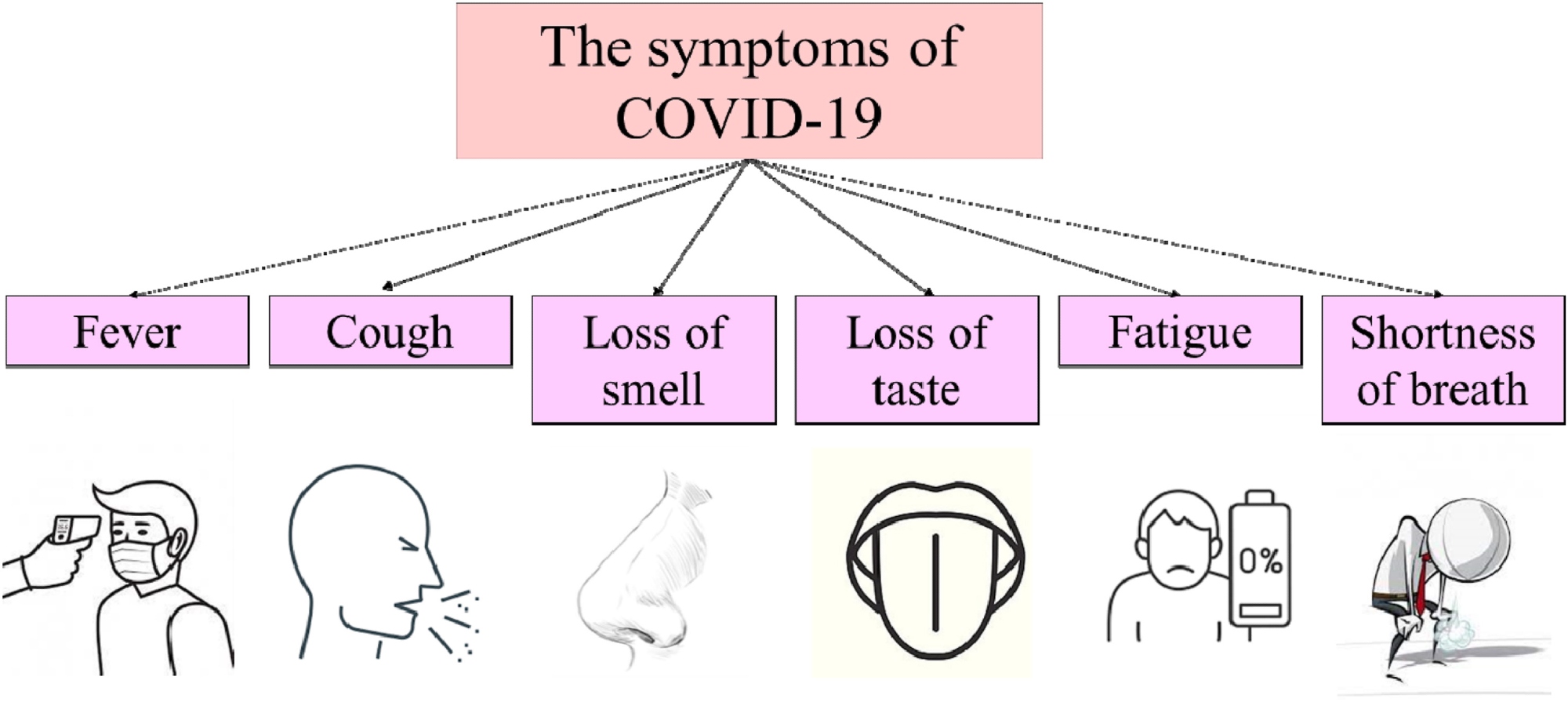
Figure 1. Symptoms of COVID-19.© Rhazouani, et al., 2021
Furthermore, GO's higher negative charge results in it possessing a high affinity for positively charged viruses; conjugation of GO with antibodies has also been shown to enable quick detection of targeted viruses.
Functionalized graphene shows good viral capture capacity and can be used as a disinfectant. Graphene sensor arrays can be implemented on standard utility textiles and for drug efficacy screening.
This study discusses the mechanism of entry of viruses into host cells and investigates GO's antiviral activity on certain viruses and through photocatalysis. Researchers also analyzed the antiviral effects of the GO-silver complex to fight viruses.
GO and graphene as biosensors for virus detection have also been discussed.
Research
Forging an effective fight against viruses requires in-depth knowledge of how they enter host cells and proliferate to form new viruses. The virus multiplication process involves various steps.
The S protein of the SARS-CoV-2 virus uses the cellular ACE2 receptor to enter the host cell. Figure 2 illustrates how the SARS-CoV-2 virus enters the human cell and proliferates.
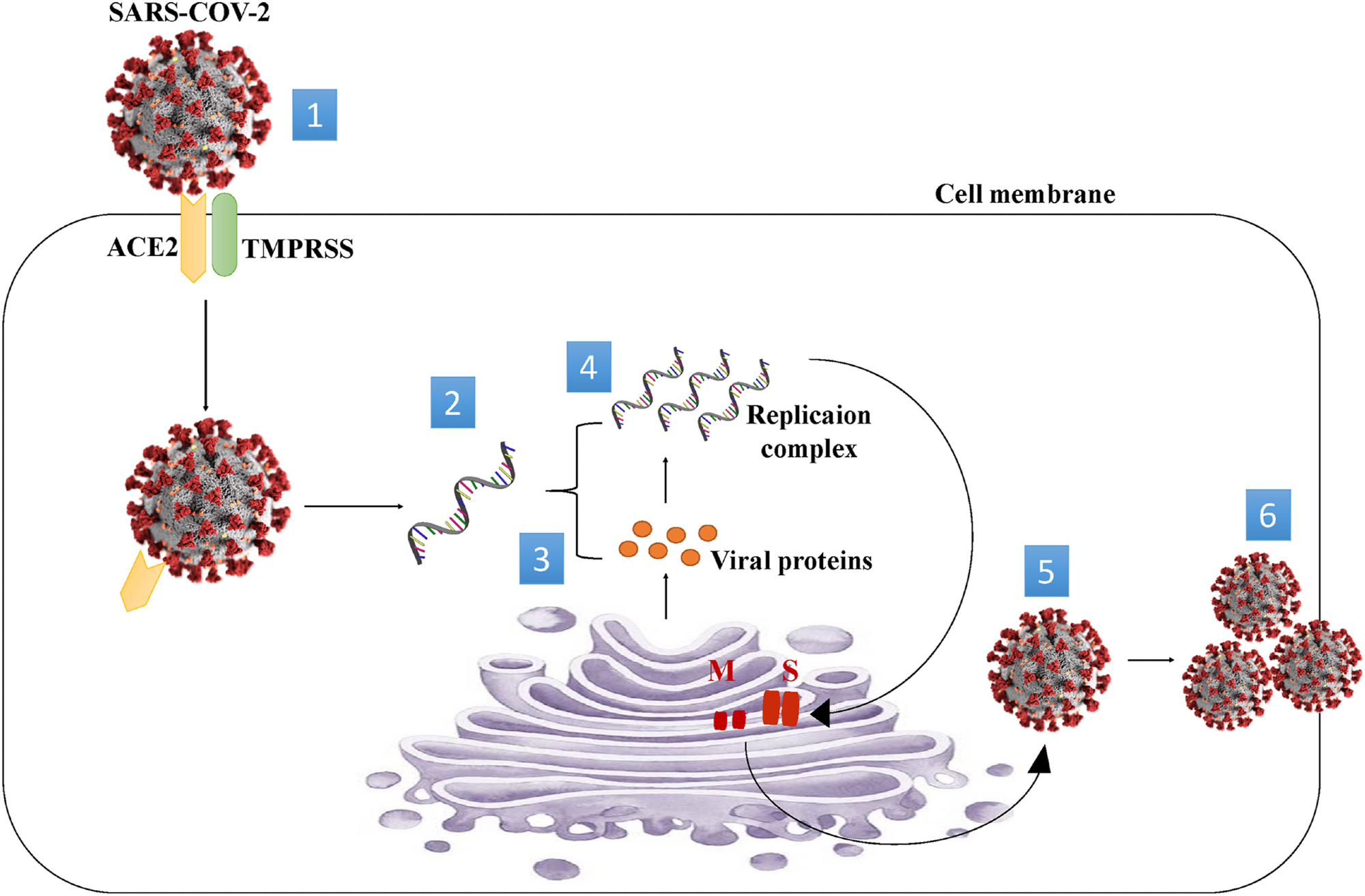
Figure 2. Mechanism of SARS-CoV-2 entry into the cell. (1) Spike protein on the virion binds to ACE2. TMPRSS2, an enzyme, helps the virion enter. (2) The virion releases its RNA. (3) Some RNA is translated into proteins by cell machinery. (4) Some of these proteins form a replication complex to make more RNA. (5) Proteins and RNA are assembled into a new virion in the Golgi and 6 Released.© Rhazouani, et al., 2021
GO exhibits a higher water dispersibility, hydrophilicity, and bonding capacity than graphene. Such physicochemical properties, extremely high mechanical strength, and high surface-to-volume ratio have led to the extensive use of GO as an antibacterial and anti-cancer agent.
Its sharp edges, two-dimensional structure, and negatively charged surface allow it to interact with viruses and break their plasma membrane or produce reactive oxygen species.
GO significantly inhibits infection of PRV, a porcine herpes virus (a DNA virus) that causes Aujesky's disease, and PEDV, a coronavirus (an RNA virus) that infects pigs and leads to dehydration and diarrhea. The strong antiviral activity of GO and rGO can be attributed to the unique monolayer structure and its negative charge.
In vivo and in vitro studies have shown that the hypericin-GO complex (GO/HY) exhibits antiviral activity against New Duck Virus Disease (NDRV), an acute infectious disease of poultry. The GO/HY complex exhibits dose-dependent inhibition of NDRV replication, which can be attributed to the inhibition of virus binding or virus inactivation (see Figure 3).
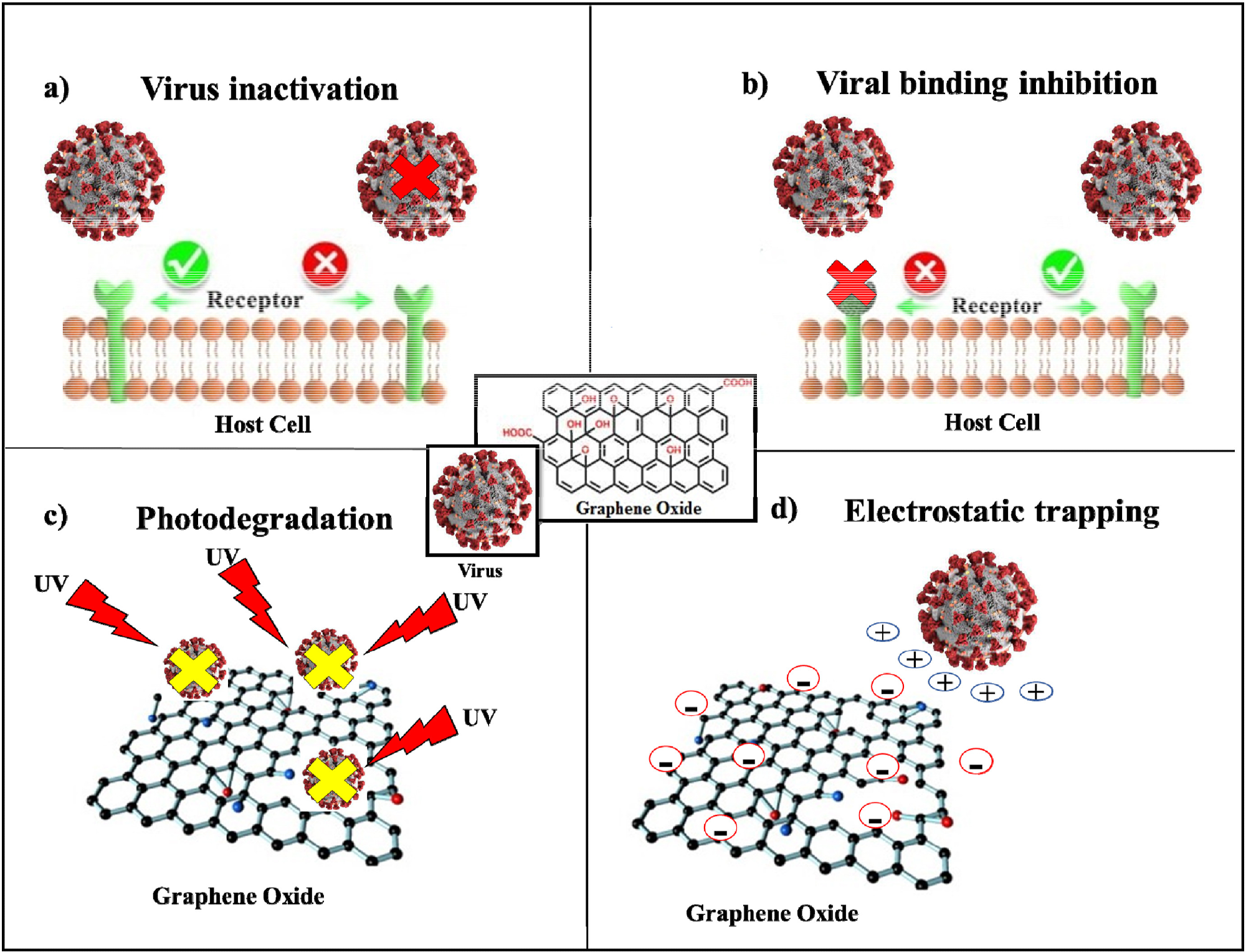
Figure 3. Description of the main mechanisms of GO's antiviral activity, (a) including virus inactivation, (b) viral binding inhibition, (c) photodegradation, and (d) electrostatic trapping. © Rhazouani, et al., 2021
Results
GO has been established as an excellent nanomaterial for high-throughput detection and disinfection of viruses, as well as great potential to inhibit environmental infections. Meanwhile, curcumin-functionalized GO acts effectively to inhibit infections caused by the respiratory syncytial virus (RSV).
Curcumin-functionalized GO was also found to be highly biocompatible with host cells, indicating a new pathway of antiviral therapy for RSV infection (see Table 1).
Table 1. The antiviral activity of GO. Source: Rhazouani, et al., 2021
| Virus |
Family |
Species |
Nucleic
acid |
Viral
envelope |
GO |
Ref |
| RSV |
Pneumoviridae |
Human
orthopneumovirus |
RNA |
enveloped |
GO |
Yang et al.
(2017) |
| VSV |
Rhabdoviridae |
Indiana
vesicolorum |
RNA |
enveloped |
GO |
Gholamiet
al. (2017) |
| H9N2 |
Orthomyxoviride |
Influenza A |
RNA |
enveloped |
GO |
Songet al.
(2015) |
| IBDV |
Birnaviridae |
Infectious Bursal
disease virus |
RNA |
Non-
enveloped |
GO-AgNPs |
Chen et al.
(2016) |
| NDRV |
Reoviridae |
– |
RNA |
Non-
enveloped |
GO/HY |
D. Xet al.
(2019) |
| FCoV |
Coronaviridae |
Alphacoronavirus 1 |
RNA |
enveloped |
GO-AgNPs |
Chen et al.
(2016) |
| PEDV |
Coronaviridae |
Porcine epidemic
diarrhea virus |
RNA |
enveloped |
GO |
Yeet al.
(2015) |
Nanocomposites with GO and partially reduced sulfonated GO suppressed infections by herpes simplex virus type 1 (HSV-1) by inhibiting the virus from binding to host cells. GO also exhibits superior photocatalytic activity that can be leveraged to suppress virus activity. The virus must stay close to the GO surface under UV irradiation (see Figure 3).
When compared to graphene, GO exhibits a higher bactericidal activity as its concentration considerably affects bacterial viability. At lower concentrations, GO forms floating scaffolds that promote bacterial growth. However, at higher concentrations, it forms scaffolds that can inhibit bacterial growth.
The combination of AgNPs with antiviral activity and GO with antimicrobial potential has exhibited antiviral activity against both non-enveloped and enveloped viruses.
Viral inhibition tests employed to determine the antiviral activity of the GO-AgNPs complex demonstrated that this complex suppressed 25% of feline coronavirus (FCoV) infection, while inhibiting 23% of IBDV infection. By contrast, GO alone was found to inhibit only 16% of FCoV infection, while showing no antiviral activity against IBDV infection.
SARS-CoV-2 infection has been detected using two main methods: the first utilizes CRISPR, while the second is based on the identification of antibodies specific to viral antigens. Studies have shown that nanosensors are regarded as the most potent way to detect the novel SARS-CoV-2.
Researchers have been able to make a new class of biosensors thanks to the evolution of various carbon-based nanomaterials, like graphene, GO, and carbon nanotubes.
Graphene is specifically regarded as a key element for making biosensors. Besides finding use as graphene-based sensors to detect human diseases, they have also been applied in the areas of agriculture, food, and aquaculture to fight specific viruses.
GO nanosheets exhibit excellent physicochemical and biological properties, and are thus used to make protective equipment against infectious diseases. These properties may provide new benefits for regulating SARS-CoV-2 and minimizing its spread.
A multifunctional cotton fabric with ultra-strong UV protective properties and high electrical conductivity has been made by applying a dispersion of GO's nanosheets to the fabric's surface through the vacuum filtration deposition method.
Figure 4 illustrates the potential multifunction of GO that may prove helpful against the virus at different levels. Thus, GO could be employed as a biosensor to detect viruses and manufacture protective equipment against infectious diseases because of its mechanical properties.
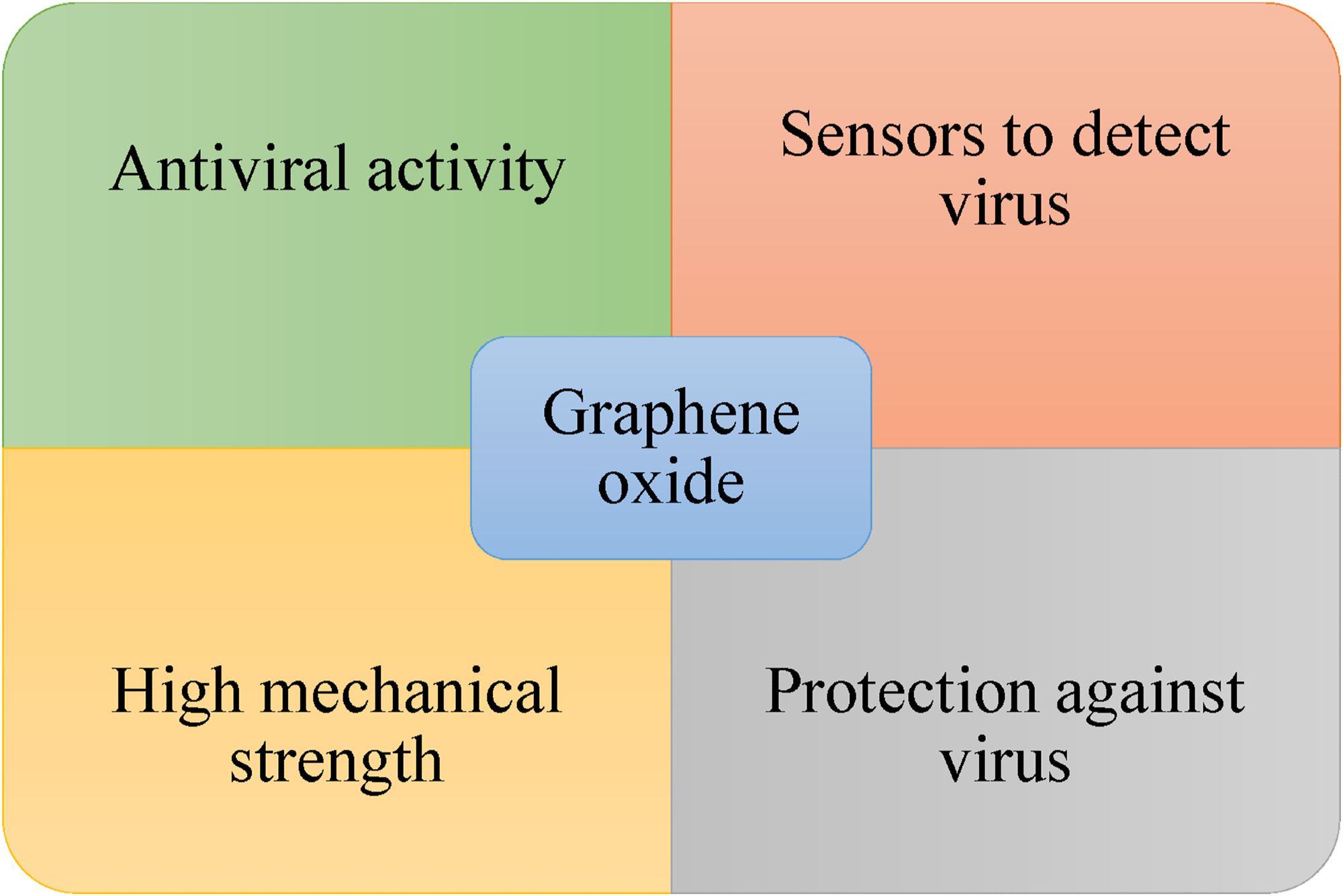
Figure 4. Representative uses of GO to combat viruses. © Rhazouani, et al., 2021
Conclusion
GO-based nanomaterials are potential candidates to tackle different types of viral infections, such as COVID-19.
GO and graphene can inactivate different viruses by various mechanisms, and have been demonstrated to be highly useful for high-throughput diagnostics that involve using a transistor-based biosensor that can detect SARS-CoV-2. Their inclusion in protective equipment against infectious disease is an area of research sure to develop.
Further research is essential to design new GO-based antivirals to combat SARS-CoV-2 and other viral agents. In particular, more insights must be gained into the toxicological mechanisms of nanomaterials to improve their integration into the biomedical field.
Continue reading: Effect of Nano-Perovskite Structure on Suppressing the SARS-CoV-2 Infection.
Journal Reference:
Rhazouani, A., Aziz, K., Gamrani, H., Gebrati, L., Uddin, M. S., Faissal, A. (2021) Can the application of graphene oxide contribute to the fight against COVID-19? Antiviral activity, diagnosis and prevention. Current Research in Pharmacology and Drug Discovery, 2, p. 100062. Available online: https://www.sciencedirect.com/science/article/pii/S2590257121000493?via%3Dihub.
References and Further Reading
- Alayande, A. B., et al. (2019) Surface morphology-dependent spontaneous bacterial behaviors on graphene oxide membranes. Separation and Purification Technology, 226, pp. 68–74. doi.org/10.1016/j.seppur.2019.05.072.
- Adams, M J & Carstens, E B (2012) Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses. Archives of Virology, 157(7), pp. 1411–1422. doi.org/10.1007/s00705-012-1299-6.
- Afroj, S., et al. (2020) Highly Conductive, Scalable, and Machine Washable Graphene-Based E-Textiles for Multifunctional Wearable Electronic Applications. Advanced Functional Materials, 30, p. 2000293. doi.org/10.1002/adfm.202000293.
- Afsahi, S., et al. (2018) Novel graphene-based biosensor for early detection of Zika virus infection. Biosensors and Bioelectronics, 100, pp. 85–88. doi.org/10.1016/j.bios.2017.08.051.
- Akhavan, O., et al. (2012) Protein Degradation and RNA Efflux of Viruses Photocatalyzed by Graphene–Tungsten Oxide Composite Under Visible Light Irradiation. Journal of Physical Chemistry C, 116(17), pp. 9653–9659. doi.org/10.1021/jp301707m.
- Ali, M. A., et al. (2020) (Sensing of COVID-19 Antibodies in Seconds via Aerosol Jet Nanoprinted Reduced-Graphene-Oxide-Coated 3D Electrodes. Advanced Materials, doi.org/10.1002/adma.202006647.
- Aliyev, E., et al. (2019) Structural Characterization of Graphene Oxide: Surface Functional Groups and Fractionated Oxidative Debris. Nanomaterials, 9(8), p. 1180. doi.org/10.3390/nano9081180.
- Bisht, H., et al. (2005) Neutralizing antibody and protective immunity to SARS coronavirus infection of mice induced by a soluble recombinant polypeptide containing an N-terminal segment of the spike glycoprotein. Virology, 334(2), pp. 160–165. doi.org/10.1016/j.virol.2005.01.042.
- Broughton, J. P., et al. (2020) CRISPR–Cas12-based detection of SARS-CoV-2. Nature Biotechnology, 38, pp. 870–874. doi.org/10.1038/s41587-020-0513-4.
- Chen, Y.-N., et al. (2021) Antiviral Activity of Graphene–Silver Nanocomposites against Non-Enveloped and Enveloped Viruses. International Journal of Environmental Research and Public Health, 13(4), p. 430. doi.org/10.3390/ijerph13040430.
- Cui, J., et al. (2019) Origin and evolution of pathogenic coronaviruses. Nature Reviews Microbiology, 17, pp. 181–192. doi.org/10.1038/s41579-018-0118-9.
- Du, X., et al. (2019) Hypericin-loaded graphene oxide protects ducks against a novel duck reovirus. Materials Science and Engineering: C, 105, p. 110052. doi.org/10.1016/j.msec.2019.110052.
- Dasari Shareena, T. P., et al. (2018) A Review on Graphene-Based Nanomaterials in Biomedical Applications and Risks in Environment and Health. Nano-Micro Letters, 10(3), p. 53. doi.org/10.1007/s40820-018-0206-4.
- Das, S., et al. (2013) Oxygenated Functional Group Density on Graphene Oxide: Its Effect on Cell Toxicity. Particle & Particle Systems Characterization, 30(2), pp. 148–157. doi.org/10.1002/ppsc.201200066.
- De Maio, F., et al. (2021) Graphene nanoplatelet and graphene oxide functionalization of face mask materials inhibits infectivity of trapped SARS-CoV-2. iScience, 24(7), p. 102788. doi.org/10.1016/j.isci.2021.102788.
- de Wilde, A. H., et al. (2017) Host factors in coronavirus replication. Current Topics in Microbiology and Immunology, 419, pp. 1–42. doi.org/10.1007/82_2017_25.
- de Wit, E., et al. (2016) SARS and MERS: recent insights into emerging coronaviruses. Nature Reviews Microbiology, 14, pp. 523–534. doi.org/10.1038/nrmicro.2016.81.
- Du, T., et al. (2018) Antiviral Activity of Graphene Oxide–Silver Nanocomposites by Preventing Viral Entry and Activation of the Antiviral Innate Immune Response. ACS Applied Bio Materials, 1(5), pp. 1286–1293. doi.org/10.1021/acsabm.8b00154.
- Ege, D., et al. (2017) Graphene Oxide/Polymer-Based Biomaterials. Advanced Engineering Materials, 19(12), p. 1700627. doi.org/10.1002/adem.201700627.
- Elechiguerra, J. L., et al. (2005) Interaction of silver nanoparticles with HIV-1. Journal of Nanobiotechnology, 3(1), p. 6. doi.org/10.1186/1477-3155-3-6.
- Gholami, M. F., et al. (2017) Functionalized Graphene as Extracellular Matrix Mimics: Toward Well-Defined 2D Nanomaterials for Multivalent Virus Interactions. Advanced Functional Materials, 27(15), p. 1606477. doi.org/10.1002/adfm.201606477.
- Hanff, T. C., et al. (2020) Is there an association between COVID-19 mortality and the renin-angiotensin system? A call for epidemiologic investigations. Clinical Infectious Diseases, 71(15), pp. 870–874. doi.org/10.1093/cid/ciaa329.
- Hoffman, M. R., et al. (1995) Environmental Applications of Semiconductor Photocatalysis. Chemical Reviews, 95(1), pp. 69–96. doi.org/10.1021/cr00033a004.
- Hoffman, M., et al. (2020) SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell, 181(2), pp. 271–280. doi.org/10.1016/j.cell.2020.02.052.
- Hu, X., et al. (2012) Covalently synthesized graphene oxide-aptamer nanosheets for efficient visible-light photocatalysis of nucleic acids and proteins of viruses. Carbon, 50(8), pp. 2772–2781. doi.org/10.1016/j.carbon.2012.02.038.
- Hu, X., et al. (2019) surface-modified graphene oxide-based cotton fabric by ion implantation for enhancing antibacterial activity. ACS Sustainable Chemistry & Engineering, 7(8), pp. 7686–7692. doi.org/10.1021/acssuschemeng.8b06361.
- Hummers, W. S., et al. (1958) Preparation of Graphitic Oxide. Journal of American Chemical Society, 80(6), p. 1339. doi.org/10.1021/ja01539a017.
- Jeremiah, S. S., et al. (2020) Potent antiviral effect of silver nanoparticles on SARS-CoV-2. Biochemical and Biophysical Research Communications, 533(1), pp. 195–200. doi.org/10.1016/j.bbrc.2020.09.018.
- Joshi, S. R., et al. (2020) Low cost synthesis of reduced graphene oxide using biopolymer for influenza virus sensors. Mater Sciences and Engineering C, 108, p. 110465. doi.org/10.1016/j.msec.2019.110465.
- Kam, Y. W., et al. (2007) Antibodies against trimeric S glycoprotein protect hamsters against SARS-CoV challenge despite their capacity to mediate FcγRII-dependent entry into B cells in vitro. Vaccine, 25(4), pp. 729–774. doi.org/10.1016/j.vaccine.2006.08.011.
- Kedambaimoole, V., et al. (2021) Reduced Graphene Oxide Tattoo as Wearable Proximity Sensor. Advanced Electronic Materials, 7(4), p. 2001214. doi.org/10.1002/aelm.202001214.
- Krishnamoorthy, K., et al. (2011) Graphene oxide as a photocatalytic material. Applied Physics Letters, 98, p. 244101. doi.org/10.1063/1.3599453.
- Krishnan, S., et al. (2011) Innate immune responses in respiratory syncytial virus infections. Viral Immunology, 17(2), pp. 220–233. doi.org/10.1089/0882824041310612.
- Kumar, A., et al. (2020) Role of graphene in biosensor and protective textile against viruses. Medical Hypotheses, 144, p. 110253. doi.org/10.1016/j.mehy.2020.110253.
- Raghav, P. K., et al. (2020) Are graphene and graphene-derived products capable of preventing COVID-19 infection? Medical Hypotheses, 144, p. 110031. doi.org/10.1016/j.mehy.2020.110031.
- Laghrib, F., et al. (2021) Current progress on COVID-19 related to biosensing technologies: New opportunity for detection and monitoring of viruses. Microchemical Journal, 160, p. 105606. doi.org/10.1016/j.microc.2020.105606.
- Lambert, T. N., et al. (2009) synthesis and characterization of Titania Graphene nanocomposites. Journal of Physical Chemistry C, 113(46), pp. 19812–19823. doi.org/10.1021/jp905456f.
- Lau, S. K. P., et al. (2018) Discovery and sequence analysis of four delta coronaviruses from birds in the Middle East reveal interspecies jumping with recombination as a potential mechanism for avian-to-avian and avian-to-mammalian transmission. Journal of Virology, 92(15), p. e00265-18. doi.org/10.1128/JVI.00265-18.
- Li, F., et al. (2016) Structure, Function, and Evolution of Coronavirus Spike Proteins. Annual Review of Virology, 3, pp. 237–261. doi.org/10.1146/annurev-virology-110615-042301.
- Abd Muain, M. F., et al. (2018) Gold nanoparticle-decorated reduced-graphene oxide targeting anti hepatitis B virus core antigen. Bioelectrochemistry, 122, pp. 199–205. doi.org/10.1016/j.bioelechem.2018.04.004.
- Möritz, M., et al. (2001) Capability of air filters to retain airborne bacteria and molds in heating, ventilating and air-conditioning (HVAC) systems. International Journal of Hygiene and Environmental Health, 203(5–6), pp. 401–409. doi.org/10.1078/1438-4639-00054.
- Mousavizadeh, L & Ghasemi, S (2021) Genotype and phenotype of COVID-19: Their roles in pathogenesis. Journal of Microbiology, Immunology and Infection, 54(2), pp. 159–163. doi.org/10.1016/j.jmii.2020.03.022.
- Navakul, K., et al. (2017) A novel method for dengue virus detection and antibody screening using a graphene-polymer based electrochemical biosensor. Nanomedicine: Nanotechnology, Biology and Medicine, 13(2), pp. 549–557. doi.org/10.1016/j.nano.2016.08.009.
- Nguyen, E. P., et al. (2020) Recent advancement in biomedical applications on the surface of two-dimensional materials: from biosensing to tissue engineering. Nanoscale, 12(37), pp. 19043–19067. doi.org/10.1039/D0NR05287F.
- Nguyen, N. H. L., et al. (2021) COVID-19 Spike Protein Induced Phononic Modification in Antibody-Coupled Graphene for Viral Detection Application. ACS Nano, 15(7), pp. 11743–11752. doi.org/10.1021/acsnano.1c02549.
- Khanam, P N & Hasan, A (2019) Biosynthesis and characterization of graphene by using non-toxic reducing agent from Allium Cepa extract: Antibacterial properties. International Journal of Biological Macromolecules, 126, pp. 151–158. doi.org/10.1016/j.ijbiomac.2018.12.213.
- Omar, N. A. S., et al. (2019) Sensitive surface plasmon resonance performance of cadmium sulfide quantum dots-amine functionalized graphene oxide based thin film towards dengue virus E-protein. Optics & Laser Technology, 114, pp. 204–208. doi.org/10.1016/j.optlastec.2019.01.038.
- Palmeri, V., et al. (2017) Bacteria Meet Graphene: Modulation of Graphene Oxide Nanosheet Interaction with Human Pathogens for Effective Antimicrobial Therapy. ACS Biomaterials Science & Engineering, 3(4), pp. 619–627. doi.org/10.1021/acsbiomaterials.6b00812.
- Palmieri, V & Papi, M (2020) Can graphene take part in the fight against COVID-19? Nanotoday, 33, p. 100883. doi.org/10.1016/j.nantod.2020.100883.
- Pan, N., et al. (2018) Fabrication of Cotton Fabrics through In-Situ Reduction of Polymeric N-Halamine Modified Graphene Oxide with Enhanced Ultraviolet-Blocking, Self-Cleaning, and Highly Efficient, and Monitorable Antibacterial Properties. Colloids and Surfaces. Available at: https://agris.fao.org/search/en/records/6473b8e913d110e4e7abfc9e.
- Paredes, J. I., et al. (2008) Graphene Oxide Dispersions in Organic Solvents. Langmuir, 24(19), pp. 10560–10564. doi.org/10.1021/la801744a.
- Rhazouani, A., et al. (2021) Synthesis and Toxicity of Graphene Oxide Nanoparticles: A Literature Review of In Vitro and In Vivo Studies. BioMed Research International, 2021. doi.org/10.1155/2021/5518999.
- Ricco, M., et al. (2020) Point-of-care diagnostic tests for detecting SARS-CoV-2 antibodies: a systematic review and meta-analysis of real-world data. Journal of Clinical Medicine, 9(5), p. 1515. doi.org/10.3390/jcm9051515.
- Sametband, M., et al. (2014) Herpes Simplex Virus Type-1 Attachment Inhibition by Functionalized Graphene Oxide. ACS Applied Materials & Interfaces, 6(2), pp. 1228–1235. doi.org/10.1021/am405040z.
- Seah, I., et al. (2020) Revisiting the dangers of the coronavirus in the ophthalmology practice. Eye, 34, pp. 1155–1157. doi.org/10.1038/s41433-020-0790-7.
- Seifi, T & Kamali, A R (2021) Anti-pathogenic activity of graphene nanomaterials: A review. Colloids and Surfaces B: Biointerfaces, 199, p. 111509. doi.org/10.1016/j.colsurfb.2020.111509.
- Seifi, T & Kamali, A R (2021) Antiviral performance of graphene-based materials with emphasis on COVID-19: A review. Medicine in Drug Discovery, 11, p. 100099. doi.org/10.1016/j.medidd.2021.100099.
- Seo, G., et al. (2020) Rapid Detection of COVID-19 Causative Virus (SARS-CoV-2) in Human Nasopharyngeal Swab Specimens Using Field-Effect Transistor-Based Biosensor. ACS Nano, 14(4), pp. 5135–5142. doi.org/10.1021/acsnano.0c02823.
- Song, Z., et al. (2015) Virus Capture and Destruction by Label-Free Graphene Oxide for Detection and Disinfection Applications. Small, 11(9–10), pp. 1171–1176. doi.org/10.1002/smll.201401706.
- Sun, L., et al. (2017) Antibacterial Activity of Graphene Oxide/g-C3N4 Composite through Photocatalytic Disinfection under Visible Light. ACS Sustainable Chemistry & Engineering, 5(10), pp. 8693–8701. doi.org/10.1021/acssuschemeng.7b01431.
- Tang, X., et al. (2015) Functionalization of cotton fabric with graphene oxide nanosheet and polyaniline for conductive and UV blocking properties. Synthetic Metals, 202, pp. 82–88. doi.org/10.1016/j.synthmet.2015.01.017.
- Walls, A. C., et al. (2020) Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell, 181(2), pp. 281–292. doi.org/10.1016/j.cell.2020.02.058.
- Wang, K., et al. (2019) Morphology- and Crystalline Composition-Governed Activity of Titania-Based Photocatalysts: Overview and Perspective. Catalysts, 9(12), p. 1054. doi.org/10.3390/catal9121054.
- Weingart, H., et al. (2004) Immunization with modified vaccinia virus Ankara-based recombinant vaccine against severe acute respiratory syndrome is associated with enhanced hepatitis in ferrets. Journal of Virology, 78(22), pp. 12672–12676. doi.org/10.1128/jvi.78.22.12672-12676.2004.
- Woo, P. C. Y., et al. (2012) Discovery of seven novel Mammalian and avian coronaviruses in the genus delta coronavirus supports bat coronaviruses as the gene source of alphacoronavirus and beta coronavirus and avian coronaviruses as the gene source of gamma coronavirus and delta coronavirus. Journal of Virology, 86 (7), pp. 3995–4008. doi.org/10.1128/jvi.06540-11.
- Yang, X. X., et al. (2017) Synergistic antiviral effect of curcumin functionalized graphene oxide against respiratory syncytial virus infection. Nanoscale, 9(41), pp. 16086–16092. doi.org/10.1039/C7NR06520E.
- Ye, S., et al. (2015) Antiviral Activity of Graphene Oxide: How Sharp Edged Structure and Charge Matter. ACS Applied Materials & Interfaces, 7(38), pp. 21571–21579. doi.org/10.1021/acsami.5b06876.
- Ziem, B., et al. (2016) Highly Efficient Multivalent 2D Nanosystems for Inhibition of Orthopoxvirus Particles. Advanced Healthcare Materials, 5(22), pp. 2922–2930. doi.org/10.1002/adhm.201600812.
- Zaleska, A., (2008) Doped-TiO2: A Review. Recent Patents on Engineering, 2(3), pp. 157–164. doi.org/10.2174/187221208786306289.
- Zhou, P., et al. (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 579, pp. 270–273. doi.org/10.1038/s41586-020-2012-7.
- Ziem, B., et al. (2017) Size-dependent inhibition of herpesvirus cellular entry by polyvalent nanoarchitectures. Nanoscale, 9(11), pp. 3774–3783. doi.org/10.1039/c7nr00611j.
- Allocution liminaire du (2020) Allocution liminaire du Directeur général de l’OMS lors du point presse sur la COVID-19 - 11 mars 2020. Available at: https://www.who.int/fr/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (Accessed 27 October 2020).
- Natarajan, A., et al. (2017) An Elegant Analysis of White Spot Syndrome Virus Using a Graphene Oxide/Methylene Blue based Electrochemical Immunosensor Platform. Scientific Reports, 7(1). doi.org/10.1038/srep46169.
- MINING.COM (2020) Graphene Ink May Be Used to Fight Coronavirus, May 01. Available at: https://www.mining.com/graphene-ink-may-be-used-to-fight-novel-coronavirus/ (Accessed 11 November 2020).
- Unicef (2020) Maladie à coronavirus (COVID-19). Available at: https://www.unicef.org/morocco/recits/maladie-%C3%A0-coronavirus-covid-19 (Accessed 27 October 2020).
- StatNano.com (2020) Synergistic Antiviral Activity of Graphene Oxide and Common Antiviral Agents. Available at: https://statnano.com/news/67505/Synergistic-Antiviral-Activity-of-Graphene-Oxide-and-Common-Antiviral-Agents (Accessed 10 October 2020).