AZoNano speaks to Dr. John Miao about his involvement in the breakthrough research that could rewrite our understanding of how substances are formed at an atomic level. Using an innovative atomic electron tomography technique, researchers were able to reveal how atoms were arranged in amorphous Pd nanoparticles and Ta thin films. Read more about the research behind this exciting development, and what it could mean for how we view the physical world around us.
Please could you introduce yourself and tell us what inspired your career into amorphous materials?
Observing the three-dimensional (3D) atomic arrangement of an amorphous solid has been my dream since I was a graduate student. That dream has now been finally realized, after 22 years of relentless pursuit, which can be seen from our two big papers (one published in Nature Materials and the other in Nature this year):
In your own words, could you summarize the results of your research?
For the past 70 years, icosahedral order has been widely considered as the prevalent atomic motif in monatomic liquids and amorphous materials. In the Nature Materials paper, we advanced atomic electron tomography (AET) to determine for the first time the 3D atomic structure of monatomic amorphous materials, including a Ta thin film and two Pd nanoparticles.
Despite different synthesis methods, we found that pentagonal bipyramids are the most abundant atomic motifs in these amorphous materials. Instead of forming icosahedral order, the pentagonal bipyramids are closely connected with each other to form networks that extend to a medium-range scale.
Our molecular dynamics simulations, coupled with experimental results, further revealed that the 3D atomic structure of monatomic liquids resembles those of the experimental amorphous materials. Moreover, we found that pentagonal bipyramid networks (PBNs) are also prevalent in the monatomic liquid.
During the quench from a liquid to metallic glass state, the PBNs quickly grow in size and form a small fraction of icosahedral order, indicating an important role of the PBNs during the glass transition.
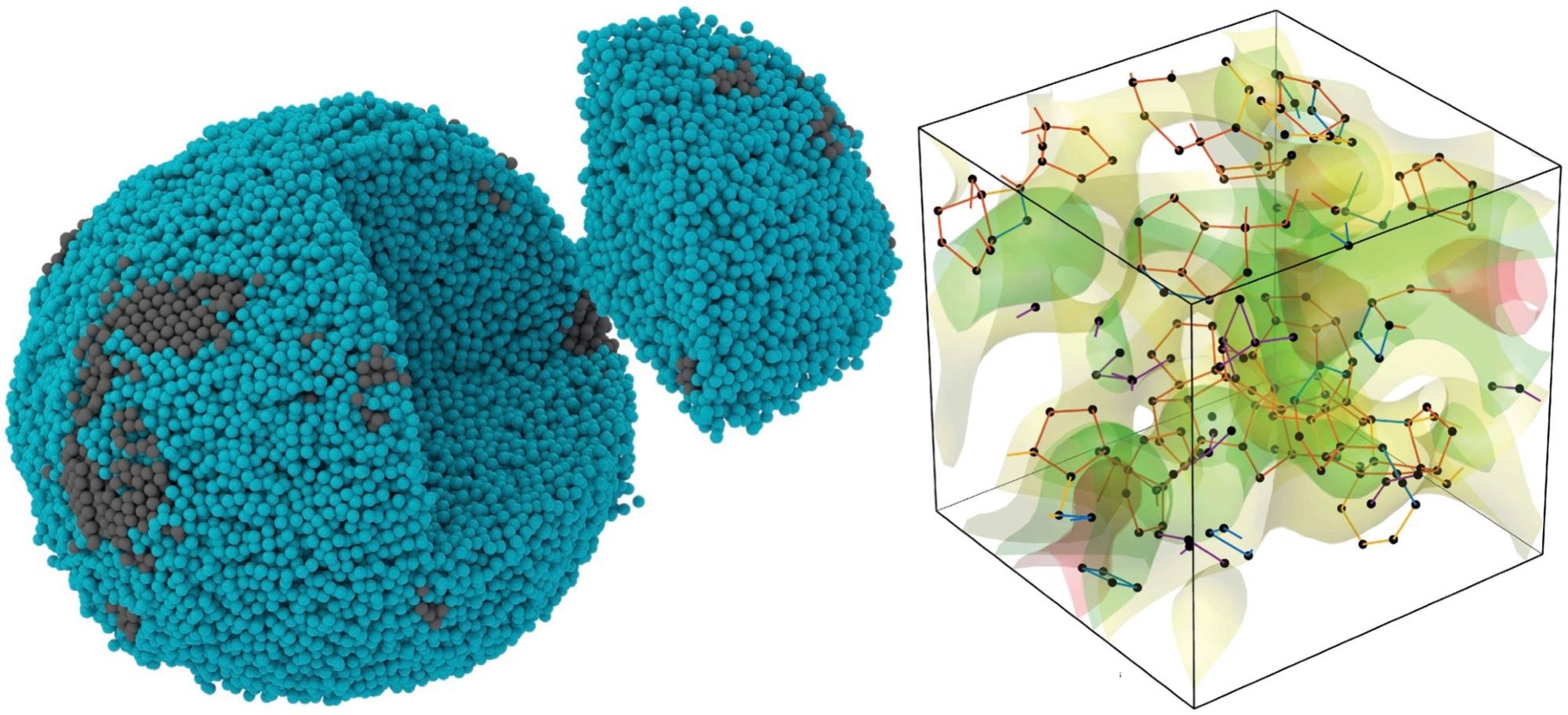
Experimental 3D atomic image of a tiny, amorphous palladium particle (left), in which the 10-sided pentagonal bipyramid (right) is the most prevalent motif for how atoms pack together; the orange lines represent the pentagonal bipyramid shape. Image Credit: Yakun Yuan and John Miao/UCLA
Results from the 3D imaging indicate a major shift from current understandings of how atoms in amorphous materials are structured. How did you approach this area of research?
Due to the lack of long-range order, the 3D atomic structure of amorphous materials had eluded direct experimental determination for more than a century. We solved this long-standing problem by developing atomic electron tomography, which can determine the 3D atomic positions of real materials without assuming crystallinity.
What were the main challenges that had to be overcome?
Achieving the 3D atomic structure of amorphous materials came out of two important advances. The experiment was optimized to expose the radiation-sensitive sample to the lowest possible dose needed to generate images, to avoid the electrons changing the sample in the course of the measurement. And advanced algorithms were developed to analyze the noisy images and then stitch them together to obtain a 3D map.
Investigations found that pentagonal bipyramids, atoms arranged in groups of seven, were the most common formation. Why might this be the case?
A pentagonal bipyramid represents the densest packing of seven atoms, but an icosahedron requires the tight packing of 12 pentagonal bipyramids. We found that the majority of pentagonal bipyramids form a network, instead of assembling into icosahedra in the amorphous materials.
Substances that are arranged into crystals are very well understood due to their repetitive pattern. However, much less is understood about substances that lack this level of order. Why is this, and what are the limitations of basing their understanding on theoretical models and indirect experiments?
Over the last century, crystallography has been broadly applied to determine the 3D atomic structure of crystalline samples.
The quantitative 3D structural information has been fundamental to the development of many scientific fields. However, for amorphous solids, their 3D structure has been primarily inferred from experimental data, where the statistical structural information can be obtained but the 3D local structure is lost.
This qualitative approach has hindered our fundamental understanding of the 3D structure of amorphous solids and related phenomena such as the crystal-amorphous phase transition and the glass transition

Image Credit: Nasky/Shutterstock.com
For many years, research and industry have assumed amorphous materials' atomic arrangement to be icosahedral groups of 13. Knowing that this is incorrect, how might this impact the nanotechnology sector?
In our Nature Materials paper (https://www.pa.ucla.edu/index.php), we also determine the 3D atomic structure of an amorphous Ta thin film.
The ability to determine the 3D atomic structure of amorphous thin films is expected to greatly expand the applicability of AET to a broad range of technologically relevant materials. Moreover, the experimental method and results reported in this paper could have an important impact on different fields, ranging from the direct determination of the 3D atomic structure of quasicrystals to the study of the physics of jamming, the amorphous–crystalline phase and glass transitions at the single-atom level.

Image Credit: Gorodenkoff/Shutterstock.com
What characteristics of Pd nanoparticles made them the ideal choice to include in this study? Are there any other nanoparticles that you would be interested in investigating in the future?
In this work, we focused on two amorphous Pd nanoparticles and an amorphous Ta thin film because our collaborators can make these samples. In the future, we will be interested in determining the 3D atomic positions of a wide range of amorphous materials.
Materials that fundamentally exist as single atoms were chosen in this study. How might more complex materials compare with the results of this research? Could more complex materials be investigated in the same manner?
We have also advanced AET to determine the 3D atomic structure of a multi-component metallic glass sample and quantitatively characterized the short- and medium-range order of the 3D atomic arrangement. Please see this Nature paper: https://www.nature.com/articles/s41586-021-03354-0.
What are the next steps for this field of research?
We will continue to apply AET to determine the 3D atomic structure of a wide range of amorphous materials, which could transform our fundamental understanding of non-crystalline materials and related phenomena.
What aspect of this research do you find personally exciting?
I consider the following papers among the best work of my scientific career thus far, which also fulfills my dream as a graduate student.
Where can our readers go to see the progression of this field of research?
UCLA press release: https://newsroom.ucla.edu/releases/first-ever-3d-atomic-imaging-amorphous-solid
Here is a Nature video featuring our research with more than 21M views on YouTube, becoming the most viewed video ever made by Nature in any discipline:
Have you ever seen an atom?
Have you ever seen an atom? Video Credit: nature video/YouTube.com
About Dr. John Miao
 Jianwei (John) Miao is a professor of physics and astronomy and California NanoSystems Institute at the University of California, Los Angeles. In 1999, he performed the seminal experiment of extending x-ray crystallography to allow structural determination of noncrystalline specimens, known as coherent diffractive imaging (CDI), lensless imaging, or computational microscopy. CDI methods such as ptychography and Bragg CDI have been broadly implemented using synchrotron radiation, x-ray free-electron lasers, high harmonic generation, optical lasers, and electrons. Miao has also pioneered atomic electron tomography (AET) and recently led a team that has determined the three-dimensional atomic structure of amorphous materials for the first time.
Jianwei (John) Miao is a professor of physics and astronomy and California NanoSystems Institute at the University of California, Los Angeles. In 1999, he performed the seminal experiment of extending x-ray crystallography to allow structural determination of noncrystalline specimens, known as coherent diffractive imaging (CDI), lensless imaging, or computational microscopy. CDI methods such as ptychography and Bragg CDI have been broadly implemented using synchrotron radiation, x-ray free-electron lasers, high harmonic generation, optical lasers, and electrons. Miao has also pioneered atomic electron tomography (AET) and recently led a team that has determined the three-dimensional atomic structure of amorphous materials for the first time.
Disclaimer: The views expressed here are those of the interviewee and do not necessarily represent the views of AZoM.com Limited (T/A) AZoNetwork, the owner and operator of this website. This disclaimer forms part of the Terms and Conditions of use of this website.