Jørgen Kjems and collaborators have achieved a remarkable breakthrough in developing tiny nano-sized pores that can contribute to better possibilities for, among other things, detecting diseases at an earlier stage. Image Credit. ACS Nano 2023, 17, 10, 9167-9177
By utilizing small nanopores and nanobodies, they have determined markers that are indicators of COVID-19 and breast cancer with impressive sensitivity and accuracy. With the availability of this technology, the future might hold quick and precise disease diagnosis using a simple blood test. The breakthrough could take us a step ahead to enhanced healthcare.
In a partnership with Groningen University, Professor Jørgen Kjems and his research group at Aarhus University have obtained an exceptional breakthrough with tiny nano-sized pores that could create better possibilities for, among other things, detecting diseases at an earlier stage.
Their study, recently reported in the scientific journal ACS Nano, displays a new innovative method for identifying particular proteins in complicated biological fluids, like blood, without having to label the proteins chemically. The study is considered to be a significant milestone in nanopore technology and could also revolutionize medical diagnostics.
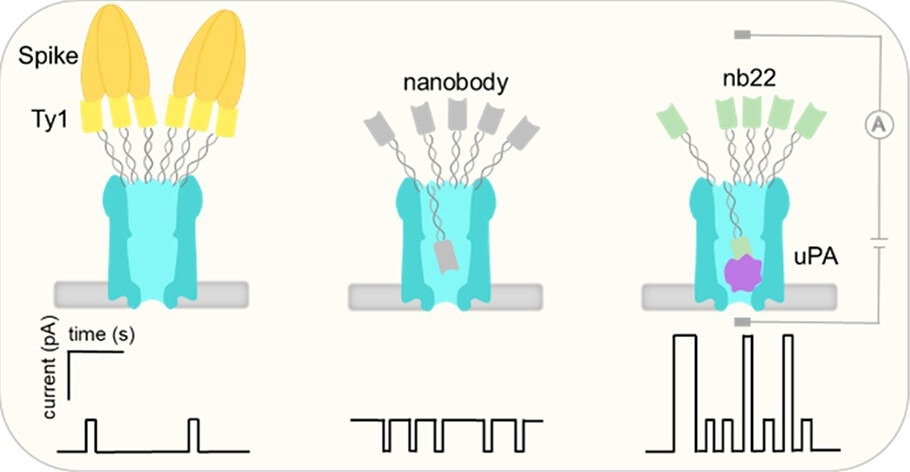
Nanopores are small channels formed in materials that could be utilized as sensors. The scientists, headed by Jørgen Kjems and Giovanni Maglia (Groningen Univ.), have taken this a step ahead by coming up with a unique kind of nanopore known as ClyA with scanner molecules, known as nanobodies, fixed to it.
Such nanobodies, derived from antibodies, have the potential to identify various proteins with surprising precision. In this study, the scientists fixed nanobodies to ClyA with the help of a DNA adapter. By utilizing a range of nanobodies, they were able to make several different nanopore sensors, which could detect a range of proteins of different sizes.
The research group made nanopores with specialized nanobodies fixed, which have the potential to detect the Spike protein of SARS-CoV-2 (the virus that results in COVID-19) and a protein marker for breast cancer known as urokinase-type plasminogen activator (uPA), respectively.
By quantifying variations in electrical currents that are attributable to the existence of such proteins, the researchers could find and determine separate proteins and even identify their concentrations.
What makes this discovery highly exceptional is that the nanopores remained highly sensitive and precise even when tested with complicated samples like blood.
Even though the nanopores are invisible to the naked eye, the importance of this study is palpable. Already, the present technologies enabled the combination of nanopores into a portable device that has the potential to make use of the nanopores’ ability to scan liquids for particular molecules.
Hence, it has the potential to envision a future where patients can quickly and precisely detect diseases like infectious diseases or cancer with a simple blood test. This may result in earlier interventions, enhanced treatment outcomes, and improved healthcare on the whole.
Even though additional studies and validation are required before this technology turns out to be extensively available, the partnership between these two universities takes us one step ahead of this reality. The discovery exemplifies the power of scientific collaboration and innovation in converting healthcare.
Journal Reference
Zhang, X., et al. (2023) Specific Detection of Proteins by a Nanobody-Functionalized Nanopore Sensor. ACS Nano. doi.org/10.1021/acsnano.2c12733.