Jan 24 2018
Cardiac arrest can be caused by severe oxygen deficiency. It is important that the blood’s oxygen content is re-established quickly, otherwise the patient may die within a matter of minutes. Therefore, in order to increase the survival rate of such patients, American scientists have developed unique air-filled microbubbles that could be utilized as an intravenous oxygen carrier. These microbubbles dissolve quickly in blood, which means the risk of embolism is greatly reduced. The study has been reported in the journal, Angewandte Chemie.
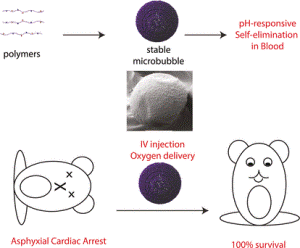 Image credit: © Wiley-VCH
Image credit: © Wiley-VCH
Whether it results from heart failure, pertussis, an asthma attack, a piece of food in the trachea or a swimming accident, asphyxial cardiac arrest accounts for about 100,000 in-hospital patients’ deaths in the United States alone.
In association with Brian D. Polizzotti and John N. Kheir at Boston Children’s Hospital and Harvard University (Boston, Massachusetts, USA), the researchers developed the stable, self-disrupting microbubbles that have the potential to save many lives because they provide physicians some amount of time to either perform a tracheotomy procedure to re-establish air flow or resolve the cause of oxygen deprivation.
The concept of utilizing microbubbles as a means to carry substances, such as contrast agents or drugs, is not a novel one. However, since these microbubbles remain in the blood for a long time, their intravenous injection invariably carries a high risk of life-threatening pulmonary embolism. Other challenges included low stability, poor control over size and morphology, and components that are impossible to break down.
However, these disadvantages are not present in the air-filled microbubbles. Their success is attributed to a unique production technique in which biocompatible polymers are nanoprecipitated at the interface between liquid and air. Dextran – a branched polymer made of glucose units – is the starting material.
Acids are an example of functional groups which are connected to give unique surface-active properties to the polymer. This polymer forms micelles when it is dissolved in an organic solvent, followed by the addition of water, in which it is insoluble. Homogenization with air produces foam-containing air bubbles that are surrounded by micelles. Upon addition of more water, increasing number of micelles come together forming solid nanoaggregates that create a shell around the air bubble.
When these stable microbubbles are introduced into the blood, the pH value of the blood causes the acid groups to form charged COO-groups. This enables water to enter the shell and eventually release the oxygen. The shells fall apart and the components dissolve fully due to the electrostatic repulsion between the charges.
In order to induce asphyxial cardiac arrest, the scientists blocked the rodents’ airways. After a gap of 10 minutes, they removed the blockage to mimic a physician re-establishing the airway. All the animals died in the control group, while all the animals in the other group lived as a result of fast and repeated injection of the microbubbles during cardiac arrest. There were no signs of a life-threatening embolism. In addition, comparison with other oxygen carriers showed that the stable, self-disrupting microbubbles were the only technique which allowed the rodents to receive large quantities of oxygen (up to 12 mL) without any complications.