May 29 2019
Scientists from the Nanoscience Center (NSC) at the University of Jyväskylä, Finland, and in the Xiamen University, China, have determined how copper particles at the nanometer scale operate in altering a carbon-oxygen bond when ketone molecules are converted to alcohol molecules.
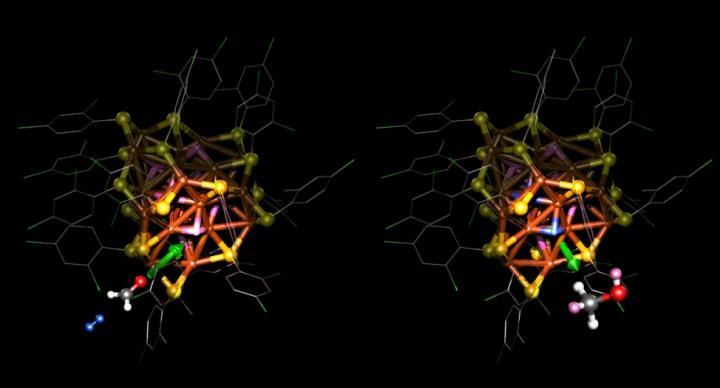 Atomic structure of the copper catalyst used in the hydrogenation reaction of a carbon-oxygen bond. Formaldehyde H2CO (on the left), used as the model molecule in the simulations, takes two hydrogens from the copper; they move to the carbon-oxygen bond so that the molecule changes into a simple alcohol (methanol CH3OH on the right). After the reaction, a hydrogen molecule (blue, on the left) in the vicinity splits into two hydrogen atoms inside the copper. (Image credit: Sami Malola, University of Jyväskylä)
Atomic structure of the copper catalyst used in the hydrogenation reaction of a carbon-oxygen bond. Formaldehyde H2CO (on the left), used as the model molecule in the simulations, takes two hydrogens from the copper; they move to the carbon-oxygen bond so that the molecule changes into a simple alcohol (methanol CH3OH on the right). After the reaction, a hydrogen molecule (blue, on the left) in the vicinity splits into two hydrogen atoms inside the copper. (Image credit: Sami Malola, University of Jyväskylä)
Alteration of the carbon-oxygen and carbon-carbon bonds present in organic molecules is a significant intermediate phase in catalytic reactions, where the source material is converted into useful end products.
Gaining insight into the operation of catalysts at the atomic structure level of a single particle enables catalysts to be developed into preferred directions, for instance, making them efficient and selective for a particular desired end-product. The research was reported in the internationally renowned ACS Nano nanoscience publication series. In Finland, the research was headed by Academy Professor Hannu Häkkinen.
The catalytic copper particles used in the research were produced and structurally characterized at the Xiamen University, and their operation in modifying a strong carbon-oxygen bond in a hydrogenation reaction was investigated by the scientists of the Nanoscience Center (NSC) at the University of Jyväskylä in computer simulations.
The precise atomic structure of the copper particles was revealed using X-ray diffraction and nuclear magnetic resonance (NMR) spectroscopy. The particles were found to include 25 copper atoms and 10 hydrogens, and there were 18 thiols guarding the particle surface. While the experimental work in Xiamen demonstrated its outstanding performance in catalytic hydrogenation of ketones, the simulations estimated that the hydrogens bound to the copper core of the particle serve as a hydrogen storage, which liberates two hydrogen atoms to the carbon-oxygen bond at the time of a reaction.
After the reaction, the hydrogen storage is refilled when a hydrogen molecule linked to the particle from its surroundings divides into two hydrogen atoms, which are attached again to the copper core. The NMR measurements conducted in Xiamen showed an intermediate product of the reaction, which corroborated the predictions of the computational model.
This is one of the first times in the whole world when it has been possible to discover how a catalytic particle works when its structure is known this accurately, thanks to a cooperation involving both experiments and simulations.
Hannu Häkkinen, Academy Professor, University of Jyväskylä
Häkkinen also led the computational part of the study.
Traditionally, expensive platinum-based catalysts are used in hydrogenation reactions. This study proves that nanoscale copper hydride particles also act as hydrogenation catalysts. The results give hope that in the future, it will be possible to develop effective and inexpensive copper-based catalysts to transform functionalized organic molecules into products with a higher added value.
Karoliina Honkala, Häkkinen’s collaborator, Professor of Computational Catalysis, University of Jyväskylä
Apart from Häkkinen and Honkala, Postdoctoral Researcher Nisha Mammen, Doctoral Student Sami Kaappa, and Senior Researcher Sami Malola from the University of Jyväskylä took part in the research. The study carried out by the teams of Häkkinen and Honkala was supported by the Academy of Finland. The computer simulations in the research were performed by the supercomputers of the CSC - IT Center for Science. The experimental work of the research was conducted by the team led by Professor Nanfeng Zheng from Xiamen University.