Apr 9 2018
A nanomaterials expert from Swansea University has been investigating how small gold particles survive when they are subjected to extremely high temperatures.
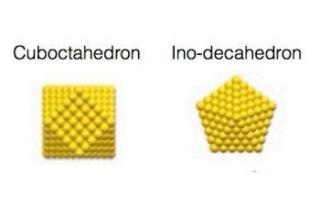 The two alternative architectures of the gold nanoclusters containing 561 atoms. (Image credit: Swansea University)
The two alternative architectures of the gold nanoclusters containing 561 atoms. (Image credit: Swansea University)
The study proves crucial for the engineering sector for certain prospective applications of nanotechnology, for instance, in aerospace and catalysis, where particles with only nanoscale dimensions are subjected to extremely high temperatures.
The outcomes of the research—which was a three-way collaboration between Swansea University, the University of Birmingham, and the University of Genoa—were reported in the Nature Communications journal this week. The research demonstrated that gold nanoparticles with a precisely chosen size of 561 ± 14 atoms prove strikingly rugged against aggregation and diffusion, although there is no change in their internal atomic arrangements.
The scientists subjected an array of size-selected gold nanoparticles, or clusters, to temperatures as high as 500 °C by using an ultrastable, variable-temperature stage in an aberration-corrected scanning transmission electron microscope and imaged them with atomic resolution. The particles were deposited from a nanoparticle source onto thin carbon or silicon nitride films.
The Two Alternative Architectures of the Gold Nanoclusters Containing 561 Atoms
The experiments indicated that when the gold nanoparticles were bound to the surface, at point defects, they were fixed strong enough, even at extreme temperatures. However, during the heat treatment, there were variations in the atomic structures of the clusters, which alternated to and fro between two main atomic configurations, or “isomers”. One of the configurations was a face-centered cubic structure, analogous to a small piece of bulk gold, and the other was a decahedral arrangement with a symmetry forbidden in an extended crystal. The scientists could even measure the minute difference in energy, of merely 40 meV, between the two different atomic structures.
These advanced experiments have allowed us to make a new measurement for nanoparticles deposited on a surface - the difference in energy between two competing atomic arrangements. It’s something that the people who use computers to calculate the properties of nanomaterials are particularly excited about, a kind of reference point if you like. And the images show that our little nanoparticles are really rather tough creatures, which bodes rather well for their applications in future industrial manufacturing.
Professor Richard Palmer, Head of the Nanomaterials Lab in Swansea University’s College of Engineering
The focus of the study carried out at Swansea Lab is to increase the production of these nanoparticles by 10 million times to the level of grams, and much more. Professor Palmer stated that “We need very small things in very large numbers to realise the true potential of nanotechnology.”