|
Since its inception, Strem Chemicals has focused on offering unique organometallic compounds for both academic and industrial research purposes. Close relationships with leading researchers in the field have enabled Strem to stay abreast of the latest scientific advances in and regularly add novel chemicals to our product portfolio. Most recently, Strem has embraced the emerging area of nanotechnology and formed a collaboration with the Max-Planck-Institut fuer Kohlenforschung. A series of including metal nanoclusters, metal nanocolloids (organosols and hydrosols), metal nanopowders, metal nanoparticles, and magnetic fluids are now available from Strem. Below is a discussion of potential applications of these nanomaterials in the energy market.
Fuel Cells and Hydrogen Storage
As the world’s fossil fuel resources dwindle, practical implementation of alternative energy systems is becoming increasingly important. Fuel cells will most likely be the energy source of the future, for vehicles, local off-grid power supply, and power sources for small and handheld electronic devices. Nanomaterials will be important as catalysts and key components of hydrogen storage systems.
|
|
|
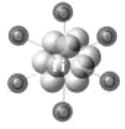
Figure 1. Diagram showing a cluster of 13 Titanium atoms and 6 THF-O atoms.
|
How Nanomaterials Are Used in Fuel Cells and Energy Storage
Both supported and unsupported colloidal nanomaterial precursors have been used to prepare electrocatalysts for fuel cells. Finely particulate colloidal platinum sols are ideal precursors for the manufacture of fuel cell electrodes.
Nanostructured metal colloids are also very promising precursors for manufacturing multi-metallic fuel cell catalysts that are truly nanosize (< 3 nm) and have high metal loadings (30 wt% of metal). The nanocolloid catalysts are typically based on bi- or tri-metallic particles and offer improved efficiency and tolerance towards contaminants. They are under investigation as catalysts in low-temperature proton-exchange membrane fuel cells (PEMFCs) and direct methanol fuel cells (DMFCs).
Using Metal Nanoclusters in Hydrogen Storage Applications and as Catalytic Centers for Carbon Nanotube Growth
For hydrogen storage applications, metal nanoclusters appear to have many potential applications. The Ti13-THF nanocluster has been found to be one of the best catalysts available for reversible hydrogen storage in alanates. Metal nanoclusters have also been utilized as catalytic centers for the growth of single walled carbon nanotubes, which are being investigated as hydrogen storage devices.
Potential Applications for Cobalt Nanoparticles and Graphite Carbon-Shells
Core-shell cobalt nanoparticles with tailored chemical compositions were prepared to protect the nanoparticles against corrosive environments. The nanoparticles can easily be removed, leaving a graphite carbon-shell. The low density shell possesses excellent permeability and electron conductivity properties and has potential application in chemical storage, including, for example, hydrogen.
Using Precious Metal Nanoparticles as Electrocatalysts in Fuel Cells and Using Nanowires as Hydrogen Sensors
Precious metal nanoparticles of various compositions have been optimized to act as effective electrocatalysts in polymer electrolyte fuel cells and direct methanol fuel cells, both at the anode and the cathode side. Electrically conducting nanowires created through deposition of atomic clusters manufactured by existing semiconductor processes are currently being used as hydrogen sensors for power distribution.
Solar Energy Systems
In addition to research on fuel cells, development of other practical alternative energy sources continues. Nanomaterials may prove vital for improving the efficiency of solar energy systems.
|
|
|
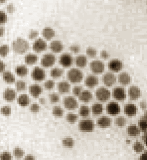
Figure 2. TEM of a nanostructured metal colloid.
|
Using Semiconductor Nanocrystals and Quantum Dots in Solar Energy Applications
High performance semiconductor nanocrystals active throughout the visible spectrum and into the near-infrared have been combined with conductive polymers to create ultra high performance solar cells that exceed the capabilities of current technology. Because the nanocrystals harvest a greater portion of the energy spectrum, these plastic solar cells have significantly improved efficiencies. Quantum dots for solar roofing tiles that are based on metal nanoparticles are expected to be commercialized within the next several years.
Why Nanoparticles Are Such Effective Materials for Conducting Heat Transfer
Nanoparticles when added to heat transfer fluids increase their performance. The solid nanoparticles conduct heat better than the liquid. Nanoparticles work best because they stay suspended in liquids longer than larger particles. They also have a much greater surface area, which is where heat transfer takes place. The smaller the particle, the greater its ability to enhance heat transfer. Nano-additives, including nanoparticles and nanopowders, could be utilized to enhance heat transfer from solar collectors to storage tanks.
Improving Current Technologies
Batteries
Nanocrystalline nickel and metal hydrides have been investigated for use in batteries. The nanostructured materials offer improvements regarding power density and durability by controlling the charge diffusion and oxidation state on a nanoscale level.
Coal Liquefaction
Metal nanocluster catalysts have been developed using an inverse micelle technique. They have been applied in both dispersed and supported forms for coal liquefaction and fossil fuel-plastics co-processing.
Transformers
The ever growing demand for greater electricity production will require upgrades of most transformers at some point in the near future at a potential cost of millions of dollars in hardware retrofits. If the heat transfer capability of existing transformers can be increased, then many of the upgrades may not be necessary. Magnetic fluids, which have significant heat transfer capabilities when exposed to magnetic fields, are being investigated as substitutes for existing coolants in transformers for electrical supply. Minimal modifications would be required before these fluids could be used.
List of Nanomaterials that Strem Can Supply to All Types of Industry
A listing of specific including metal nanoclusters, metal nanocolloids (organosols and hydrosols), metal nanopowders, metal nanoparticles, and magnetic fluids offered by Strem is available upon request or via our website. Application sheets discussing the potential use of these products in the medical and pharmaceutical, defense and security, chemical, automotive, and energy fields, and as magnetic fluids, can also be obtained from Strem. More information is also available in the form of a reference sheet listing literature source materials.
|