Dec 9 2008
Using the Canadian Light Source, a team of researchers from Quebec's Laval University and Australia's La Trobe University has discovered how to improve the nanometers-thick layer of polymer used to coat cardiac stents. A stent is a small, springy tube made from stainless steel that is used to open arteries to the heart, that become blocked from atherosclerosis or other forms of heart disease. The findings, published at the end of August in the journal Langmuir, could lead to fewer complications when treating blocked arteries.
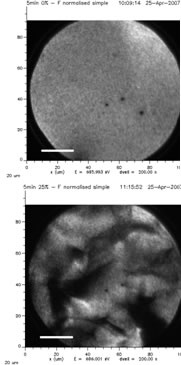 X-ray Photoemission Electron Microscope images of a 35 nanometer-thick stent coating, before (top) and after (bottom) mechanical deformation. Source: Paula Horny and Stephane Turgeon, Laval University.
X-ray Photoemission Electron Microscope images of a 35 nanometer-thick stent coating, before (top) and after (bottom) mechanical deformation. Source: Paula Horny and Stephane Turgeon, Laval University.
Stents have been used to treat heart disease for close to fifteen years. Implanted during angioplasty, they shore up the walls of the newly unclogged artery. But the effectiveness of stents is hampered by their potential involvement in a complication called restenosis, a re-narrowing of the repaired artery due to a buildup of scar tissue.
"We are trying to apply physics and chemistry to improve the clinical performance of medical devices like stents," says Diego Mantovani of Laval's Laboratory for Biomaterials and Bioengineering, and University Hospital Research Centre. "Thirty to forty percent of patients have had the artery reocclude and develop restenosis four, six, eight months after the stent was deployed."
Coating the stents with polymers impregnated with clot-busting drugs can help reduce complications and new blockages. These drug-eluting stents, however, can run into problems: cracks may form in the surface coating because of mechanical deformation caused by the stent's expansion during implantation, with further degradation caused by the body itself. Mantovani's team realized that the cause of the coatings' troubles arose because the polymer was not bonded to the stent itself, but to a thin layer of chromium-rich oxide covering the stent. Fractures in the fragile oxide layer contribute to the cracking of the polymer film.
"Our group started by developing a coating that we hoped would not crack, by resisting deformation, and attach it strongly to the stainless steel in the stent, in place of the oxide layer" explains Mantovani. "We then needed a way to look at what happens to the coated stent after immersion in test solutions that mimic conditions inside the heart in the body."
The research is part of a joint strategic project funded by Canada's Natural Sciences and Engineering Research Council (NSERC), in collaboration with Montreal-based Plasmionique, a company that specializes in manufacturing thin film coatings.
Evaluating the stability and effectiveness of coated stents has been an ongoing challenge for the stent industry since the U.S. Food and Drug Administration identified the issue in 2005. The difficulty stems from a lack of standards and technologies capable of making the coatings and seeing cracks in a layer of material that is only some hundreds of atoms thick. Even with some of the best 'bench top' tools available, Mantovani says his team could not visualize what was going on in their coated stents after deformation.
"When we first looked at samples in our lab we were happy because we didn't find any cracks," says Mantovani. "But the synchrotron's X-PEEM (X-ray photoemission electron microscope) revealed pinhole defects made during the coating's manufacture. We've been able to take what we found and apply it to improving our coating technology."
Professor Mantovani hopes that their new and improved coatings will be available on stents in the next two to five years. He believes that similar coatings can be used on other medical equipment and in applications where a drug or antimicrobial chemical could be embedded in the coating to control the spread of viruses and bacteria. For example, his lab is currently working to put coatings on air filters, to curb the spread of airborne germs on the International Space Station.
"The way to test these kinds of coatings, improve the polymers and see how they bond with their substrate is now clear," says Mantovani. "With the light source, there are no limits other than our time and energy to carry out this kind of analysis."
Reference: P. Hale, S. Turgeon, P. Horny, F. Lewis, N. Brack, G. Van Riessen, P. Pigram, D. Mantovani 2008. Langmuir 24 (15), pp. 7897-7905. DOI: 10.1021/la8002788.