Image Credit: University of Surrey.
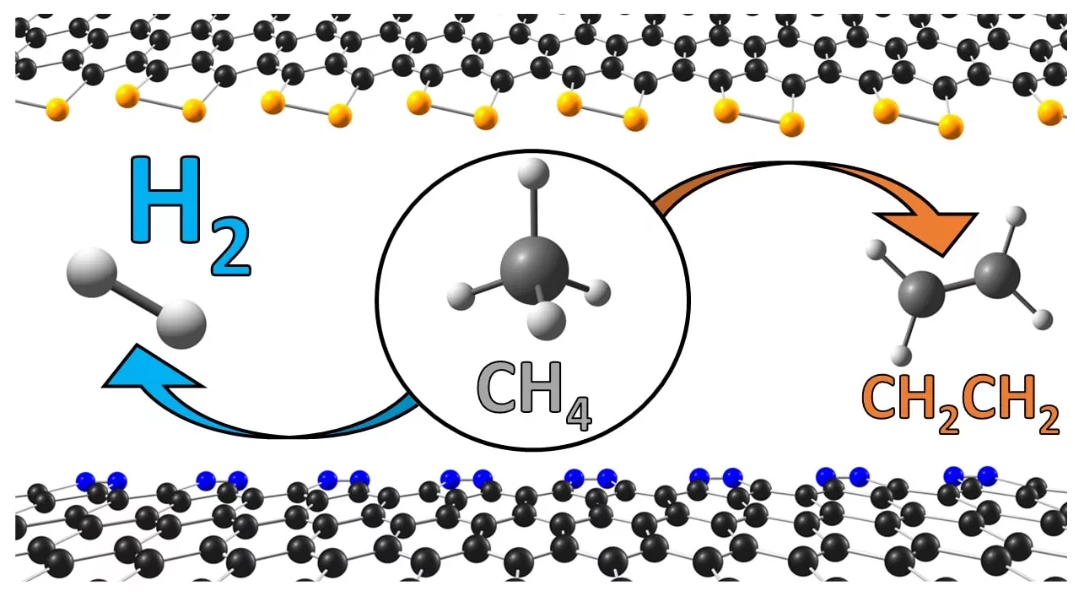
The study has demonstrated favorable results for applying edge-decorated nano carbons as metal-free catalysts for the instant conversion of methane, which is also a robust greenhouse gas, into hydrogen.
Among the nano carbons explored, nitrogen-doped nano carbons presented the maximum level of performance for hydrogen production at elevated temperatures.
Importantly, the scientists also discovered that the nitrogen-doped and phosphorous-doped nano carbons had powerful resistance to carbon poisoning, a typical problem with catalysts in this process.
Our results suggest that using edge-decorated nano carbons as catalysts could be a game-changer for the hydrogen industry, offering a cost-effective and sustainable alternative to traditional metal catalysts. At the same time, this process gets rid of methane, which is a fossil fuel involved in global warming.
Dr Neubi Xavier Jr, Research Fellow, University of Surrey
Dr. Neubi Xavier Jr performed the material science simulations for this study.
Hydrogen fuel is a source of clean and renewable energy that has the potential to decrease carbon emissions and reduce reliance on fossil fuels. Hydrogen can drive vehicles, heat buildings, and produce electricity when employed as a fuel. The sole by-product of hydrogen fuel is water vapor, ensuring it is an eco-friendly option to conventional fossil fuels.
However, the manufacture of hydrogen fuel is dependent on fossil fuels, which cause carbon emissions during the making, and metal catalysts, which require excessive energy for mining and manufacturing, can negatively impact the environment.
Formulating sustainable hydrogen production techniques and catalytic materials is vital to comprehending the complete potential of hydrogen fuel as a source of clean energy.
The study was carried out by a team headed by Dr. Marco Sacchi from the University of Surrey, an expert in sustainable energy and computational chemistry, who integrated thermodynamics, quantum chemistry, and chemical kinetics to establish the most successful edge decoration for hydrogen fabrication.
One of the biggest challenges with catalysts for hydrogen production is that they can get poisoned by carbon. But our study found that nitrogen and phosphorous-doped nano carbons are pretty resistant to this problem. This is a huge step forward for sustainable hydrogen production.
Dr Marco Sacchi, Study Lead Author, University of Surrey
Journal Reference
Xavier Jr, N. F., et al. (2023) First-Principles Microkinetic Modeling Unravelling the Performance of Edge-Decorated Nanocarbons for Hydrogen Production from Methane. ACS Applied Materials & Interfaces. doi.org/10.1021/acsami.2c20937.