Oct 24 2014
Successful techniques for cryopreserving bulk biomaterials and organ systems would transform current approaches to transplantation and regenerative medicine. However, while vitrified cryopreservation holds great promise, practical application has been limited to smaller systems (cells and thin tissues) due to diffusive heat and mass transfer limitations, which are typically manifested as devitrification and cracking failures during thaw.
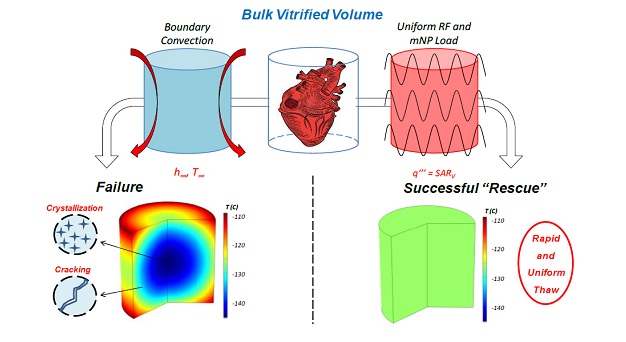 Rapid and uniform heating of magnetic nanoparticles provides a new approach for rewarming bulk, vitrified biomaterial systems that is much less susceptible to the typical failures encountered with traditional thawing approaches (including cracking and devitrification).
Rapid and uniform heating of magnetic nanoparticles provides a new approach for rewarming bulk, vitrified biomaterial systems that is much less susceptible to the typical failures encountered with traditional thawing approaches (including cracking and devitrification).
Here we leverage a clinically proven technology platform, in magnetically heated nanoparticles, to overcome this major hurdle limiting further advancement in the field of cryopreservation. The excitation fields used (alternating magnetic fields at hundreds of kHz) are relatively transparent to biological tissues, but generate significant heating in mNPs, which can be distributed throughout macro- and microscopic tissue structures. The homogeneity in heating then depends on the mNP distribution, which we show here can accommodate unloaded sections up to several millimeters in dimension, while still distributing heat effectively enough to avoid devitrification (i.e. crystallization) and cracking. One major advantage is also the tunability of the heating rate, where the applied field can be adjusted to better control rewarming protocols (e.g. annealing to reduce thermal stresses around the glass transition). Finally, we show CT imaging can be used as a planning and quality control tool by measuring density changes imparted by the cryoprotectants, mNPs, and phase change(s) during processing of the biomaterials.
Greg Fahy, Ph.D. a cryobiologist and Vice President and Chief Scientific Officer at Twenty-First Century Medicine, Inc. an expert in organ cryopreservation by vitrification, says, "This is a very welcome engineering contribution to the problem of more rapidly warming large cryopreserved organs and opens up important new opportunities for exploration. " In addition, Michael Taylor, Ph.D., an expert cryobiologist, Adjunct Professor at Carnegie Mellon and Vice President of Cell and Tissue Systems says, "Avoiding ice formation and the associated tissue injury during cryopreservation of bulky biomaterials has led to a focus on vitrification (vitreous "ice-free" cryopreservation) in recent times. While this approach has been successfully applied to small cell aggregates and thin tissues, extrapolation to larger bulk tissues of a clinically relevant size has thus far failed due principally to the challenges of uniform cooling and heating at rates that promote and stabilize the vitreous state. Progress in developing appropriate cooling technologies to achieve vitrification has advanced more rapidly than complimentary warming techniques, which now present the principal stumbling block. The introduction of radiofrequency excited magnetic nanoparticles offers a highly encouraging path forward and conceptually the only feasible approach on the horizon for achieving uniform warming of bulky vitrified systems."
While these proof-of-principle experiments and modeling clearly demonstrate the physical potential of this new approach, continuing work plans to establish these capabilities in scaled biological systems, working towards the ultimate goal of bulk tissue systems and clinical application. This includes heating in larger bulk systems and investigating both the mNP and tissue interactions at higher field intensities. In addition, one critical area of study not directly addressed here is the perfusion and distribution of the mNPs in tissue systems. Mesoporous silica coatings have been shown to provide stability against aggregation and biocompatibility and we are leveraging this towards an optimized nanoparticle platform for cryopreservation applications.
Co-authors for this work include - Michael L. Etheridge, Ph.D., Jeunghwan Choi, Ph.D., and John C. Bischof, Ph.D., from the University of Minnesota - Twin Cities; Leoni Rott and Birgit Glasmacher, Ph.D., from the Institute for Multiphase Processes, Leibniz Universitaet Hannover; and Yi Xu, Ph.D., from the University of Shanghai for Science and Technology.
This work was supported by the University of Minnesota (MN Futures and Institute for Engineering Medicine Seed Grants), the NSF/CBET (1066343 and 1133285), and the Alexander von Humboldt Foundation. Leoni Rott and Dr. Yi Xu also received funding from the Dr. J¨¹rgen und Imgrad Ulderup Stiftung (foundation) and the Science and Technology Commission of Shanghai Municipality (13ZR1428600), respectively. We would like to acknowledge our collaboration with the Institute for Multiphase Processes at Leibniz Universit?t Hannover for previous CT imaging studies that supported this work and Leoni Rott's Master's research on this topic. Micro-CT imaging was carried out in the Minnesota Dental Research Center for Biomaterials and Biomechanics under the guidance of Dr. Alex Fok and Yung-Chung Chen; with additional consultation from Dr. Brian Bagley. The authors would finally like to acknowledge: Dr. Yoed Rabin (Carnegie Mellon) for his input on the manuscript; Steven Jackson for help in developing the cooling protocol; and Dushyant Mehra for assistance with the DSC measurements.