Molecular dynamics (MD) simulations were carried out to study ice nucleation and growth. Different factors affecting the nucleation like the surface configuration, surface hydrophilicity, hindering effects, lateral expansion of ice nucleus, boundary misorientation were studied in detail. This microscopic study of nucleation and growth inhabitation will help in the design of anti-icing surface systems.
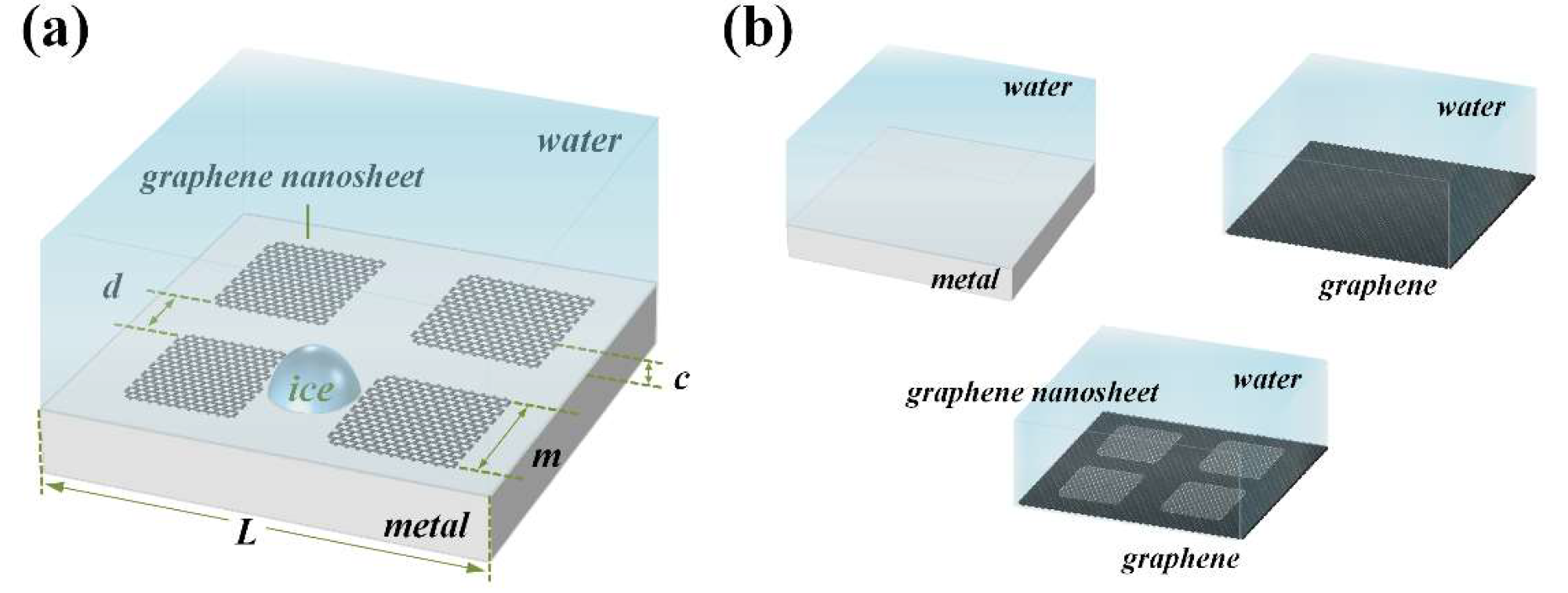
Illustrations of simulation systems. (a) Model of the metal–graphene nanosheet surface that was covered with a box of liquid water. A typical ice nucleus formation is illustrated on the surface. (b) Models of three other simulation systems. Pure metal, pure graphene and graphene–graphene nanosheet surface systems. © Jiang, B. et al (2022)
Uses of Graphene
Graphene has an effect on the nucleation and growth of the ice as it can restrain the formation of ice under certain conditions. Different researches have shown that graphene is light in weight and can be used as a heater in anti-icing systems.
Controlling Factors
There are different controlling factors for the formation of the heterogeneous ice nucleation: the coupling of the surface hydrophilicity and surface crystallinity, and the dynamics between the surface hydrophilicity and the surface morphology.
What the Researchers Did
The team used a graphene nanosheet to create a surface hydrophilicity configuration on a metal surface, while MD was used to examine the features of the nucleation and growth of ice on the surfaces.
Different samples of the surface configurations were taken, and the process and initialization of ice nucleation and growth under specific conditions were studied. All these samples were of different surface hydrophilicity discrepancies between a graphene nanosheet and metal substrate. The growth rate and the disorientation between crystals of the ice nucleus were also calculated through the molecular dynamics process.
Molecular Dynamics Simulation Method
In order to explore the delay in freezing of the surface, different MD simulations were carried out.
Metal–graphene nanosheets were used in the system for MD simulations. In addition, three more simulation systems were built for comparison, which included pure metal, pure graphene, and graphene–graphene nanosheets surface simulations.
These simulations were carried by varying interaction strength as the main element. A monoatomic water (mW) model was used to represent the interactions between water molecules in this study.
Excellent structural properties and a melting point close to the experiment characterize this particular water model. Short-range two-body and three-body interactions between water molecules were considered part of the mW model. Because it is a single atom, its dynamics were much faster, which reduces the computational burden of ice-freezing models.
Each cooling simulation was run five times to ensure that the results were statistically sound. During the simulations, the start and stop of the icing process were monitored. When the number of water molecules in the ice nucleus began increase rapidly, the icing process began. On the other hand, the end of icing was chosen as the point when the ice nucleus became stable.
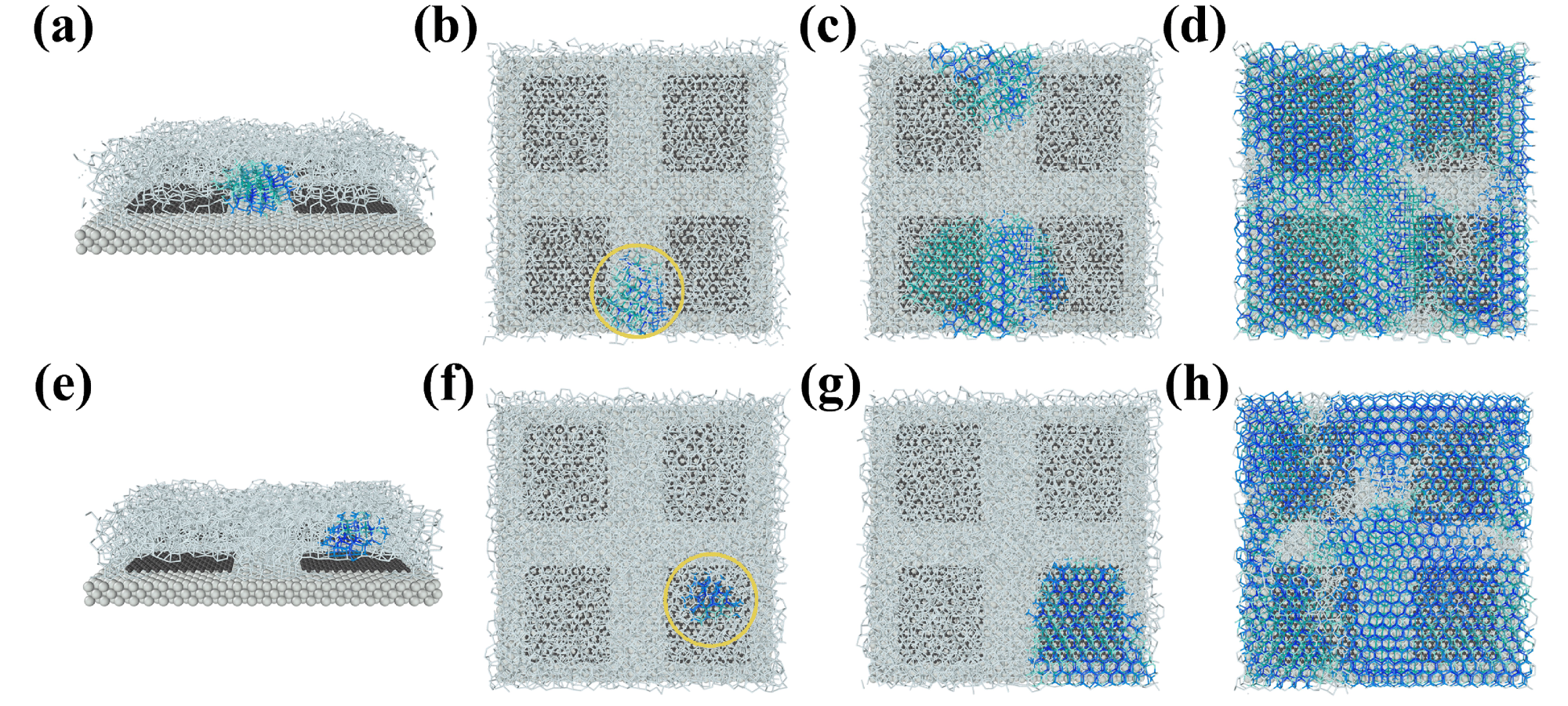
Snapshots of the preferential site of ice nucleation and subsequent growth of ice crystals on a metal–graphene surface. (a) Ice nucleation at the clearance of the surface, εw-m = 4.0 kcal/mol. (b–d) Representative growth trajectories of an ice nucleus that appeared at the clearance, which is indicated by a yellow circle (from the top view). (e) Ice nucleation at the top of the graphene nanosheet, εw-m = 1.0 kcal/mol. (f–h) Representative growth trajectories of an ice nucleus that appeared at the top of the graphene nanosheet. Silver and black spheres represent metal and carbon atoms. White sticks covered on them represent liquid water molecules, and blue and green sticks connect pairs of ice molecules with hexagonal and cubic structure orders, respectively. © Jiang, B. et al (2022)
Important Findings of Study
In this study, it was possible to explore the freezing delay capabilities of metal–graphene nanosheets surfaces using molecular dynamics simulations and to get microscopic insight into the processes that limit the ice nucleation and growth in the clearance configuration.
When surface hydrophilicity of the metal substrate εw-m was higher than 1 kcal/mol, preventing other water molecules from interfering with the metal substrate, it created an interfacial layer of water. A surface hydrophilicity discrepancy of 5.87 kcal/mol delayed ice nucleation by up to 5.25 ns inside the εw-m range of 4.0 to 14.0 kcal/mol. When εw-m was 8.0 kcal/mol, the ice nucleus developed at the slowest pace. Since the metal substrate surface hydrophilicity εw-m exceeded 1.0 kcal/mol, the ice embryo's nucleation site migrated from the top of graphene nanosheets to the clearance.
Future Prospects
This study presents graphene nanosheet's configuration of surface hydrophilicity towards the inhibition of ice nucleation and growth. In addition, the stunting effect, which is proportional to the contact area between ice crystals, can restrict ice growth when the boundary between ice crystals is disoriented. These findings give a basic model to examine the mechanism of nano-sized graphene and its derivatives on ice nucleation, growth, and their potential application for anti-icing materials.
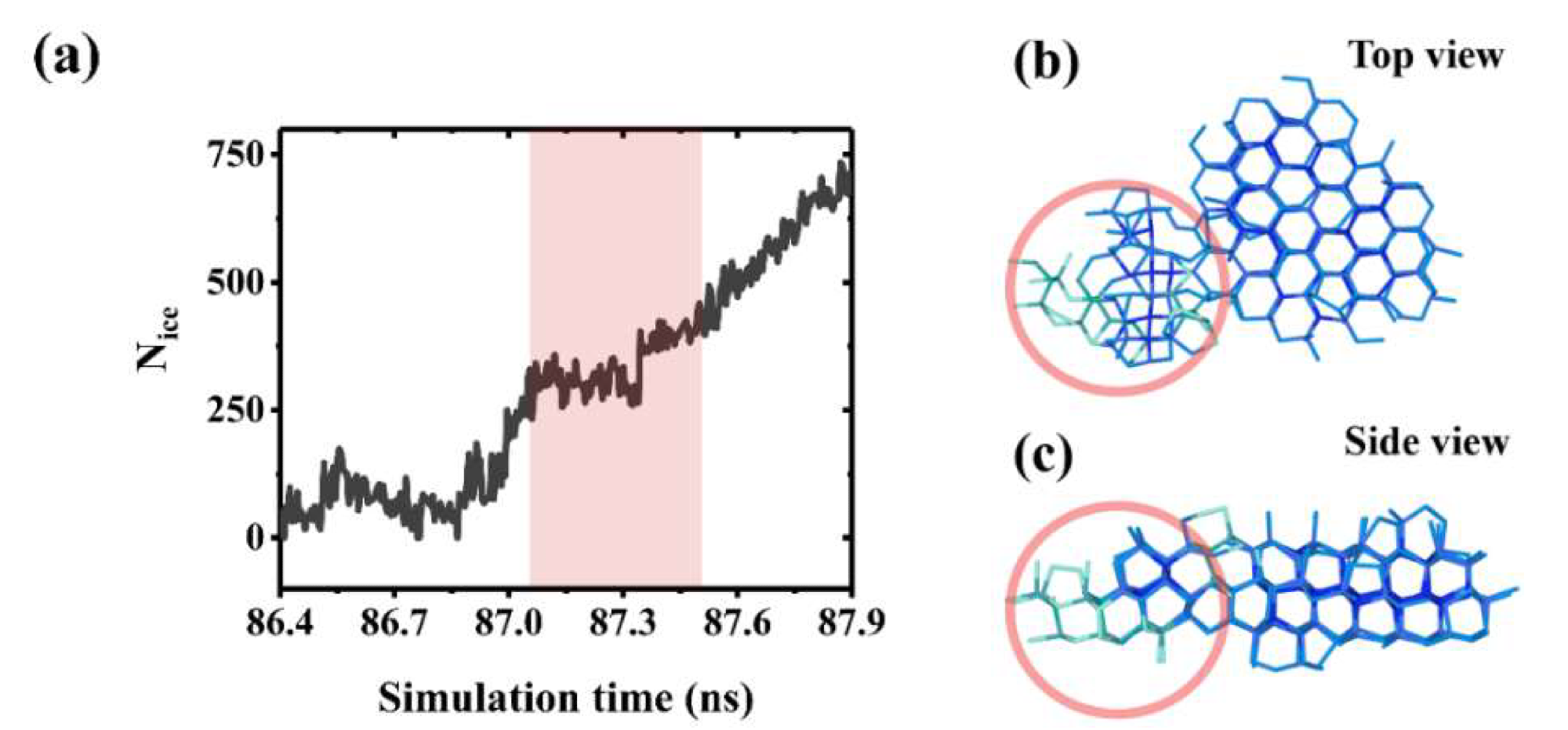
Stunting effect of a boundary misorientation during ice growth. (a) The inset shows the stunting period (shaded red) in the ice growth process caused by boundary misorientation between ice crystals. (b) Top view snapshots of boundary misorientation between ice crystals. (c) Side view of the boundary misorientation between ice crystals. © Jiang, B. et al (2022)
Continue reading: Heating up Graphene to Study Ice Formation
Reference
Jiang, B. et al (2022) Patterning Configuration of Surface Hydrophilicity by Graphene Nanosheet towards the Inhibition of Ice Nucleation and Growth. Coatings. Available at: https://www.mdpi.com/2079-6412/12/1/52
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.