
Image Credit: Egorov Artem/Shutterstock.com
Synthesized graphene can be made by mixing water, detergents, and solvents with graphite powder at ambient temperature and atmospheric pressure. This method was developed by Jonathan N. Coleman, a professor at Trinity College Dublin.
During the process, graphite powder fragments into nanosheets, and the solvents in the mixture prevent the graphene from aggregating. Generally, the exfoliation process generates small flakes, also called platelets, of graphene, that are between one and six layers thick, as opposed to one single layer. The monolayer content can be raised to 75 percent with an additional step.
Coleman produced other 2D materials using this mixing process, such as boron nitride and molybdenum disulfide. Coleman’s method has led some market analysts to claim that the less precise and smaller graphene flakes made via this method are suitable for 90 percent of graphene’s applications in areas such as coatings and plastic composites. In comparison, liquid-phase exfoliation does not produce the large, blemish-free sheets required for making displays and high-end electronics like CVD synthesis does.
Process Details
Liquid-phase exfoliation (LPE) disperses graphite flakes into a solution using intercalating agents, which are molecules that place themselves between layers, and surfactants. It is supported by mechanical processing steps, like controlled ball milling, or solvothermal or sonochemical processes. This method generates a scalable form of graphene that consists of multiple layers, making micron-scale platelets called graphene nanoplatelets. (GNPs) at a low cost.
For this process, the most widely used solvent is NMP (N-Methyl-2-pyrrolidone), which is available in high volumes because it is often used in the petrochemical industry. Although it is labeled as a toxic substance, it is still easy to handle, but it can prove costly for production, in particular when marketing the graphene as ink.
However, issues surrounding choosing a solvent can be avoided if the right surfactants are used. The surfactants work as a type of wall that forms and surrounds the graphene surfaces, blocking the re-aggregation of the graphene flakes into graphite while submerged in an aqueous solution. The surfactants are also able to mask the thermal and electrical performance of the materials.
The type of surfactants used in this method is a key difference between producers. In the first step of the process, natural graphite is submerged in a solvent such as NMP or likewise. The bath is then vibrated ultrasonically in order to exfoliate the graphene. The result is graphene that is a blend of multi- and single-layer sheets that vary in flake sizes. After this, several rounds of solution filtering take place to separate the graphene according to size and the number of layers.
When each of these steps has been completed, the graphene is ready to use, semidispersed, in an organic solvent. Further processing is needed for the graphene to become a functional ink or masterbatch.
Sonication
Sonication is one of the steps used in LPE methods and produces monolayer or few-layer graphene at relatively high concentrations. The key mechanism through which sonication induces physical or chemical changes is through its triggering of cavitation bubbles. These cavitation bubbles are produced as the ultrasonic waves proliferate through the medium, and create both high-pressure and low-pressure areas that push and pull at the molecules.
There are two types of sonication that can be used separately or in conjunction: bath sonication and tip sonication.
Sonication-assisted liquid-phase exfoliation (LPE) involves three steps:
- The dispersion of graphite in a solvent
- The exfoliation of dispersion via sonication
- The purification (filtration) of the graphene
The degree of exfoliation depends on the power of the sonication, the liquid medium used to disperse the graphene sheets, and the rate of centrifugation. Centrifugation is responsible for removing large and unstably dispersed graphite particles or aggregates.
High Shear Mixing
LPE also makes use of high-shear mixing as an exfoliation technique. High-shear mixing and sonication are alike in that aqueous liquids can be used to allow graphite to be exfoliated and provide stable dispersions of graphene, which means there is no need to use harmful organic liquids.
Again, Coleman was at the forefront of the move towards high-shear mixing when he and his colleagues used high-shear exfoliation. Together they were able to demonstrate that high-shear mixing of graphite in certain successful solvents could result in highly concentrated dispersions of graphene nanosheets. The graphene flakes featured no oxygen molecules on their surface and were generally free of defects. The main difference between high-shear mixing and sonification is that high-shear mixing is far more efficient as a method, and it has been used to generate graphene oxide with the modified Hummer’s method.
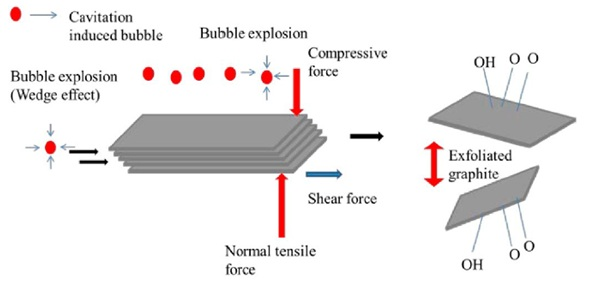
Figure 1. Illustration of Graphite Exfoliation Mechanism. Source: Multidisciplinary Digital Publishing Institute, 2016.
Microfluidization.
Microfluidization is a high-pressure homogenization technique. It exerts high pressure on the fluid, which pushes the fluid through a microchannel. The main benefit of microfluidization is that the mild exfoliation it causes can lower the number of defects in the graphene.
Quantum dots and graphene-based conductive inks have recently been produced via microfluidization techniques. Microfluidization is superior in that when compared to sonication and high-shear mixing, the high shear rate is present in the whole fluid region. Exfoliation in microfluidization is caused by cavitation, shear stress, and collision, and in the process, the dispersion of graphite is forced into the chamber channel and the shear force-controlled exfoliation.
Micro jets cause the collisions and produce shear forces, in which monolayer or few-layer graphene can be self-peeled down because of their capacity for lateral self-lubrication. The function of cavitation is similar to the sonication assisted LPE.
Scalability and Market Position
In the production of bulk graphene, liquid-phase exfoliation has become one of the top techniques of choice. It is highly scalable because the precursor for the process is graphite, and it also offers low production costs. If the filtering stage of LPE is eliminated, there are only a small number of processing steps and solvents needed.
Crucially, the whole process is physical, which leaves almost no damage to the resulting graphene structure. This is in stark contrast to reduced graphene oxide (rGO graphene), where the rGO process permanently damages the graphene lattice because of the chemical routes involved.
As discussed, the liquid phase exfoliation process can easily be scaled up. The size of the chamber can be changed without needing to consider strict condition controls, unlike chemical reactions. However, the challenge in scaling up LPE is in keeping yields high while also retaining low production costs. This is due to the fact that a lot of the graphite is not converted to graphene, but remains as a micro-graphite material. The unused graphite can be recycled for further use if the process is well designed.
As the resulting graphene is not permanently degraded or damaged, its use could extend to higher-end applications, similar to the ways in which CVD graphene is used. Although the resulting graphene does not create in the large-area sheets seen in CVD graphene, it can be carried out more cheaply than CVD techniques.

This information has been sourced, reviewed and adapted from materials provided by The Graphene Council.
For more information on this source, please visit The Graphene Council.