Jun 12 2017
An innovative technique for enhancing the functionality of next-generation molecular electronic devices by making use of graphene has been developed by an international research team headed by the University of Bern and the National Physical Laboratory (NPL).
The outcomes of the research can be used to design smaller devices with greater performance that can be used in different applications such as flexible electronics, molecular sensing and energy conversion and storage, and also robust measurement systems for resistance standards.
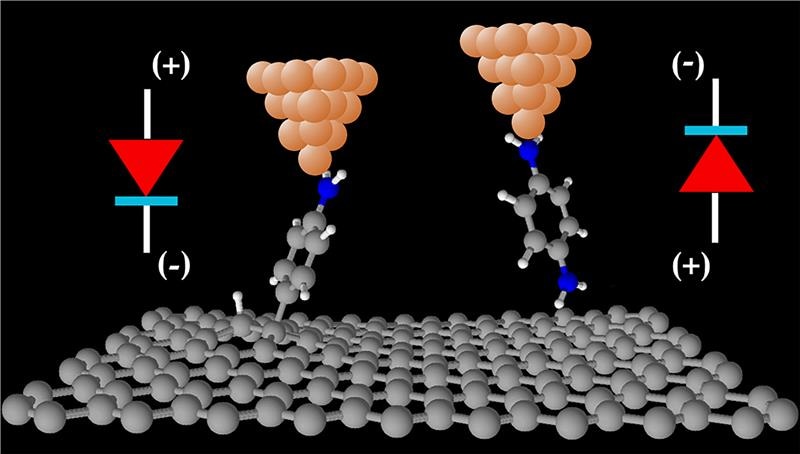 The Researchers performed the characterization of graphene-based molecular electronic devices at room-temperature and demonstrated that molecules covalently attached to mechanically robust graphene substrates are ideal candidates for next-generation molecular electronic devices. CREDIT: Alexander Rudnev, University of Bern.
The Researchers performed the characterization of graphene-based molecular electronic devices at room-temperature and demonstrated that molecules covalently attached to mechanically robust graphene substrates are ideal candidates for next-generation molecular electronic devices. CREDIT: Alexander Rudnev, University of Bern.
The goal of a number of Researchers working in the field of nanoscale molecular electronics is to construct electronic devices by using individual molecules as the fundamental units, so that the functionality is enhanced and developers can accomplish an unparalleled level of control and device miniaturization. The main hurdle to this advancement is the lack of stable contacts between the metals and molecules used that have the ability to function at room temperature and generate reproducible outcomes. In addition to its exceptional mechanical stability, graphene also has considerably higher thermal and electronic conductive characteristics, making it very propitious for different prospective applications in molecular electronics.
An experimentalist group at the University of Bern and theoreticians from NPL (UK) and the University of the Basque Country (UPV/EHU, Spain) has worked in collaboration with Researchers from Chuo University (Japan) to show the stability of multi-layer graphene-based molecular electronic devices down to the single molecule limit. The outcomes of the research have been reported in the Science Advances journal and prove to be a significant step forward in developing graphene-based molecular electronics, wherein the reproducible characteristics of covalent contacts between the molecules and graphene overcome the drawbacks of the prevalent modern technologies based on coinage metals, even under ambient conditions.
Connecting single molecules
When particular molecules are adsorbed on graphene-based electronic devices, the device functionality can be enhanced, chiefly by changing its electrical resistance. Yet, it is not easy to correlate the gross device characteristics with the characteristics of the adsorbed individual molecules. This is because averaged quantities are not adequate to identify propitiously greater discrepancies across the surface of graphene.
Dr. Alexander Rudnev and Dr. Veerabhadrarao Kaliginedi at the Department of Chemistry and Biochemistry of the University of Bern measured the electric current that flows though single molecules linked to graphite or multi-layered graphene electrodes. For this, they used a distinctive low-noise experimental method, which enabled them to resolve the molecule-to-molecule discrepancies. The Researchers made use of theoretical calculations of Dr. Ivan Rungger (NPL) and Dr. Andrea Droghetti (UPV/EHU) to show that discrepancies on the graphite surface are very minute and that the properties of the chemical contact of a molecule with the top graphene layer determines the functionality of single-molecule electronic devices.
“We find that by carefully designing the chemical contact of molecules to graphene-based materials, we can tune their functionality,” stated Dr. Rungger. “Our single-molecule diodes showed that the rectification direction of electric current can be indeed switched by changing the nature of chemical contact of each molecule,” stated Dr. Rudnev.
We are confident that our findings represent a significant step towards the practical exploitation of molecular electronic devices, and we expect a significant change in the research field direction following our path of room-temperature stable chemical bonding.
Dr. Veerabhadrarao Kaliginedi, The Department of Chemistry and Biochemistry, The University of Bern
The outcomes of the study will also assist Scientists focusing on electro-catalysis and energy conversion research design graphene/molecule interfaces in their experimental systems to enhance the efficiency of the device or catalyst.