Supercapacitors: Why are They Important?
The current advancement of smart and adaptable electronic products necessitates the creation of a solid-state and high-performance energy storage technology.
Batteries have been widely employed in many industrial applications as conventional energy storage systems. However, they have sluggish charge-discharge cycles due to their diffusion-controlled energy storage system. Batteries also have a poor energy capacity and a short life cycle because of slow reaction kinetics.
Supercapacitors possess notable benefits over typical rechargeable batteries, such as an extended life cycle, greater power density, greater reliability, faster charging and discharge, and reduced pollution.
Supercapacitors are classified as either electrical double-layer capacitors (EDLCs) or pseudocapacitors based on their charge-storing technique. EDLCs store charges by electrostatically trapping electrolytic ions onto the interface of electrode substances without charge transmission, while pseudocapacitors operate through a rapid and reversible interfacial redox process.
Limitations of Presently Used Supercapacitor Electrode Materials
Adaptable solid-state supercapacitors (SSCs) for wearable electronic devices are typically made by encasing a gel electrolyte between two porous electrodes. The scalable production of electrode materials with high power and energy densities is crucial for creating supercapacitors with potential applications in the automotive and power generation industries.
Supercapacitor electrode materials should have excellent electrical conductance, strong chemical and thermal stability, and a large surface area per unit volume. Researchers have made considerable effort in developing two-dimensional (2D) nanoparticles as high-performance electrode substances. However, the issue of restacking during the preparation phase disturbs charge transfer and reduces the number of functional sites.
Many nanomaterials studied as electrode materials to improve the interlayer gap of two-dimensional (2D) materials in supercapacitors possess inherent limitations for large-scale and uninterrupted manufacturing because of their expensive and complicated production methods.
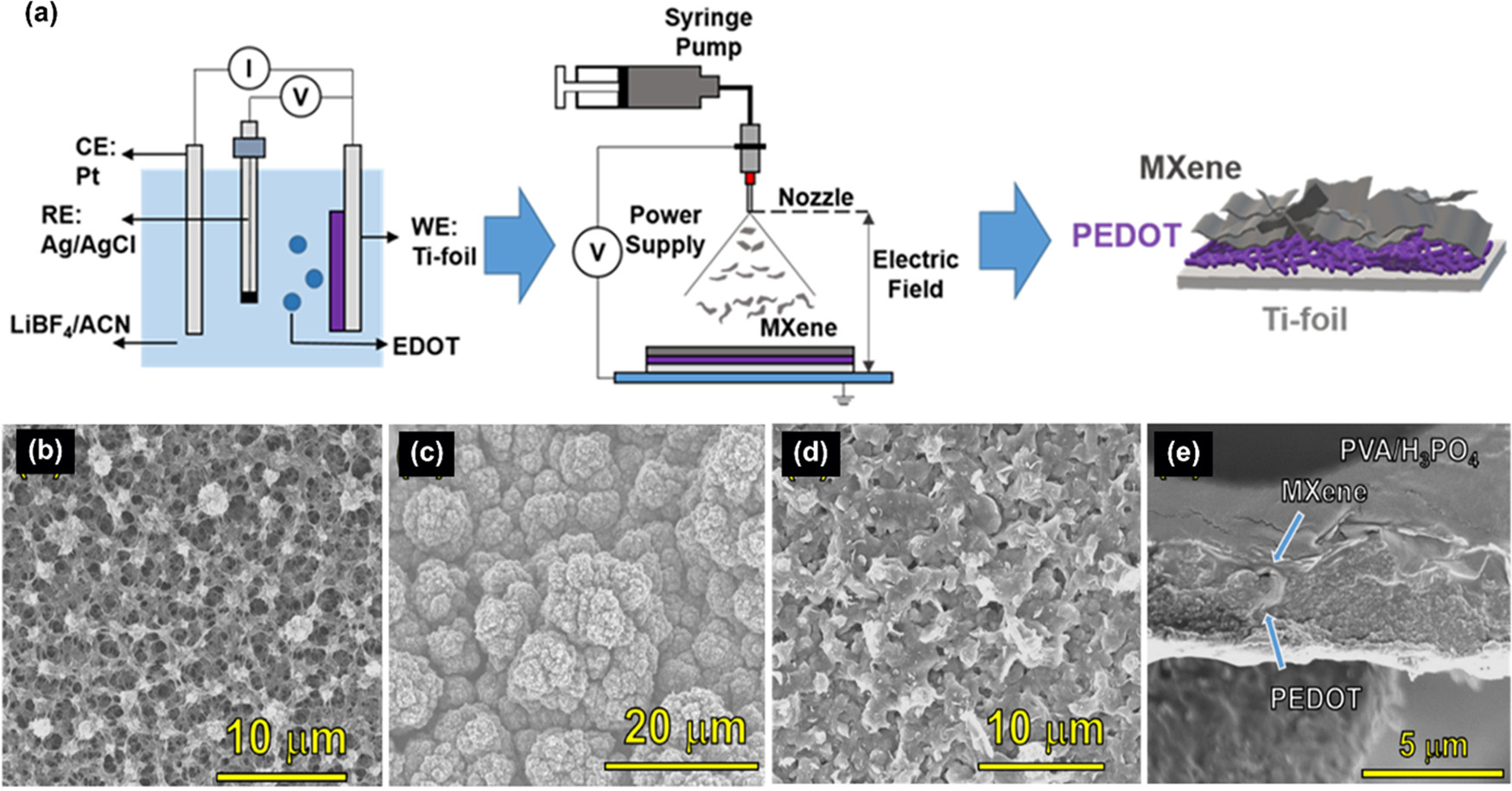
Fabrication process and morphology: (a) schematic illustration of the fabrication process for PEDOT/MXene layered composite electrodes. Morphology of PEDOT and PEDOT/MXene electrodes: scanning electron microscopy (SEM) images from the surface of (b) PEDOT-30, (c) PEDOT-60, and (d) PEDOT-60/MX. (e) Cross-sectional image of PEDOT-90/MX. (See Figure S1 for SEM and TEM images of MXen only and the SEM image of PEDOT90, and Figure S2 for the MXene structure identification).
MXene: A Novel Electrode Material for Supercapacitors
MXene, a revolutionary two-dimensional (2D) substance, has captivated scientists and engineers working in areas such as energy storage systems, supercapacitors, and adaptable batteries.
The use of pure MXene and its composites in different classes of supercapacitors is expanding dramatically due to its exceptional mechanical, physical, photonic, electromagnetic, and electrolytic capabilities.
The assembly of hybrid MXene materials results in a mixture of functional qualities that cannot be produced from MXene alone. Multiple kinds of MXene-based composites can be created by mixing electroactive components such as metallic compounds and conducting polymers (CPs) with MXene.
A synergistic effect is formed as a result, where MXene provides exceptional electrical conductivity, expanding the accessible surface area and stabilizing the material's internal structure. In addition, these materials serve as spacers and enhance the interlayer gap, thus inhibiting the restacking of MXene nanosheets.
Highlights and Key Developments of the Current Study
In this study, solid-state supercapacitors (SSCs) were manufactured by the electrochemical crosslinking of PEDOT and consequent electrospray application of MXene onto the PEDOT film. The fractal properties of the electropolymerized PEDOT and the compacted MXene flakes combined to generate a highly porous accessible structure.
This porous shape allowed for the filling of the gel electrolyte without void formation, resulting in a large electrocatalytic reactive surface and efficient ion transport pathways for increased electrocatalytic activity.
PEDOT demonstrated significant conductivity owing to total contact with the gel electrolyte despite the absence of polystyrene sulfonate (PSS) as an activator, which permeated deep into the porous surface of PEDOT to provide high conductance.
The device's electrolytic capacitance was increased even more by introducing salt to the gel electrolyte. The correct quantity of salt considerably increased the electrolyte's ionic conductance, resulting in remarkably high capacitance.
These results show that the as-prepared device surpasses most aqueous electrolyte supercapacitors. Moreover, the PEDOT/MXene composite electrode manufacturing technique is straightforward, affordable, and readily adaptable to the practical manufacture of large-scale flexible solid-state supercapacitors.
Reference
Cho, S., et al. (2022). Flexible Solid Supercapacitors of Novel Nanostructured Electrodes Outperform Most Supercapacitors. ACS Omega. Available at: https://doi.org/10.1021/acsomega.2c04822
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.