Dec 11 2017
Minute pores at the opening to a cell can function as tiny bouncers that let in certain electrically charged atoms, that is, ions, but obstruct others. These “ion channels” function as acutely sensitive filters and have a significant role in biological functions such as firing of brain cells and muscle contraction.
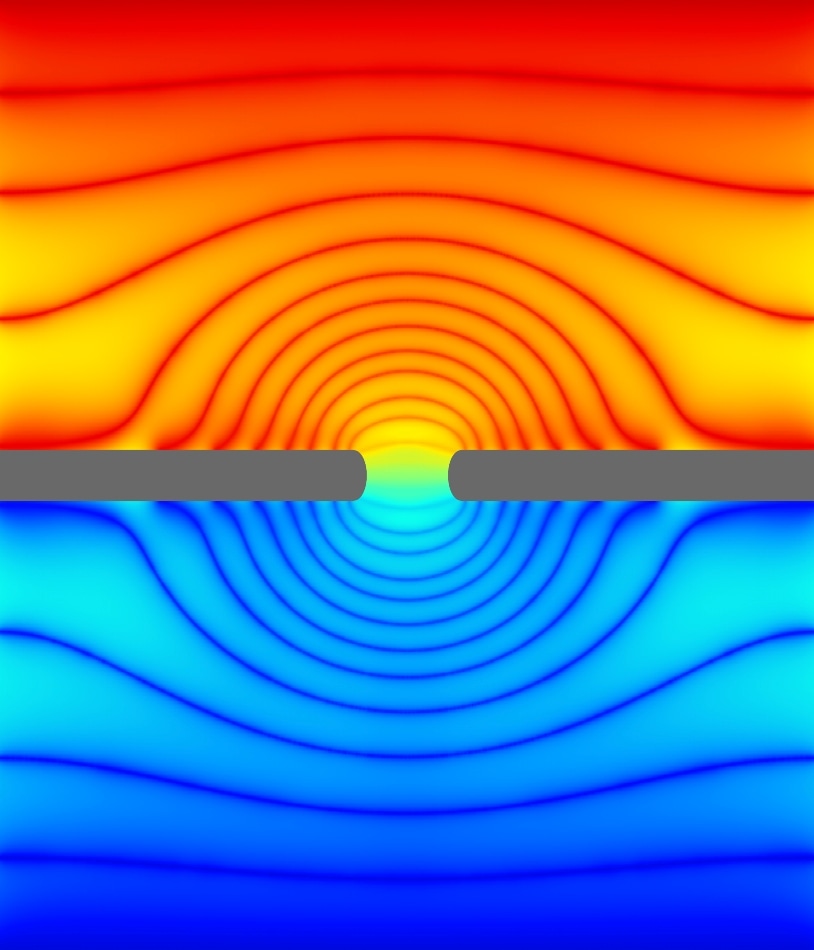 In this simulation, a biological membrane (gray) with an ion channel (center) is immersed in a solution of water and ions. This cross section of a simulation “box” shows the electric potential, the externally supplied “force” that drives ions through the channel. A dazzling pattern emerges in this potential due to the presence of the channel—the colors show the lines of equal potential. The slowly decaying nature of this pattern in space makes simulations difficult. The golden aspect ratio—the chosen ratio of height to width of this box—allows for small simulations to effectively capture the effect of the large spatial dimensions of the experiment. CREDIT: NIST.
In this simulation, a biological membrane (gray) with an ion channel (center) is immersed in a solution of water and ions. This cross section of a simulation “box” shows the electric potential, the externally supplied “force” that drives ions through the channel. A dazzling pattern emerges in this potential due to the presence of the channel—the colors show the lines of equal potential. The slowly decaying nature of this pattern in space makes simulations difficult. The golden aspect ratio—the chosen ratio of height to width of this box—allows for small simulations to effectively capture the effect of the large spatial dimensions of the experiment. CREDIT: NIST.
In order for the right ions to be quickly transferred through the cell membrane, the minute channels depend on a complicated interplay between the ions and their surrounding molecules—specifically water—that have an attraction for the charged atoms. However, these molecular processes have conventionally been challenging to model, and hence to perceive, by means of computers or artificial structures.
At present, scientists from the National Institute of Standards and Technology (NIST) and their collaborators have shown that nanoscale pores formed in layers of graphene (atomically thin carbon sheets well known for their conductivity and strength) can offer an uncomplicated model for the complicated functioning of ion channels. This model enables researchers to evaluate a number of characteristics in relation to the transport of ions. Moreover, graphene nanopores might eventually become viable with effective mechanical filters appropriate for processes such as recognizing defective DNA in genetic material and eliminating salt from oceanic water.
Michael Zwolak, an NIST researcher, collaborated with Subin Sahu—jointly affiliated with NIST, the University of Maryland NanoCenter, and Oregon State University—to find out an innovative technique to mimic specific features of ion channel behavior, at the same time explaining computationally expansive details such as molecular-scale variations in the shape or size of the channel.
To be able to enter into the ion channel of a cell, which is a protein assemblage including a pore with a width of merely few atoms, ions have to lose a portion of or the entire water molecules linked with them. By contrast, the energy needed to achieve this is normally restrictive, which is why ions require some additional assistance, which they acquire from the ion channel lined with molecules with charges opposite to specific ions, thereby assisting in attracting them. Furthermore, the arrangement of such charged molecules offers an improved fit for specific ions and not others, thus developing an exceptionally selective filter. For example, negatively charged molecules are lined along specific ion channels, where the molecules are distributed such that they can easily house potassium ions and not sodium ions.
The researchers hope to acquire an in-depth understanding of the selectivity of ion channels not only to understand the way biological systems operate but also because the functioning of these channels might propose a propitious way to engineer non-biological filters for a number of industrial applications.
Zwolak, Sahu, and Massimiliano Di Ventra from the University of California, San Diego, adopted a much uncomplicated system (i.e. graphene nanopores) to mimic the conditions that are similar to the functioning of actual ion channels. For instance, the simulations performed by the researchers, for the first time, showed that nanopores can be rendered to allow only specific ions to pass through them by altering the diameter of the nanopores formed in a single graphene sheet or by adding more sheets. However, in contrast to biological ion channels, this selectivity occurs due to the elimination of just the water molecules, a procedure called dehydration. Graphene nanopores will enable this dehydration-only selectivity to be evaluated under disparate conditions, which is another achievement. The outcomes of the study have been published in the latest issues of Nano Letters and Nanoscale.
In two earlier preprints, Zwolak and Sahu have addressed a portion of the complexity in mimicking constriction and transport of the ions through the nanopore channels. At the time theorists simulate a process, they select a “box” of specific size in which they carry out the simulations. Based on the detail and breadth of the computation, the box may be smaller or bigger. The researchers demonstrated that if the simulation volume dimensions are selected such that the ratio of the height of the volume to its width is of a specified numerical value, the simulation can concurrently record the impact of the surrounding ionic solution and such minute specifications as nanoscale variations in the diameter of the pores or the presence of charged chemical groups. According to Zwolak, this latest finding, which the researchers term “the golden aspect ratio” for simulations, will largely simplify computations and will enable to obtain an in-depth knowledge of the functioning of ion channels.