Feb 14 2018
Scientists from Japan have devised a technique to develop two materials, where each is formed of three graphene layers. Graphene is stacked distinctively in each of the materials and has distinctive electrical characteristics. Their study offers inferences for developing innovative electronic devices, for example, photo sensors that transform light into electrical energy.
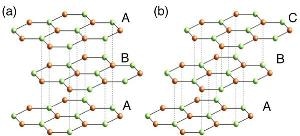 The schematic crystal structures of (a) ABA- and (b) ABC- stacked three-layer graphene.
The schematic crystal structures of (a) ABA- and (b) ABC- stacked three-layer graphene.
In 2004, two researchers found out that they had isolated a single layer of carbon atoms on a scotch tape used for cleaning a graphite crystal. From then on, graphene has attracted the attention of scientists due to its exhilarating characteristics: it is nearly 200 times stronger when compared to steel, highly flexible, and an exceptional conductor of electricity.
The carbon atoms in graphene are arrayed into hexagons, producing a honeycomb-form lattice. When a single layer of graphene is placed on top of another layer, bilayer graphene is formed. The layers can be arrayed in one of two positions. In the first, all of the center points of the hexagons containing carbon atoms in each layer are arranged directly above each another (AA-stacking). The second option is that they can be displaced forward so that the center of the hexagon in one layer is positioned above a carbon atom (AB-stacking). AB-stacking of two graphene layers results in the formation of a material with semiconducting characteristics, by exerting an external electric field.
Intentionally stacking three graphene layers has been challenging. However, achieving this can assist scientists to investigate the change in physical characteristics of trilayer materials depending on the way the layers are stacked. This might result in the creation of innovative electrical devices.
At present, scientists from Tohoku University and Nagoya University in Japan have developed two distinctive types of trilayer graphene with distinctive electrical characteristics.
They adopted one of two techniques to heat silicon carbide. In one method, they heated silicon carbide to 1510 °C under an atmosphere of pressurized argon. In the other procedure, silicon carbide was heated to 1300 °C under a high vacuum condition. Then, both materials were sprayed with hydrogen gas where the bonds were broken to form single hydrogen atoms. Subsequently, two kinds of trilayer graphene were formed.
The silicon carbide heated under the pressurized argon atmosphere formed ABA-stacked graphene, where the hexagons in the top and bottom layers were accurately aligned, whereas the middle layer was displaced to a certain extent. The silicon carbide heated under vacuum formed ABC-stacked graphene, where each layer was displaced to a certain extent in front of the layer underneath it.
Then, the scientists investigated the physical characteristics of each material and discovered that their electrons behaved distinctively.
Like monolayer graphene, the ABA graphene was an exceptional electrical conductor. In contrast, the ABC graphene behaves more like AB graphene, that is, it had semiconducting characteristics.
“The present success in selective fabrication of ABA and ABC trilayer graphene would widen the feasibility of graphene-based nano-electronic devices with variable layer numbers and stacking sequences,” the scientists concluded in their work published in the NPG Asia Materials journal.