May 24 2018
The conventional techniques and materials used for fabricating advanced integrated circuits are heading toward, or have likely already reached, their utmost physical restrictions with respect to the size of the final product.
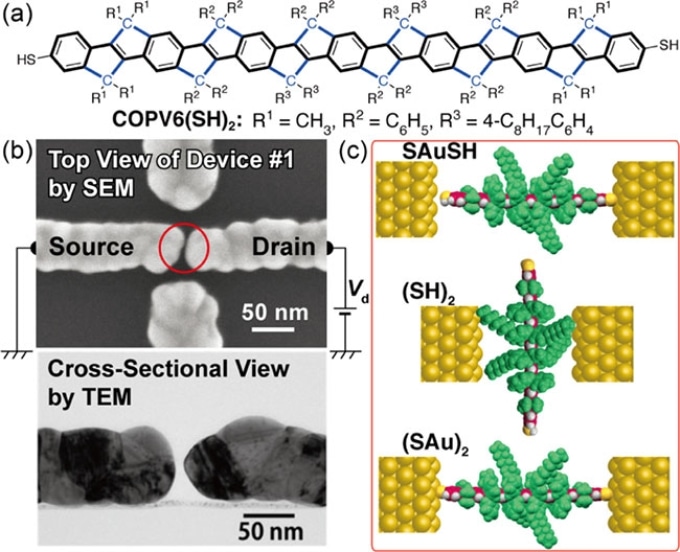 The COPV6(SH)2 molecule and how it binds with the ELGP nanogap. (a) Molecular structure of the COPV6(SH)2 molecule. (b) Top and cross-sectional views of an ELGP nanogap electrode. (c) Different ways in which the molecule binds with the nanogap. The thermally stable device is obtained when the first type of binding (SAuSH) occurs. (Image credit: Tokyo Institute of Technology)
The COPV6(SH)2 molecule and how it binds with the ELGP nanogap. (a) Molecular structure of the COPV6(SH)2 molecule. (b) Top and cross-sectional views of an ELGP nanogap electrode. (c) Different ways in which the molecule binds with the nanogap. The thermally stable device is obtained when the first type of binding (SAuSH) occurs. (Image credit: Tokyo Institute of Technology)
Put differently, it is almost impossible to achieve further miniaturization of electronic devices without examining other kinds of materials and technology, for instance, organic molecular electronic devices. Yet, in general, this category of devices functions well only at very low temperatures due to the thermal fluctuations of the organic molecules as well as the metal electrodes.
Although special electroless gold-plated nanogap electrodes, known as ELGP electrodes, have exhibited excellent thermal stability at their gap, it is most important to develop innovative categories of molecular wires to overcome the problem mentioned above. To achieve this, a research team, including Professor Yutaka Majima from Tokyo Tech, worked with a 4.5-nm-long molecule known as disulfanyl carbon-bridged oligo-(phenylenevinylene), or COPV6(SH)2.
This molecule includes a rigid rod-like pi-conjugated arrangement, in which four 4-octylphenyl groups isolate it spatially and electronically from its surrounding. The two sulfhydryl terminals present in the molecule may or may not bind chemically with the opposing gold surfaces in an ELGP nanogap. Fascinatingly, the scientists discovered that if the COPV6(SH)2 molecule binds with gold surfaces in a particular manner, termed SAuSH, the ensuing device exhibits the attributive behavior of coherent resonant electron-tunneling devices, which find a range of prospective applications in electronics and nanotechnological fields.
Most significantly, it was found that the ensuing device was thermally stable, exhibiting similar current versus voltage curves at 9 and 300 K. This could not be realized earlier when flexible organic molecular wires were used. Moreover, the COPV6(SH)2 molecule binds at the ELGP nanogap in multiple ways, and the researchers are now clueless how to control the type of device that they obtain.
In spite of all this, they were successful in measuring the electrical properties of the devices they got to elucidate in detail the hidden quantum mechanisms that govern their behavior. Moreover, they corroborated their outcomes with theoretically deduced values, and through this, they gained further insights into the operating principle of the SAuSH device and the other probable configurations.
The subsequent step would be to realize a better yield of the SAuSH device since their yield was less than 1%. The researchers are of the notion that the high molecular weight and rigidity of the molecule, and also the stability of ELGP electrodes, would govern the high stability of the ensuing device and its low yield. As several probable variations of the COPVn category of molecules and several ELGP nanogap configurations exist, the challenge in obtaining a better yield might be overcome through adjustments in the techniques and the properties of the molecules and gaps used. The data reported in this study will serve as the basis for forthcoming molecular-scale electronic studies.