Thermostable and pH Stable Cellulases
Even at sub-optimal operating temperature and pH, cellulose materials demonstrate high stability. In the biomass industry, these temperature and pH-stable enzymes are in great demand. As fungal strains have a higher cellulolytic index, they are usually favored to generate cellulose enzymes over bacterial strains.
Nanomaterials, which operate as catalysts due to their exceptional physicochemical features, such as high surface reactivity and great adsorption capacity, have been extensively investigated for enhancing the thermal and pH stability of enzymes.
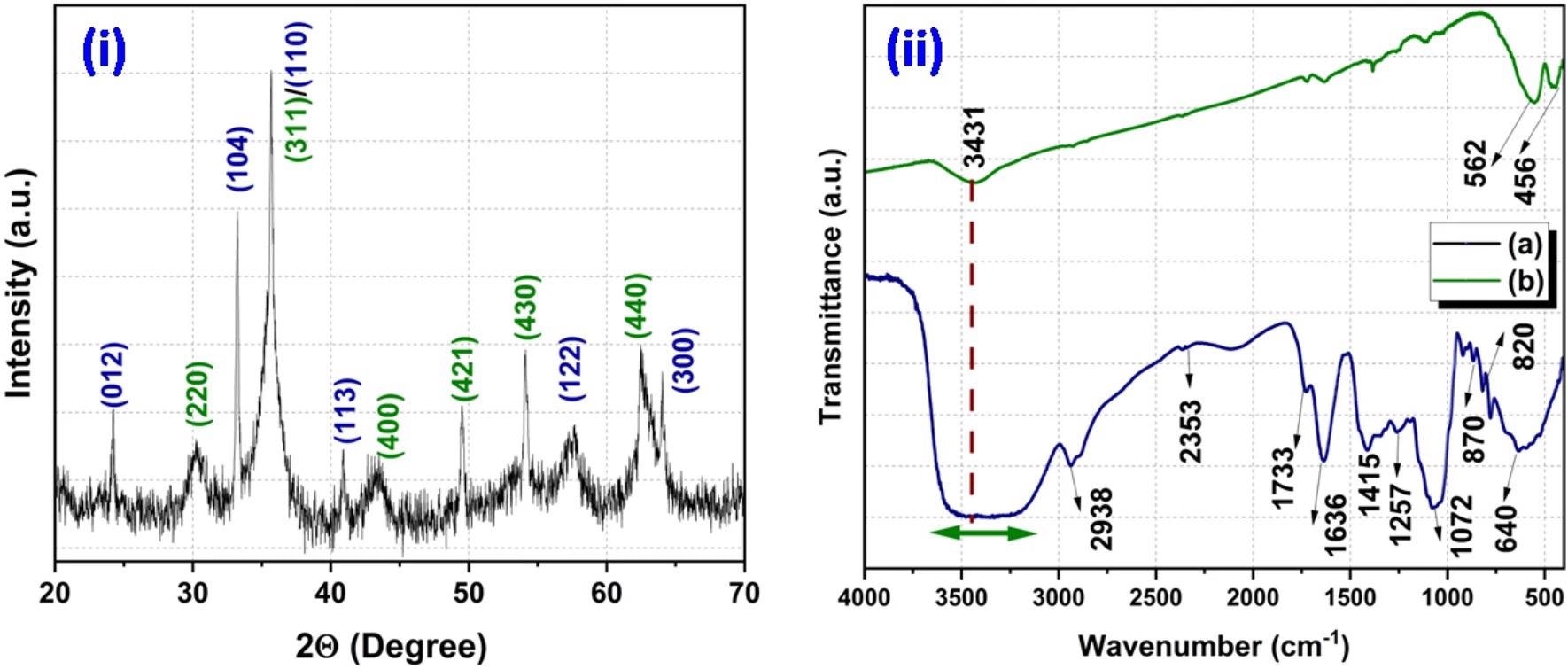
XRD pattern of synthesized product (i) and FT-IR spectra of waste pulp extract of Syzygium cumini (a) and Fe3O4/α-F2O3 nanocomposite (b) (ii). © Srivastava, N. et al. (2021).
Synthesis of Fe3O4/α-Fe2O3 Nanocomposite
In this research, the researchers prepared the Fe3O4/α-Fe2O3 nanocomposite using a modified approach.
To generate the Fe3O4/α-Fe2O3 nanocomposite through this method, a 1 M solution of ferric nitrate (50 mL) was first produced. Thereafter, WPE and ferric nitrate mixture were combined in a 2:1 ratio until the solution became brownish.
The solution was centrifuged to obtain the precipitate after being incubated for 10 minutes for settling down. To achieve the final product, the precipitate was thoroughly washed with Distilled water before being calcined at 300 °C for 10 minutes in a prepared furnace.
Effect of Fe3O4/α-Fe2O3 Nanocomposite on Thermal Stability of Enzyme
Temperature is a critical characteristic that has a significant impact on cellulase synthesis and activity. In the presence of Fe3O4/α-Fe2O3 nanocomposite, the effect of different incubation temperatures on cellulolytic enzyme activity was examined (NCs).
The Fe3O4/α-Fe2O3 nanocomposite treated enzyme had optimal activity at temperatures between 50 and 60 °C, according to their findings. Furthermore, the enzyme retains 100 % pure enzyme reactions for one hour at this temperature range, while the reference could only maintain its optimal activity at 50 °C.
At 75 °C and 95 °C, the enzyme maintains a half-life of up to 2.5 hours and 1 hour, respectively. The high temperature which degrades the enzyme configuration might explain a significant decline in enzyme half-life over 70 °C.

FE-SEM micrograph of Fe3O4/α-F2O3 nanocomposite at two different magnifications (a, b), selected micrograph for the elemental mapping (c), elemental mapping for the iron (d), oxygen (e) and the overlapping of the iron and oxygen element (f). © Srivastava, N. et al. (2021).
Effects of Fe3O4/α-Fe2O3 Nanocomposite on pH Stability
Researchers tested the impact of Fe3O4/α-Fe2O3 on enzyme reactivity at different pH levels to determine its stability. The findings demonstrate that in an acidic environment (pH 3.5–6.0), the Fe3O4/α-Fe2O3 treated enzyme has 100 % activity, whereas its half-life is at pH 8.0. It is worth mentioning that the reference enzyme (untreated with nanocomposite) only showed 100 % relative activity with a half-life of 7.5 in the pH range of 4.0–4.5.
Properties of Synthesized Fe3O4/α-Fe2O3 NCs
The UV-Visible spectrograph obtained in the region of 250–650 nm was used to evaluate the optical properties of produced Fe3O4/α-Fe2O3 NCs. The UV–Vis spectra had a peak around 350 nm. In addition, the optical band gap was computed using the Tauc plot, and was determined to be ~2.72 eV using the Tauc plot. The result was in excellent accord with previous research.
Researchers used VSM measurements to investigate the magnetic characteristics of the Fe3O4/α-Fe2O3 nanocomposite. The presence of an M-H loop indicates that the produced sample is superparamagnetic. This feature determined the existence of nanoparticles with single magnetic domains that are exceedingly tiny in size.
The Fe3O4 phase was primarily responsible for the Fe3O4/α-Fe2O3 nanocomposite's superparamagnetic characteristic since the α-Fe2O3 phase exhibits weak magnetic capabilities at ambient temperature.
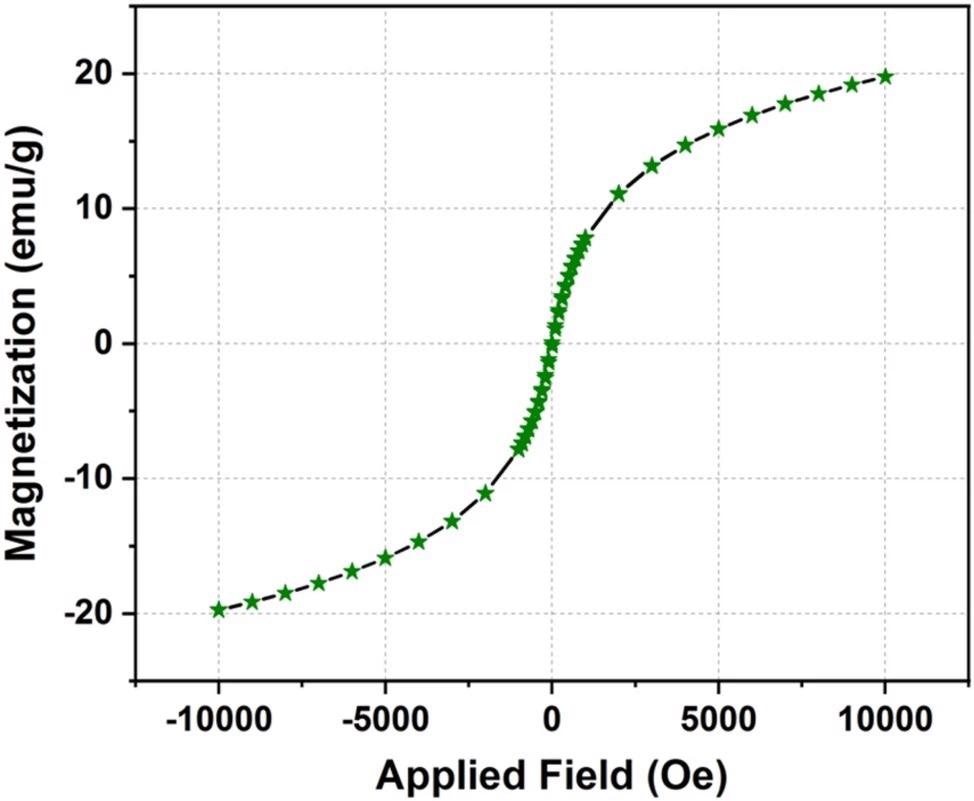
M-H graph of Fe3O4/α-F2O3 nanocomposite measured with an applied magnetic field of 10 KOe. © Srivastava, N. et al. (2021).
Because of its excellent immobilization and photocatalytic activity, the Fe3O4/α-Fe2O3 nanocomposite was predicted to operate as a barrier, enhancing enzyme stability for a longer time at moderate temperatures. Cellulose immobilized on Fe3O4 NPs was shown to be stable at 65 °C for up to 5 hours.
Conclusion and Prospects
In this study, the researchers described the synthesis of Fe3O4/α-Fe2O3 nanocomposite from the waste pulp of Jamun extract. Crude cellulase treated with Fe3O4/α-Fe2O3 nanocomposite demonstrated optimal reaction temperature and thermal stability in the extended temperature range of 50–60 degrees Celsius for 15 hours, as well as pH stability in the range of 3.5–6.0.
Furthermore, the researchers concluded that the enzyme's efficiency might be increased even further by using surface functionalizations of nanoparticles to form covalent bonds.
The efficiency of the enzyme system may be substantially enhanced in this manner, which has a lot of promise for industrial applications.
Continue reading: Lipid Nanoparticle Drug Delivery System Created from Grapefruits.
Reference
Srivastava, N. et al. (2021). Sustainable green approach to synthesize Fe3O4/α-Fe2O3 nanocomposite using waste pulp of Syzygium cumini and its application in functional stability of microbial cellulases. Scientific Reports, 11, 24371. Available at: https://www.nature.com/articles/s41598-021-03776-w
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.