Scientists have explored and produced novel superhydrophobic films fascinated by naturally water-resistant substances such as lotus leaves. Generally, a material with water contacting angle higher than 150 degrees is considered superhydrophobic.
To prevent hydration, a material's surface tension must be less than that of the wetting fluid, as per Young's model. Furthermore, surface abrasion influences contacting angle and wettability, as indicated by the Wenzel and Cassie-Baxter models. As a result, not just the chemical composition of the surface, but also the substance's surface topological structure are critical in achieving superhydrophobic behavior.
Advantages of Incorporating Magnetic Features into Superhydrophobic Materials
The incorporation of magnetic characteristics into superhydrophobic substances, or vice versa, allows for distant operation of the substance while resisting water, opening up new possibilities for promising uses.
Magnetic phase-reinforced nanoscale composites have been created in research thus far. Magnetically reactive foams, for example, have been claimed to extract organic pollutants from water after being made by adding magnetic nanoparticles (NPs) into a bulk polymeric matrix.
Furthermore, a magnet elastomer with a superhydrophobic coating has been designed to enable particle mobility and switching off their dynamic wetting properties.
Nanoscale Superhydrophobic Particles with Magnetic Properties
Magnetized superhydrophobic nanoparticles/microparticles (NPs/MPs) have piqued the interest of scientists due to their ease of control, minimal remanence, and nanoscopic applications. In this case, raw superparamagnetic Fe3O4/-Fe2O3 nanoparticles and ferromagnetic iron particles complexed with substances that create low surface tensions are directly used, without the requirement for a bulk ceramic or polymeric framework.
Magnetic superhydrophobic nanoparticles have been employed in this method to create magnetized liquid-state marbles, which are non-sticking particles covered with minimal surface tension magnetic NPs/MPs that exhibit exceptionally little traction during sliding or rolling motions on solid surfaces.
Tiny liquid-phase particles were controlled by a magnetic field (MF) because of the magnetic characteristics of liquid marbles. Furthermore, by adjusting the strength of the MF, the casing of the liquid marbles may be reversibly closed and opened, allowing liquids to be removed and inserted.
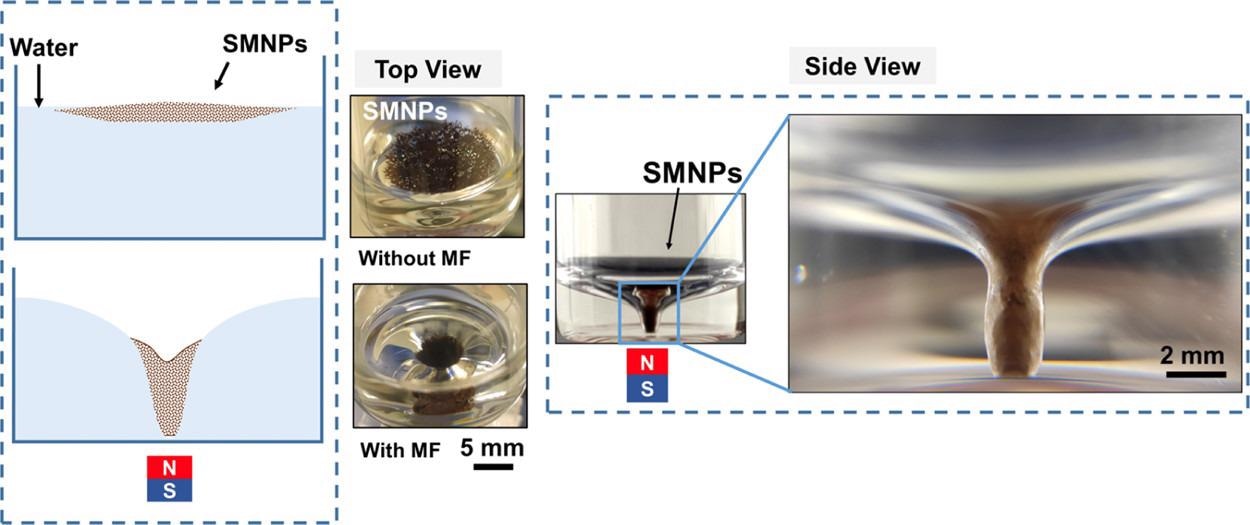
Deformation of the SMNPs layer on the water–air interface, under a MF of 42 mT. From left to right: schematic diagram of the formation of the twister, pictures of the top and side view of the magneto twister. © Gunatilake, U. B., Morales, R., Basabe-Desmonts, L., & Benito-Lopez, F. (2022)
Recent Developments in the Field
These magnetized beads have lately been employed for fluid transport, tiny microscale reactors, magneto-thermal furnaces, and digitized microfluid devices, among many other things.
It is worth noting that superhydrophobic magnetized nanoparticles have been used to tidy up oil spillage or organic pollutants on bodies of water, in which the superhydrophobic droplets formed a colloidal dispersion with the oil spillage but not with the water due to the non-polar attractive forces among the oil and the outer layer of the droplets.
As a result, using a magnetic field, such magnetic colloids were readily removed from water bodies. In recent experiments, superhydrophobic magnetized molecules have been used as remotely controllable systems.

(a) Schematic illustration of the formation of the plug. (b) Picture of the magnetic plug in an open surface PMMA channel. (c) Picture showing two different color liquids on both sides of the plug, demonstrating the absence of leaking or diffusion after partition. (d) Picture of the open channel after withdrawing some liquid from the right side partition. (e) Pictures of the partition of phenolphthalein (pH indicator) and NaOH solutions by the magnetic plug. (f) Pictures of the removal of the plug by removing the MF, observing the mixing of the two partitions, visually indicated by the formation of the purple/pink color of the phenolphthalein in alkaline media. © Gunatilake, U. B., Morales, R., Basabe-Desmonts, L., & Benito-Lopez, F. (2022)
Salient Features of the Study
This work introduces a unique minimal surface tension superhydrophobic magnetized particle complex termed the magneto twister, which is a robust, magnetically-adjustable and moveable, inverted, cone-shaped, solid–water interface in the form of a cyclone twister.
The magneto twister was completely described to understand the system's features and limits. This device's usefulness was demonstrated for three separate possible microtechnology uses.
To begin, the magnetic twister was utilized to manage drops of water in a water setting by simply putting a water drop over the twister and transferring the drop by shifting the imposed MF, hence allowing water droplet transportation in aqueous environments.
Because of the magnetic twister's durability, the drops were managed in a steady way, which is a typical challenge in magnetized liquid marble particle arrangements.
The magnetic twister was also used as a magnetic stopper to segregate fluids inside an exposed surface route. It was also used to gather and eliminate buoyant microscale plastics from the water's surface by simply pushing the magneto-twister in the direction of the microplastic to capture and then eliminate it using magnetic guidance.
This study paves the door for the use of superhydrophobic magnetized nanoparticles in water–air interface-assisted systems.
Reference
Gunatilake, U. B., Morales, R., Basabe-Desmonts, L., & Benito-Lopez, F. (2022). Magneto Twister: Magneto Deformation of the Water–Air Interface by a Superhydrophobic Magnetic Nanoparticle Layer. Langmuir. Available at: https://pubs.acs.org/doi/10.1021/acs.langmuir.1c02925
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.