May 4 2009
The highly porous MOFs usually reveal two different types of structural elements. Transverse bars comprised of organic molecules are connected by inorganic coupling units containing metal ions. Mixing these reactants and heating leads to self-organized formation of MOFs. These metal-organic frameworks are of global interest because they can be loaded with the most diverse of materials.
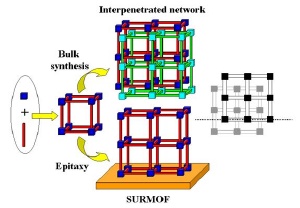 Principle of SURMOF synthesis. Using conventional synthesis, often two equivalent networks (colored red and green) are formed at the same time. Using liquid phase epitaxy the equivalence of these two networks is lifted by the presence of the organic template (dotted line on the right) and the formation of interpenetrating networks is suppressed, yielding SURMOFs containing only one network and much larger pores.
Principle of SURMOF synthesis. Using conventional synthesis, often two equivalent networks (colored red and green) are formed at the same time. Using liquid phase epitaxy the equivalence of these two networks is lifted by the presence of the organic template (dotted line on the right) and the formation of interpenetrating networks is suppressed, yielding SURMOFs containing only one network and much larger pores.
Prof. Wöll explained that the spectrum ranges from the storage of liquid hydrogen in the tank of a car all the way to a storage site for drugs. These “porous” materials are also of interest for heterogeneous catalysis. For this purpose, metal particles are embedded in the pores - this does, however, necessitate relatively large void spaces. Prof. Fischer pointed out that this has been a fundamental problem in the synthesis of MOFs to date. If the pores are too large, numerous instances of the MOF lattice develop simultaneously, forming an interlaced network of numerous structures. This in turn leads to reduction in the size of the individual voids within the frameworks.
Layer-by-layer production of larger frameworks
The scientists at the Departments of Physical Chemistry (Wöll) and Inorganic Chemistry (Fischer) at the Ruhr-University in Bonn (RUB) can now bypass this interpenetration problem. They developed a new synthesis procedure, described as liquid phase epitaxy, which differs from the usual synthesis (i.e. mixing all the reactants in solution and subsequent heating thereof). Intelligent surfaces coated with substrates are alternately dipped into pots each of which contains only one type of MOF structural element. The organic surfaces ensure that only one structural alternative of the MOF develops, thus avoiding interpenetration and yielding the desired large void spaces. Prof. Wöll is pleased to announce that it is thus now possible to produce materials with significantly larger pores than had been the case to date. Currently the scientists are trying to store metal clusters in the spacious voids. These in turn could be used for heterogeneous catalysis and sensorics.
Intelligent surfaces
The scientists produce the intelligent surfaces that ensure that exactly the desired MOFs develop by self-assembly: simply dipping metal substrates into solutions of so-called organothiols (sulphurous organic molecules) yields a high-quality organic coating. The sulphur atoms bind tightly to the metal substrate, thus acting as anchors for the organic molecules, yielding self-assembled monolayers (SAMs). The growth of the frameworks on the surface of the SAMs can then be controlled by the particular choice of the organothiol. It is even possible to “dictate” their orientation by using “tailor-made” SAMs