Jul 29 2016
Building upon their previous research, engineering faculty at the University of Pittsburgh Swanson School of Engineering and Carnegie Mellon University College of Engineering were awarded grants from the National Science Foundation to develop a novel computational framework that can custom design nanoparticles.
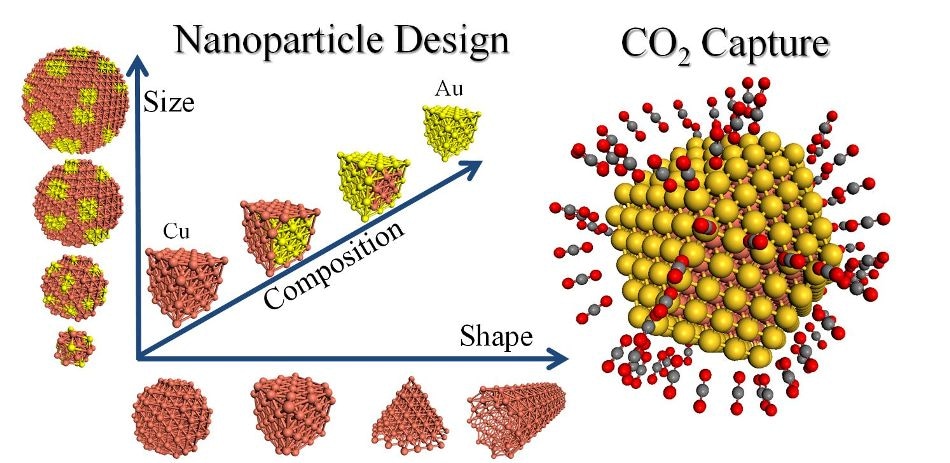 Capturing carbon dioxide via custom-designed bimetallic nanoparticles.(Credit: Mpourmpakis C.A.N.E.LA. Lab Group)
Capturing carbon dioxide via custom-designed bimetallic nanoparticles.(Credit: Mpourmpakis C.A.N.E.LA. Lab Group)
In particular, the group is investigating bimetallic nanoparticles to more effectively control their adsorption properties for capturing carbon dioxide from the atmosphere.
The three-year grant, "Collaborative Research: Design of Optimal Bimetallic Nanoparticles," is led by Giannis Mpourmpakis, assistant professor of chemical and petroleum engineering at Pitt and group leader of the Computer-Aided Nano Energy Lab (C.A.N.E.LA.). Co-investigators are Götz Veser, professor of chemical and petroleum engineering at Pitt and associate director of the University's Center for Energy; and Chrysanthos Gounaris, assistant professor of chemical engineering at Carnegie Mellon University. The NSF Division of Civil, Mechanical and Manufacturing Innovation (CMMI) awarded $350,395 to Pitt and $199,605 to CMU to support computational research and targeted experiments.
"Bulk metals behave differently than their related nanoparticles, and our research has shown that bimetallic nanoparticles exhibit unique adsorption properties," Dr. Mpourmpakis explained. "Our previous research focused on the size and shape of gold nanoparticles toward their catalytic behavior, and now we are investigating copper nanoparticles and their ability to adsorb and activate carbon dioxide."
The researchers will utilize Pitt's Center for Simulation and Modeling to computationally identify bimetallic nanoparticles that maximize their performance for a given application. By optimizing the shape, size and metal composition of bimetallic nanoparticles through computer simulation, the researchers can reduce the need for expensive and time-consuming experiments in the lab, which are often based on extensive trial and error.
"Because we know that copper-based bimetallics effectively adsorb CO2, we can now fine-tune the nanoparticle morphology to maximize adsorption," Dr. Mpourmpakis said. "The benefit to the environment of being able to capture CO2 and potentially convert it to a useful chemical would be profound."