Nov 8 2016
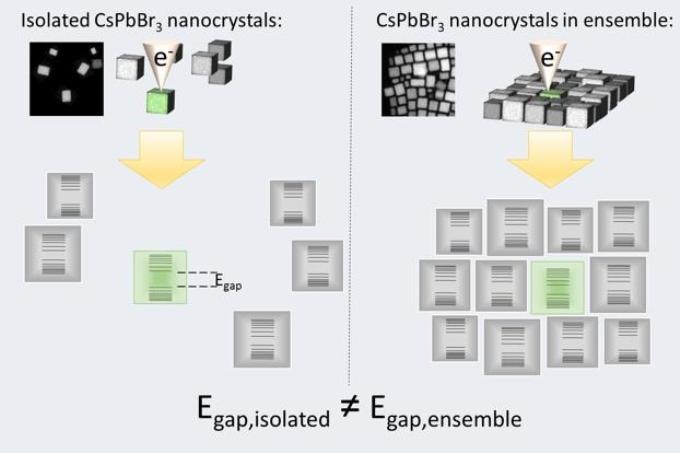 (Left) A completely isolated NC is studied using an electron beam (EELS), to determine its bandgap energy. (Right) A NC of the same size surrounded by neighbors is similarly probed, for which the measured bandgap energy is different. Hence, there must be a coupling between adjacent NCs in order for them to 'average' their bandgap energies. CREDIT: University of Amsterdam, National Institute of Advanced Industrial Science and Technology and Osaka University
(Left) A completely isolated NC is studied using an electron beam (EELS), to determine its bandgap energy. (Right) A NC of the same size surrounded by neighbors is similarly probed, for which the measured bandgap energy is different. Hence, there must be a coupling between adjacent NCs in order for them to 'average' their bandgap energies. CREDIT: University of Amsterdam, National Institute of Advanced Industrial Science and Technology and Osaka University
The relation between bandgap energy of single cesium lead bromide nanocrystals (CsPbBr3 NCs) and their shape and size has been determined directly by a global research team headed by Tom Gregorkiewicz, professor at the University of Amsterdam and Yasufumi Fujiwara, professor at Osaka University's Graduate School of Engineering, together with his Japanese team at National Institute of Advanced Industrial Science and Technology. The researchers analyzed separate NCs either surrounded by 'neighbors' or isolated in order to explicitly visualize for the very first time band structure modification introduced by efficient coupling between semiconductor NCs upon close contact.
Nanocrystals and perovskites
NCs are very small, about thousand times smaller than the width of a strand of human hair. This small size results in the energy structure of the crystals to be significantly different from that of bulk material. The bandgap energy actually depends on the size of the NC.
Perovskites, named after the Russian mineralogist Lev Perovski, refers to a particular class of materials comprising of a crystal structure in the form ABX3. Perovskites have recently attracted increased attention because of their potential for high-efficiency and low-cost photovoltaics. In CsPbBr3 NCs, the benefits of NCs and perovskites are combined, and they are thus considered to be a promising material for a wide range of optoelectronic applications.
The experimental setup
The researchers used a modern technique called low-loss electron energy loss spectroscopy (EELS), which arises from low energy excitations i.e. valence electrons. This technique is thus an analogy to absorption spectroscopy. The researchers further used EELS together with a scanning electron transmission (STEM) microscope with ultrahigh special resolution for measuring the NC location and dimensions with uniquely high precision, in parallel. In this manner, the energy absorption is directly mapped onto separate NCs that are either completely isolated or embedded in an ensemble (they have neighbors). This results in establishing an intimate relation between the NC shape, size and energy bandgap.
Interaction and coupling between proximal nanocrystals
The team determined the energy bandgap of several nanocrystals as a function of their size and then discovered that small NCs that are isolated comprise of a greater bandgap energy when compared to a NC surrounded by neighbors and that is of the same size. A bigger NC when isolated will have lower bandgap energy than when embedded in an ensemble. This result highlights that two adjacent NCs average their bandgaps instead of simply merging upon interaction and posing as a larger crystal. This indeed provides direct evidence of an effective coupling between NCs where their energy bandgap and thus energy structure, is influenced by the neighbors. These unique insights based on the interacting behavior of neighboring NCs makes room for purposeful designing of bigger quantum structures and quantum-dot-solids, comprising of NCs with specific features serving as building blocks.