Owing to the presence of stable conjugate acid and rigid alkyl functionality, the selected organic bases could easily reduce the CNTs.
Doping the CNTs
The organic-inorganic hybrid or molecular-based devices require n- and p-type material to create p/n junctions. The material polarity can be altered for inorganic semiconductors through doping technology. Moreover, the density and charge carrier type determine the material polarity and thus the functions of the corresponding device. Since the beginning of CNT research, spectroscopic characterization and doping reagents have helped explore the effects of doping.
Doping the CNTs with charge carriers depends on the charge transfer between the π-electron system of CNT and dopants enclosed on the inside or the surface of the nanotube. While electron-accepting materials induce a p-doped state in CNT, the electron-donating materials induce n-doped states.
Due to the presence of surface-adsorbed oxygen, p-type doping on CNT is easier than n-type doping. Additionally, the n-type doping in CNTs is air-sensitive due to dopant volatilization or atmospheric oxygen-induced autoxidation.
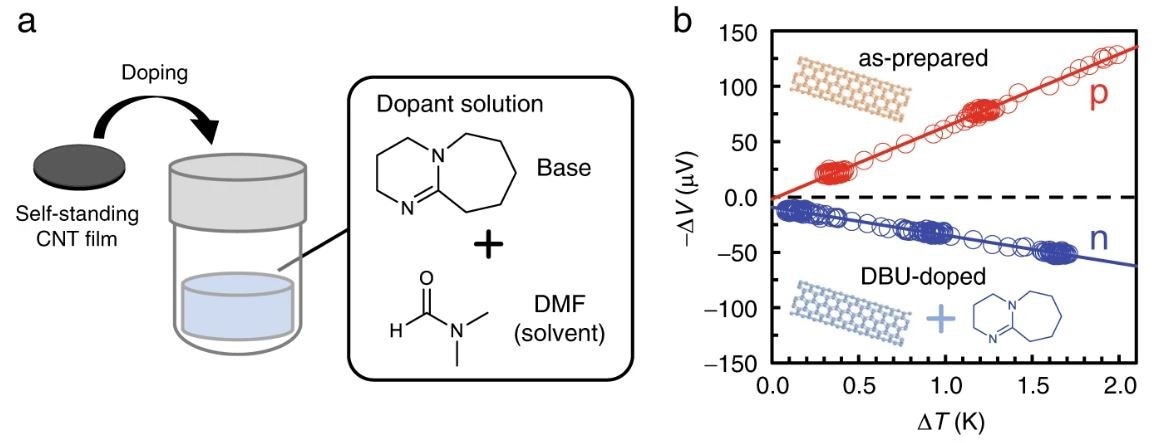
Schematic of doping by immersing the CNT films in a base solution. b Representative plots of thermopower (−ΔV) vs. the supplied temperature difference (ΔT) measured using the as-prepared (p-type) and DBU-doped (n-type) films made from EC1.5-CNT. Seebeck coefficients (S) were determined from the slopes in the plots (S = − ΔV/ΔT). Note that the potential of the lower temperature side was defined as ground. © Horike, S., et al. (2022)
Bicyclic-Ring Base Doping Induced N-Type Conduction in CNTs
In the present study, the researchers used commercially available bicyclic molecular bases as electron donors for CNTs. They tested if the bicyclic guanidine bases could induce thermal stability in n-type CNTs. Thermoelectric charge carrier analysis revealed that employing a solution-based method helped in the successful conversion of fabricated p-type CNTs into thermally stable n-type CNTs.
The excellent thermal stability (for over six months at 100 degrees celsius), facile doping process and base molecule's versatility in structure, meant that the proposed method in this study offered a new route to produce n-type CNTs for molecular electronics.
Research Findings
Thermal stability for doped CNT films with n-type polarity was examined, and after heating at 100 degrees celsius, the electrical conductivity and Seebeck coefficient were measured in air. The researchers observed a significant change in the thermal stability of n-doped CNT material in air based on the applied base’s molecular structures.
The researchers observed that the 1,1,3,3-tetramethylguanidine (TMG) and 1,8-diazabicyclo [5.4.0]-7-undecane (DBU) doped n-type CNT films had low thermal stability. After 222 hours at 100 degrees celsius, the TMG doped n-type CNTs retreated to p-type with a decrease in electrical conductivity to 500-siemens per centimeter. Thus, the TMG-doped CNTs suffered dedoping on heating.
The DBU-doped CNT film was more stable than the TMG-doped CNT. However, its n-type polarity was reverted to p-type after heating for 696 hours. Simultaneously, the electrical conductivity decreased by 70% on heating.
Under the same heating conditions, 1,5,7-triazabicyclo [4.4.0] dec-5-ene (TBD) and methyl (Me)-doped TBD CNT films were more stable than the TMG and DBU-doped counterparts. Both the TBD and Me-TBD showed a negative Seebeck coefficient after approximately 5000 hours. After heating for approximately 2000 hours, a slight increase in negative Seebeck coefficients and a decrease in electrical conductivity revealed the partial de-doping in n-doped CNTs.
With the operation, the electronic and energy devices become heated. Thus, it is crucial to persevere the physical stability of the materials and the thermal stability of n-type doping.
The stability difference based on the molecular structure of bases was due to the difference in (i) surface adsorption ability of bases on nanotubes or their volatilization, (ii) the stabilization of CNT's n-doped state, and (iii) the ability to block oxygen impurity from air causing de-doping.
Among the bases considered for the study (DBU, TBD, TMG, and Me-TBD), regardless of the concentration of the dopant in dimethyl formamide (DMF) solution, TBD- and Me-TBD doped CNT films exhibited the highest power factors. Furthermore, the higher power factors for TBD doping were achieved using polar aprotic solvents due to higher electrical conductivities.
Conclusion
In conclusion, the researchers successfully applied the organic superbases and converted the as-prepared p-type CNTs into their n-type counterparts with negative Seebeck coefficient, good thermal stability in the air, and excellent electrical conductivity. The electron transfer from bicyclic-ring guanidine to CNTs was achieved even at room temperature and the n-type polarity was retained for over six months at 100˚C in air.
Using these results, the researchers highlighted the potential of molecular structure design to explore and develop doping reagents since minor changes in the dopant's molecular structure significantly affected the n-type CNT's stability.
Reference
Shohei Horike, Qingshuo Wei, Kouki Akaike, Kazuhiro Kirihara, Masakazu Mukaida, Yasuko Koshiba et al. (2022) Bicyclic-ring base doping induces n-type conduction in carbon nanotubes with outstanding thermal stability in air. Nature Communications. https://www.nature.com/articles/s41467-022-31179-6
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.