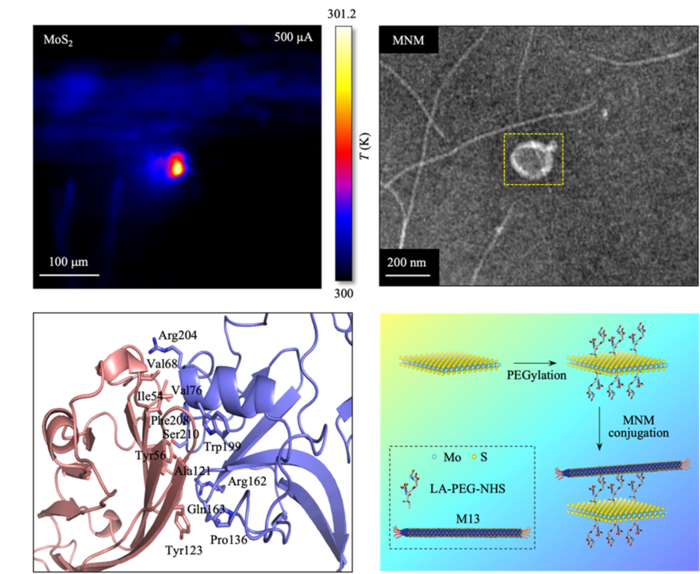
Top left, Thermograph of the sample upon application of electrical signal; Top right, Transmission electron microscopy image of MNM; Bottom left, Binding interface of the virus-cancer cell protein structure; Bottom right, Schematic diagram of the composition and process used to construct the MNM. Image Credit: SUTD
Two-dimensional (2D) materials are injected into cancer cells during electrothermal therapy, and the cells are then subjected to electrical currents. As a result, the materials heat up and destroy nearby cancer cells.
However, weak cancer cell ablation can lead to the failure of conventional electro-thermal therapy using 2D materials. The limited number of materials formed on the cancer cells and the modest Joule heating produced in the material are to be blamed for this.
To address these problems, the researchers created a hybrid nanomaterial called a MoS2 Nanostructure with the M13 virus (the authors call it MNM) by depositing the M13 virus on layered materials made of molybdenum disulfide (MoS2). In addition, polyethylene glycol (PEG) was used to modify the nanomaterial surfaces to increase biocompatibility.
The M13 virus insertion enhances the effectiveness of electro-thermal therapy. Due to the enhanced selectivity of the M13 virus’s attachment to cancer cells, more MNM assembles on the cancer cells compared to conventional 2D materials. Strong Joule heating is also produced as a result of the MoS2 material’s high electrical conductivity.
As a result, more heat is generated within the nanomaterials, which could then be used to destroy more cancer cells. For instance, the MNM nanosystem can reduce the proportion of cancer cells by 23%, which is almost twice as much as what is possible with the current generation of thermal-based therapy nanosystems.
For many years, cancer eradication has been the dream of cancer patients and researchers. Eliminating recurrence and metastasis of the cancer in the body is of vital importance. However, a single conventional therapy is unable to eradicate cancer cells completely, due to the diversity, heterogeneity, and complexity of the cancer cells. This is why we aimed to design a simple nanosystem or nanomaterial to synergistically eradicate and treat cancer cells.
Desmond Loke, Study Lead Investigator and Assistant Professor, Singapore University of Technology and Design
Yaw-Sing Tan from the Bioinformatics Institute, Agency for Science, Technology, and Research (A*STAR), Wey-Chyi Teoh from Changi General Hospital, Denise Lee, J. Shamita Naikar, Shao-Xiang Go, and Natasa Bajalovic from SUTD were the other researchers that participated in this study.
Journal Reference
Meivita, M. P., et al. (2023) An Efficient, Short Stimulus PANC-1 Cancer Cell Ablation and Electrothermal Therapy Driven by Hydrophobic Interactions. Pharmaceutics. doi:10.3390/pharmaceutics15010106