Feb 28 2019
The 2018 Nobel Prize in Chemistry was awarded to three scientists who developed the approach called directed evolution, which permanently transformed protein engineering. Directed evolution imitates natural evolution and guides the production of proteins with new or enhanced functions.
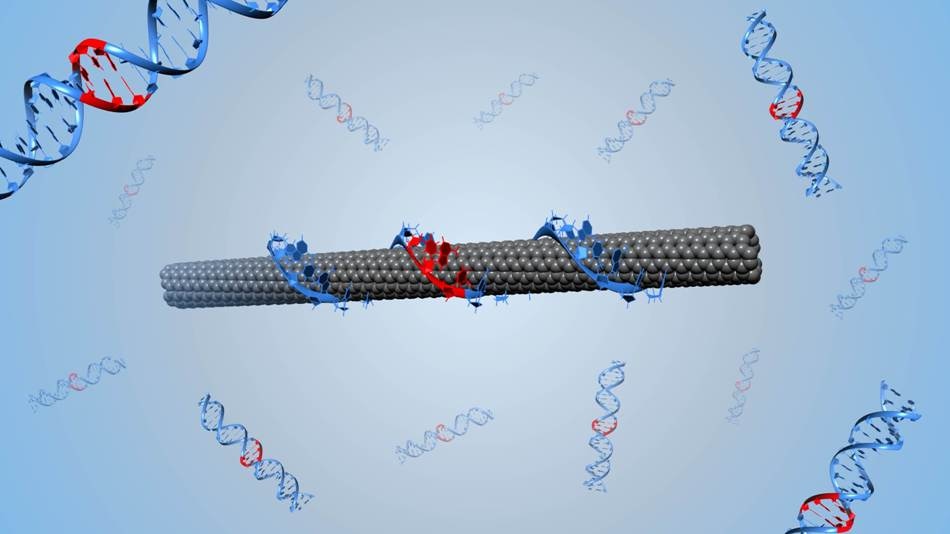 Illustration of a DNA-wrapped single-walled carbon nanotube. (Image credit: Benjamin Lambert, EPFL)
Illustration of a DNA-wrapped single-walled carbon nanotube. (Image credit: Benjamin Lambert, EPFL)
Initially, the original protein is mutated to develop a range of mutant protein variants. Then, the protein variants that exhibit enhanced or more favorable functions are chosen. These chosen proteins are then mutated again to produce another array of protein variants for another round of selection. This cycle is done again and again until a final, mutated protein is developed with superior performance when compared to the original protein.
Presently, researchers from the lab of Ardemis Boghossian at EPFL have been able to use directed evolution to develop synthetic nanoparticles and not proteins. These nanoparticles are employed as optical biosensors, which are small devices that detect biological molecules in water, blood, or air using light. Optical biosensors are extensively employed in drug development, biological research, and medical diagnostics, for example, real-time monitoring of glucose and insulin in diabetics.
The beauty of directed evolution is that we can engineer a protein without even knowing how its structure is related to its function. And we don't even have this information for the vast, vast majority of proteins.
Ardemis Boghossian, EPFL
Her team employed directed evolution to alter the optoelectronic properties of DNA-wrapped single-walled carbon nanotubes (or DNA-SWCNTs), which are nano-sized tubes of carbon atoms that look like rolled-up sheets of graphene enveloped by DNA. When they identify their target, the DNA-SWCNTs release an optical signal that can penetrate through complex biological fluids, such as blood or urine.
Common Principle of the Directed Evolution Method Applied to the Nanoparticle DNA-SWCNT Complexes
The initial complex is a DNA-SWCNT with a weak optical signal. This is advanced through directed evolution, which involves the following steps:
- Random mutation of the DNA sequence
- Covering the SWCNTs with the DNA and screening the optical signal of the complex
- Choosing the DNA-SWCNT complexes showing an enhanced optical signal
After numerous cycles of evolution, DNA-SWCNT complexes that exhibit improved optical behavior can be obtained (Credit: Benjamin Lambert, EPFL)
With the help of a directed evolution method, the team led by Boghossian was able to synthesize new DNA-SWCNTs with optical signals that are improved by up to 56%—and they achieved it with just two evolution cycles.
The majority of researchers in this field just screen large libraries of different materials in hopes of finding one with the properties they are looking for. In optical nanosensors, we try to improve properties like selectivity, brightness, and sensitivity. By applying directed evolution, we provide researchers with a guided approach to engineering these nanosensors.
Ardemis Boghossian, EPFL
The research demonstrates that a fundamental bioengineering technique can be used to more rationally modify the optoelectronic properties of particular nanomaterials. Boghossian explains: “Fields like materials science and physics are mostly preoccupied with defining material structure-function relationships, making materials that lack this information difficult to engineer. But this is a problem that nature solved billions of years ago—and, in recent decades, biologists have tackled it as well. I think our study shows that as materials scientists and physicists, we can still learn a few pragmatic lessons from biologists.”
This study was funded by an SNSF AP Energy Grant.