The sponge was made in a single-step process, which offers the benefits of ease of use and increased robustness. The research optimized the effect of several coupling agents and coupling durations on the hydrophobicity of the surface of the sponge.
Oil-Water Separation
As a result of rapid economic expansion, the rise of the petrochemical industry, and the advancement of maritime transport, marine oil spills have become a common and serious environmental hazard.
As a result of being aggregated in fish and seafood, the spilled oils will ultimately be ingested by people, presenting a significant hazard to human health and aquatic ecosystems. Excellent oil-water separation (OWS) is therefore critical.
On-site burning, surface skimming, dispersing agents, and absorption have all been employed to clean up oil spills. The absorption technique is commonly employed for OWS due to its ease of use and good separation.
Conventional absorption elements, including cellulose, zeolites, and activated carbon, are largely microporous. As a consequence, these materials exhibit relatively poor adsorption capability, resulting in an inferior OWS effectiveness.
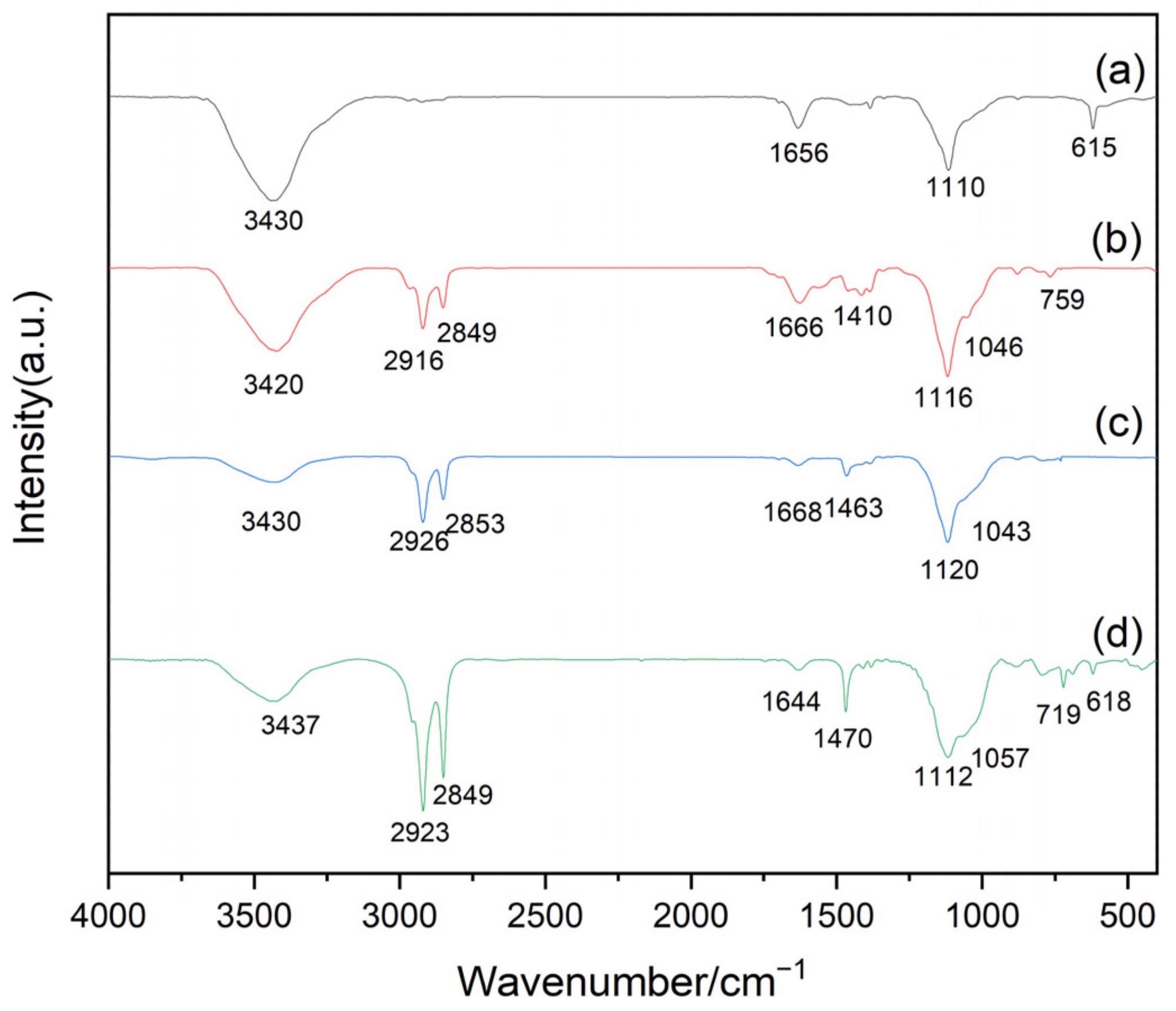
Fourier transform infrared (FT-IR) spectrophotometry of: (a) pre-treated polyurethane (PU) sponge, (b) octadecyldimethyltrimethoxy-carbon nanotubes (CNTs)/PU sponge, (c) hexadecyltrimethoxylsilane-CNTs/PU sponge, and (d) octadecyltrichlorosilane (OTS)-CNTs/PU sponge. Image Credit: Liu, D., Wang, S., Wu, T., & Li, Y.
Studies have sought to develop low-cost and efficient OWS materials.
Substantial pore size and a high specific area are the attractive features of three-dimensional adsorbents, which can achieve OWS by capturing oil on the material's surface. Furthermore, not only are they affordable, but they also exhibit a high capacity for absorption.
Unfortunately, since the surface wettability of three-dimensional adsorbents is generally hydrophilic, they have an inadequate water-repellant oleophilic capability. Their oleophilic performance and OWS efficiency, in particular, are often sub-par.
What are Ultra-Hydrophobic Materials?
An ultra-hydrophobic substance has a water contact angle larger than 150 degrees. Ultra-hydrophobic substances may be created by enhancing the hardness of the specimen surface while decreasing the surface free energy.
Due to their ability of selective oil absorption, 3D materials with high porosity and ultra-hydrophobic/ultra-lipophilic characteristics have gained a lot of interest. Still, surfaces of the sponges contain carboxyl and hydroxyl groups, which are normally hydrophiles; therefore, modifying the sponge surface is necessary.
To improve sponges, nanomaterials such as SiO2 or Fe3O4 nanoparticles, reduced graphene oxide, and nano-diamonds have been employed.
Due to their superior electric, mechanical, and optical capabilities, multi-walled carbon nanotubes (MWCNTs) are employed in a variety of use-cases. Furthermore, because of their remarkable chemical stability in the environment, large aspect ratio, and small density, MWCNTs can be utilized as adsorbents.
Ultra-hydrophobic sponges can perform swift and selective absorption of oils and carbon-based solvents with varying density and viscosity while remaining completely water-repellant.
Given that oil and carbon-based solvent spills often take place in diverse environmental circumstances, it is critical to understand the behavior of ultra-hydrophobic sponges under a variety of settings. Variations in environmental conditions may impact the oleophilic efficiency of ultra-hydrophobic sponges.
Temperature, for example, may impact the oil's viscosity, while ionic strength may alter the perceived wettability of the ultra-hydrophobic sponges, hence influencing the ultra-hydrophobic sponges' oleophilic efficiency.

Low magnification (A) and high magnification (B) Scanning electron microscopy (SEM) images of (a) pretreated sponge, (b) octadecyldimethyltrimethoxysilylpropylammonium chloride-CNTs/PU, (c) hexadecyltrimethoxylsilane-CNTs/PU, and (d) OTS-CNTs/PU with coupling time = 24 h. Image Credit: Liu, D., Wang, S., Wu, T., & Li, Y.
Key Findings of the Research
Several coupling agents and coupling durations were used in this study to develop ultra-hydrophobic sponges. The addition of long-chain alkyl groups renders the sponge ultra-hydrophobic, increasing the water contact angle up to 151 degrees.
The ultra-hydrophobic sponge is made in a single-step process, allowing the optimal coupling agent – Octadecyltrichlorosilane (OTS) - to engage with the hydroxyl groups on carbon nanotubes, but also with hydroxyl groups on sponges with preliminary treatment, increasing the durability of the created sponges.
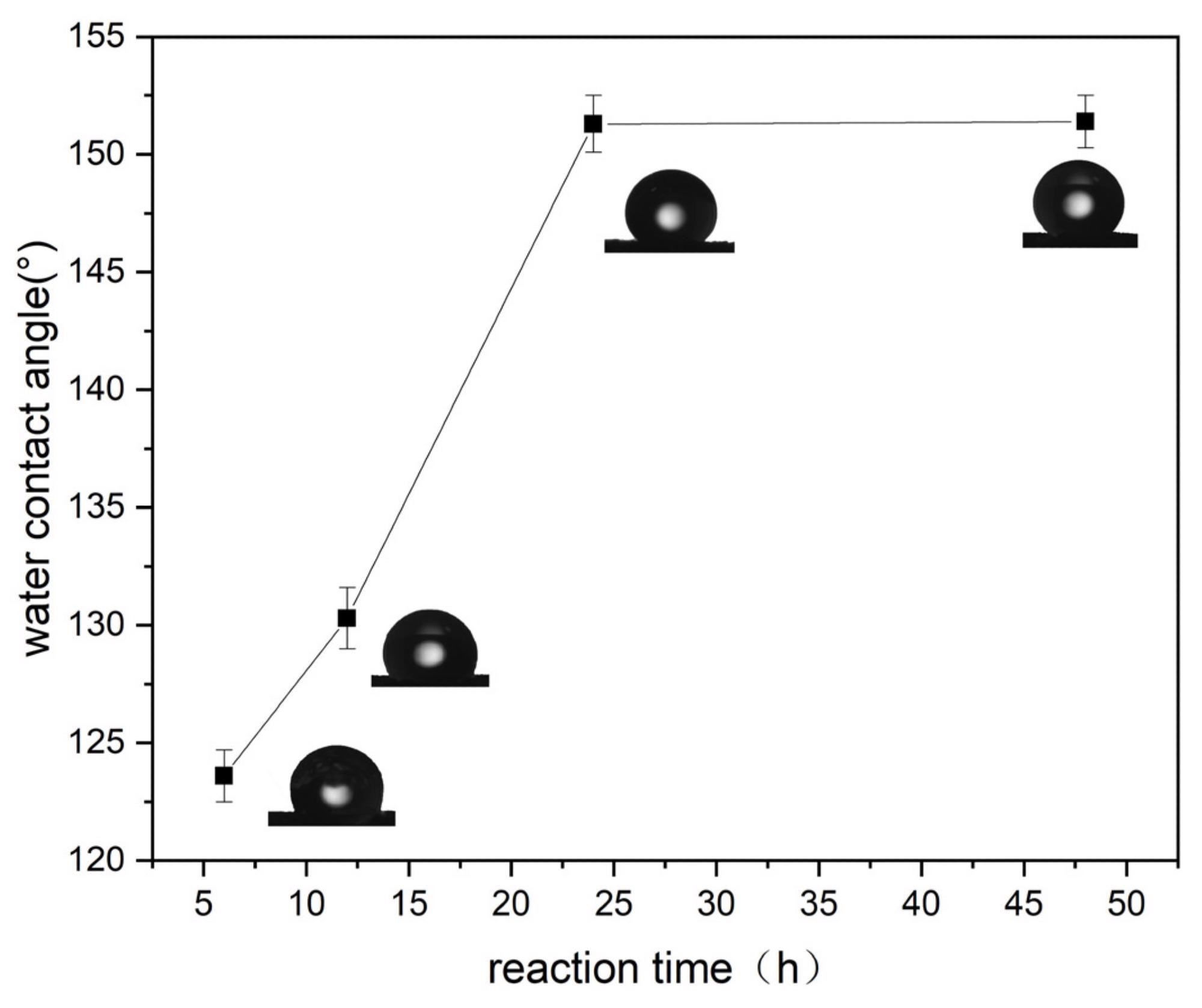
Effect of coupling time on the WCA (experimental conditions: pH = 7, coupling time = 24 h at 25 °C). Image Credit: Liu, D., Wang, S., Wu, T., & Li, Y.
For analyzing the absorbing capacity, six oils with varied densities and viscosities were chosen as sample pollutants.
The ultra-hydrophobic sponge prepared by the team has exceptional mechanical qualities as well as a high absorption potential, capable of soaking up oils 15 to 86 times its own weight. Furthermore, it can be repeatedly utilized up to 10 times by squeezing, without any major depreciation in its absorption potential.
Temperature and ionic strength were also examined, and it was discovered that oil absorption by the ultra-hydrophobic sponge is dependent on capillary and hydrophobic forces. As a result, the OTS-CNTs/PU sponge was found to be a promising option for oil spill absorption.
Continue reading: Replenishing Water Sources with Electrospun Nanofiber Membranes.
Reference
Liu, D., Wang, S., Wu, T., & Li, Y. (2021) A Robust Superhydrophobic Polyurethane Sponge Loaded with Multi-Walled Carbon Nanotubes for Efficient and Selective Oil-Water Separation. Nanomaterials, 11(12). Available at: https://www.mdpi.com/2079-4991/11/12/3344
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.