Oct 25 2017
Thermal noise and friction have a strong impact on transitions—such as folding of a protein or a chemical reaction—that take place in nanoscale systems. Nearly eight decades earlier, Hendrik Kramers, a Dutch Physicist, proposed that these transitions take place often at intermediate friction, an impact called Kramers turnover.
At present, a research team from the ETH Zurich, ICFO in Barcelona, and the University of Vienna has evaluated this effect for a laser-trapped particle, thereby providing direct experimental confirmation of Kramers’ theory for the first time. The research has been published in the journal Nature Nanotechnology.
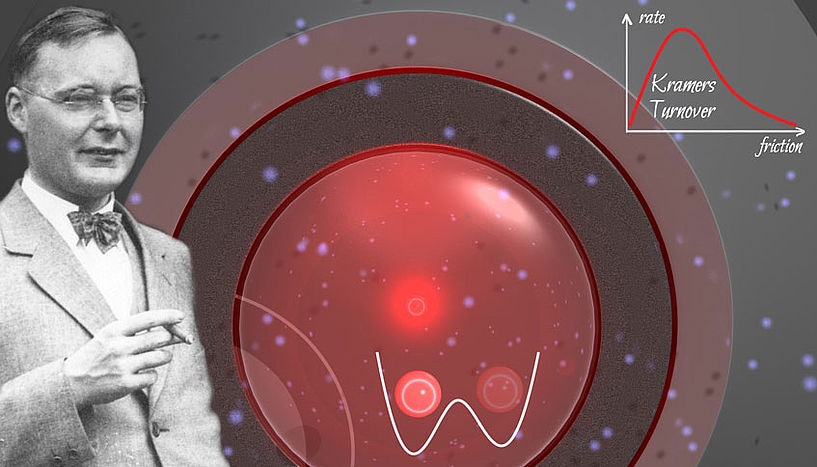 In 1940, Hendrik Kramers (left) predicted theoretically that in a double-well system (center bottom) transitions between the stable states happen most frequently at intermediate friction (upper right). The background shows a detail of the laser system used to confirm Kramers’ prediction experimentally. Copyrights: Jan Gieseler. CREDIT: Image of H. Kramers courtesy of AIP Emilio Segrè Visual Archives, Goudsmit Collection.
In 1940, Hendrik Kramers (left) predicted theoretically that in a double-well system (center bottom) transitions between the stable states happen most frequently at intermediate friction (upper right). The background shows a detail of the laser system used to confirm Kramers’ prediction experimentally. Copyrights: Jan Gieseler. CREDIT: Image of H. Kramers courtesy of AIP Emilio Segrè Visual Archives, Goudsmit Collection.
In the year 1827, Robert Brown, an English Botanist made a less significant observation that had a pivotal role in developing the atomic theory of matter. Viewing through a microscope, he observed that pollen grains that floated on water continuously wriggled around, which seemed as if they were acted upon by an invisible force—a phenomenon presently called Brownian motion.
Many years later, Scientists found out that the irregular motion of the pollen grains is the result of continual buffeting of water molecules around each pollen grain. Albert Einstein’s theoretical investigation into this occurrence offered significant evidence for the presence of atoms.
The interactions between the pollen grain and the water molecules have two significant impacts on the movement of the grain. First, they produce friction, which slows down the grain. Second, their thermal perturbation makes the grain move continuously. The balance between these two challenging forces leads to Brownian motion.
Thermal motion and friction generated by the surrounding also strongly impact transitions among long-lived states, for instance, phase transitions such as melting or freezing. The long-lived states — such as different phases of an individual chemical species or a material — are isolated by a high energy barrier. This barrier between the wells stops the physical system from quickly inter-transitioning between the two states. As a result, the system jiggles around in one of the wells for a longer period of time and seldom leaps from one well to the other. These transitions — such as the folding of proteins and phase transitions to chemical reactions — are highly significant for various processes in nature as well as technology.
Friction’s unexpected influence on transitions
So how many times do these rare barrier crossing events take place within a specific time period? Hendrik Kramers addressed the above question theoretically in the year 1940. He used a simple model system to mathematically demonstrate that the rate of occurrence of these transitions rapidly decreases when there is an increase in barrier height. Astonishingly, Kramers proposed that the transition rate is also reliant on the friction in a surprising manner. In the case of strong friction, the system moves very languidly, resulting in a small transition rate.
When the friction decreased, the system moved more readily and the transition rate increased. However, at adequately lower friction, the transition rate started to reduce again as it takes a longer time for the system to gain enough energy from the surroundings to cross the barrier. The ensuing transition rate maximum at intermediate friction is known as the Kramers turnover.
Measuring Kramers’ prediction with laser-trapped nanoparticles
As part of an international collaboration, Researchers from the ETH Zurich, ICFO in Barcelona, and the University of Vienna have been successful in directly viewing the Kramers turnover in the case of a levitated nanoparticle. During the experiment, a nanoparticle is positioned in a laser trap including an energy barrier isolating two wells. Quite similar to the pollen grain noticed by Brown, the nanoparticle continuously collided with the molecules in its environment, and the random collisions sometimes pushed the nanoparticle above the barrier.
The Researchers observed the nanoparticle motion for a longer period of time and determined the crossover rate of the nanoparticle between the wells for a broad array of frictions, which could be precisely adjusted by altering the pressure of the gas surrounding the nanoparticle. The rate observed in the experiment evidently validates the turnover proposed by Kramers nearly eight decades back.
These results improve our understanding of friction and thermal motion at the nanoscale and will be helpful in the design and construction of future nanodevices.
Christoph Dellago, one of the Authors of the research